* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Get
Psychoneuroimmunology wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Lymphopoiesis wikipedia , lookup
Immune system wikipedia , lookup
Major histocompatibility complex wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Adaptive immune system wikipedia , lookup
Innate immune system wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Molecular mimicry wikipedia , lookup
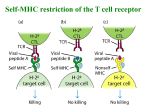
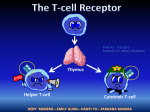
Why transplant? “Cure for organ specific disease” (or islets since 2000) Adapted from “Immunology” by Goldsby, Kindt, Osborne, Kuby ISOGRAFT AUTOGRAFT ALLOGRAFT XENOGRAFT (Experimental) ISOGRAFT AUTOGRAFT ALLOGRAFT POTENT IMMUNE RESPONSE XENOGRAFT (Experimental) Immune rejection of organ transplants is a major barrier to success 3 manifestations defined by when and how occur -Hyperacute -Acute -Chronic Hyperacute rejection - within hours - Ab mediated Adapted from “Immunology” by Goldsby, Kindt, Osborne, Kuby Problematic pre-existing antibodies and how to deal with these Allotransplants -natural antibodies to ABO blood group antigens -anti-HLA antibodies raised during previous transfusion, transplant or pregnancy Solution: test recipient serum for ABO compatibility and negative crossmatch Xenotransplants -natural antibodies to Gala1-3Gal epitope present in lower mammals Solution: agalactosyl transferase knockout pig Acute rejection - within weeks Adapted from “Immunology” by Goldsby, Kindt, Osborne, Kuby T cells play a central role in acute rejection Adapted from “Immunology” by Goldsby, Kindt, Osborne, Kuby Induction in draining lymph node T cells are activated by 2 pathways Host T cell Host DC Indirect (classical antigen presentation by host DC) Transplant-derived peptide on host MHC/HLA interacts with host T cell receptor Response is weak for allografts but strong for xenografts Transplant Host T cell Direct (antigen presentation by passenger donor DC) Donor DC Transplant-derived peptide on donor MHC/HLA or donor MHC/HLA alone interacts with host T cell receptor Response is strong for allografts but weak for xenografts Destruction by various effectors at graft site DC allo xeno Adapted from “Immunology” by Goldsby et al Solutions to acute rejection MHC match Serologic methods Cellular assays Molecular biological methods Critical for bone marrow and helpful for kidney, but limited organ availability and highly polymorphic MHC means often not practical. No MHC match for xenografts Immunosuppress recipient Cocktail of drugs with 3 distinct modes of action Actions of immunosuppressive drugs 1. Inhibit T cell signaling e.g. CyclosporinA or FK-506 block calcineurin activity and thereby IL2 synthesis. 2. Anti-proliferative e.g. Azathioprine or mycophenolic acid inhibit synthesis of purines required for cell division. Inhibit B and T cell proliferation. 3. Anti-inflammatory e.g. Corticosteroids bind to intracellular steroid receptors and thereby regulate transcription of a number of genes including cytokines, adhesion molecules and class II molecules. Chronic rejection - months or years after grafting Poorly understood combination of immunological + other factors Damage at time of grafting + acute rejection episodes + ongoing indirect response Pascual et al, N. Engl. J. Med. 346:580 (2002) result in low grade damage to graft vascular endothelium, smooth muscle proliferation and migration leading to vascular occlusion Solution: No effective immunosuppressive regimen to date, but predict better control of the immune response will decrease incidence of chronic rejection. Organ transplantation is a life saving procedure, but there are major problems…… Problem 1 Systemic immunosuppression facilitates graft survival at a price - drug targets are not limited to the immune system resulting in toxicity for other organs -systemic immunosuppression increases susceptibility to cancer and infection Possible solutions under investigation -more specific drugs? (several in clinical trials) -induce graft tolerance? (“Holy Grail”) -local immunosuppression? (Our approach) Graft tolerance by chance and deduction Transplant girl's blood change a 'miracle' The Sydney Morning Herald. January 25, 2008 N ENGL J MED 358: 369-374 (2008). Case history •Hepatitis/liver failure •Profound lymphopenia O- 9 Y.O. liver graft O+ 12 Y.O. Immunosuppression O+ No immunosuppression •Predominantly donor white cells but approx. 2%host B cells •IgG on RBC progressing to active hemolysis O+ •Donor liver survival •Loss of pre-existing immunity/successful reimmunization •TCR excision circles How was tolerance established ? Presumed events resulting in graft tolerance •Hepatitis/liver failure •Profound lymphopenia Donor leukocytes/stem cells not rejected + space for expansion O- 9 Y.O. liver graft O+ 12 Y.O. Immunosuppression O+ No immunosuppression •Predominantly donor white cells but approx. 