* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Supplemental information
Survey
Document related concepts
Transcript
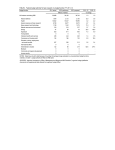
Supplemental information Supplemental Results and Discussion Both PC and SM de novo syntheses are inhibited in Fas signaling. Experiments have been carried out to monitor simultaneously de novo synthesis of PC and SM. PC and SM de novo syntheses were both inhibited upon FasL treatment in a time-dependent manner (Supplemental Fig. 1). Of note, the pool of radiolabelled PC remained 50 to 60-fold higher than that of SM, suggesting that a large quantity of PC was potentially available for SM synthesis (Supplemental Fig. 1). However, since it is unknown how local PC synthesis feeds into SM synthesis in the Golgi, we cannot rule out that the decrease of PC synthesis may account (at least in part) for the decreased SM synthesis. FasL triggered cell death (Supplemental Fig. 2A) and SMS activity inhibition (Supplemental Fig. 2B) in a dose-dependent manner, those two events being highly correlated (R2=0.94). FasL also induced the inhibition of SMS in activated human peripheral blood T lymphocytes (data not shown). In addition, in vitro SMS specific activity, as measured in the presence of an excess of PC, was significantly inhibited in protein extracts derived from FasL-treated Jurkat cells (Supplemental Fig. 2C). FasL-mediated SMS inhibition, as evaluated both in situ (Supplemental Fig. 2B) and in vitro (Supplemental Fig. 2C), was significantly prevented by zVAD-fmk, further indicating caspase involvement. Collectively, our data indicate that the inhibitory effect on SM de novo synthesis may be the consequence of diverse molecular events, including not only the inhibition of SMS specific enzyme activity itself but also possibly the inhibition of PC synthesis. FasL promotes caspase-dependent SMS1 relocation and disappearance. In HeLa cells stably over-expressing V5-tagged SMS1, the over-expressed enzyme was localized in the 1 perinuclear region in accordance with a Golgi localization (Supplemental Fig. 3). Upon FasL treatment, SMS1 was relocated throughout the cytoplasm in cells exhibiting nuclear condensation and was hardly detectable in cells having fragmented nuclei (Supplemental Fig. 3). zVAD-fmk prevented FasL-triggered SMS1 relocation and disappearance (Supplemental Fig. 3), pointing to caspase involvement in both phenomena. SMS2 is not as potent as SMS1 to negatively regulate FasL-induced apoptosis in HeLa cells. The role of SMS2 in FasL-induced apoptosis has also been investigated in HeLa cells. SMS2 was cleaved in FasL-treated cells, this phenomenon being prevented by zVAD-fmk (data not shown). In addition, V5-tagged SMS2 was cleaved in cell-free system by recombinant caspases-8 and -9 (data not shown). Endogenous SMS2 knock-down in HeLa cells significantly decreased specific SMS activity (Supplemental Fig. 4A) and slightly, but not significantly, increased FasL-induced apoptosis (Supplemental Fig. 4B). Whereas SMS2 overexpression triggered a significant SMS activity increase (Supplemental Fig. 4C), it slightly, yet not significantly, decreased FasL-triggered cell death (Supplemental Fig. 4D). Finally, SMS2 knock-down in SMS2 over-expressing cells led to a significant sensitization to FasL (Supplemental Fig. 4E). Collectively, our data indicate that (i) SMS2 is a putative caspase substrate in Fas signaling ; (ii) under some experimental conditions, SMS2 may also negatively regulate FasLinduced apoptosis in HeLa cells, but not as potently as SMS1. 