2%host B cells •IgG on RBC progressing to active hemolysis O+ •Donor liver survival •Loss of pre-existing immunity/successful reimmunization •TCR excision circles Presumed events contributing to graft tolerance •Hepatitis/liver failure •Profound lymphopenia O- 9 Y.O. liver graft O+ 12 Y.O. Donor leukocytes/stem cells not rejected + space for expansion Immunosuppression O+ No immunosuppression •Predominantly donor white cells but approx. 2%host B cells •IgG on RBC progressing to active hemolysis Donor leukocytes outgrow host leukocytes but residual host B cells make antibody to donor red blood cells O+ •Donor liver survival •Loss of pre-existing immunity/successful reimmunization •TCR excision circles Aggressive host B cells rejected by donor T cells upon withdrawal of immunosuppression Presumed events contributing to graft tolerance •Hepatitis/liver failure •Profound lymphopenia O- 9 Y.O. Immunosuppression Active thymus liver graft O+ 12 Y.O. Stem cells O+ No immunosuppression •Predominantly donor white cells but approx. 2%host B cells •IgG on RBC progressing to active hemolysis O+ •Donor liver survival •Loss of pre-existing immunity/successful reimmunization •TCR excision circles Thymic engraftment Donor cells educated to be tolerant to host and donor but responsive to foreign antigen Thymic education T cell precursor T cell receptor gene rearrangement TCR+ Immature thymocyte TCR+ TCR+ Positive selection of cells whose T cell receptor bind MHC Epithelial cell TCR+ TCR+ Death of cells that do not interact with MHC TCR+ TCR+ TCR+ Mature T cells tolerant of self into circulation TCR+ DC Negative selection and death of high affinity self reactive cells TCR+ Thymic education in mixed chimera T cell precursor T cell receptor gene rearrangement TCR+ TCR+ TCR+ TCR+ Positive selection of cells whose T cell receptor bind MHC Epithelial cell Immature thymocyte TCR+ TCR+ TCR+ TCR+ Death of cells that do not interact with MHC TCR+ TCR+ TCR+ TCR+ TCR+ TCR+ TCR+ Mature T cells Tolerant of host and donor into circulation TCR+ DC DC TCR+ TCR+ TCR+ TCR+ Negative selection and death of high affinity host-reactive and donor-reactive cells Tolerance is ideal, but for most patients the risks are too great. What else can we do….. Local immunosuppression Engineer graft to produce its own immunomodulatory molecules thus concentrating immunosuppression in the graft microenvironment “Helping the graft help itself” Type 1 diabetes Autoimmune destruction of insulin producing beta cells Usually strikes in childhood or early adulthood Affects over 140,000 Australians Up to 6 insulin injections each day for rest of life Parenteral insulin is not good enough Quic kTime™ and a Photo - JPEG decompress or are needed to see this pic ture. Adapted from Pancreatic islet transplantation is a cure for Type 1 Diabetes Somatostatin ( cell) Pancreatic polypeptide (PP cell) Insulin cell Glucagon a cell Adapted from seungkimlab.stanford.edu/ islet.html Islet transplantation www.jdrf-hms-islets.org Westmead Hospital Sydney portal vein infusion of islets into the liver Generating local immunosuppression at the graft site systemic Local Methods of expressing immunomodulatory molecules in primary tissues Viral Transient eg. adeno or stable eg. lenti Transgenic Stable Reproducible Reproducible Control expression level Select expression level xenotransplants Allo/xenotransplants Immunohistolgy from seungkimlab.stanford.edu/ islet.html Problem 2 Shortage of donor organs Number of Deceased Donors Solid Organ Transplants and Patients on the Waiting List 2002 - 2007 3000 Deceased Donors Transplants Waiting List 2500 2000 1788 1824 Australia 3000 2500 1663 1716 1757 1690 1500 2000 1500 1000 721 634 500 179 206 789 218 736 1000 740 668 500 204 202 198 0 0 2002 2003 2004 2005 2006 2007 Source:ANZOD Registry (www.anzdata.org.au) Donor organ shortage and Type 1 diabetes >140,000 Australians with Type 1 diabetes 198 organ donors in 2007 Xenotransplantation may provide the solution Why pigs? •Reproduce quickly and have large litters •Organs of similar size to humans •Beta cells regulate blood glucose appropriately in humans & pig insulin used for many years in humans •Relatively easy to rear in conditions free of particular pathogens •Can be genetically modified to reduce the risk of immune rejection Why not pigs? •Ethical objections •Religious objections •Risk of xenozoonosis (transmission of infection between species) •Breed free of known pathogens •Breed free of potential pathogens (PERVS) •Long-term monitoring of recipients •Current moratorium in Australia, but experience in approx. 150 people worldwide show no evidence of pig derived infection Pigs can be modified to remove detrimental and add beneficial molecules Remove target epitopes Add modulators of host pathways eg. Gala(1,3)Gal eg. complement modulators eg. anticoagulant molecules Add pro-survival molecules Add local immunosuppression eg. costimulation blockade eg. depleting antibodies