2 Supplemental Materials and Methods Cells and reagents- HeLa cells stably over-expressing human SMS2 harboring V5 tag at the C-terminus 1 were kindly provided by Dr. J. Holthuis (Utrecht, The Netherlands). Wild- type and SMS2-V5-overexpressing HeLa cells were transiently transfected with either 50 nM siRNA (siRNA On target smart pool) against SMS2 (Perbio sciences, Brebières, France) or control (non-targeting smart pool) siRNA using Hiperfect reagent (Qiagen, Courtaboeuf, France) following the manufacturer’s instructions. De novo PC and SM syntheses, Determination of in situ and in vitro SMS activities, Determination of cell viability- Protocols have been described in the Materials and Methods section of the core Manuscript. Fluorescent microscopy analysis- Cells were grown on glass coverslips for 24 hours in DMEM supplemented with 10% FCS. Cells were incubated for one hour with or without 40 μM zVAD-fmk and further incubated for 8 hours in the presence or absence of 125 ng/mL FasL. The indirect immunofluorescence protocol was carried out as indicated for confocal microscopy analysis. Cells were examined under a Leica fluorescence-equipped microscope using a 40-oil objective lens. Supplemental references 1. Huitema, K, van den Dikkenberg, J, Brouwers, JF and Holthuis, JC, (2004) Identification of a family of animal sphingomyelin synthases. Embo J 23: 33-44. 3 Supplemental legend to figures. Supplemental Figure 1, PC and SM de novo syntheses in Fas signaling. 3x106 Jurkat cells (clone A3) were incubated in the presence of [3H]choline (1 Ci/mL) for 4 hours to allow PC labeling and further incubated for the indicated times in the presence or absence of 500 ng/mL FasL. [3H]choline-labeled PC and SM were quantified after alkaline methanolysis. Data are representative of three independent experiments. Supplemental Figure 2, FasL triggers caspase-dependent inhibition of SMS activity. Wild-type (A3) Jurkat cells were pre-incubated or not for one hour with 40 M zVAD-fmk and further incubated for 8 hours with or without the indicated FasL concentrations. Cell viability was evaluated by MTT assay (A). In situ SMS activity was measured by adding 2.5 M C6-NBD-ceramide to the cell culture medium two hours before stopping the reaction (B). Results are expressed as the percentage of the values measured in the absence of FasL. Data are means s.e.m. of three independent experiments. B right, representative TLC plate of NBD-sphingolipid analysis. C, Jurkat cells (A3) were pre-incubated or not for one hour with 40 M zVAD-fmk and further incubated for 8 hours with or without 500 ng/mL FasL as indicated. Specific SMS activity was measured on 200 g protein extracts (basal activity: 288 pmol/h/mg). Results are expressed as the percentage of untreated cells. Data are means s.e.m. of three independent experiments. Supplemental Figure 3, FasL triggers caspase-dependent SMS1 relocation and disappearance. SMS1-V5 over-expressing HeLa cells were pre-incubated for one hour with or without 40 M zVAD-fmk and further incubated in the presence or absence of 125 ng/mL FasL for 8 hours. HeLa cells were labeled with DAPI and anti-V5 antibody. Analysis was 4 performed by fluorescence microscopy and data are representative of three independent experiments. Supplemental Figure 4, Effect of SMS2 on FasL-induced apoptosis in HeLa cells. A and B, HeLa cells were transfected with control siRNA (si cnt) or siRNA to SMS2 (si SMS2) and further incubated for 72 hours. A, Specific SMS activity was measured on 200 g protein extract and results obtained in cells transfected with siRNA to SMS2 are expressed as the percentage of cells transfected with the control siRNA. B, Cells were further incubated for 8 hours in the presence or absence of FasL (125 ng/mL). The proportion of cells exhibiting apoptotic nuclei was evaluated by microscopic analysis. C, In situ SMS and GCS activities were measured in mock-transfected (pcDNA3.1) and SMS2-V5 over-expressing (SMS2-V5) HeLa cells. Data obtained in SMS2 over-expressing HeLa cells are expressed as the percentage of those measured in mock-transfected cells. D, mock-transfected (pcDNA3.1) and SMS2-V5 over-expressing (SMS2) HeLa cells were incubated for 8 hours in the presence or absence of FasL (125 ng/mL) and cell viability was assessed by MTT assay. Results are expressed as the percentage of untreated cells for each cell line. E, SMS2 over-expressing HeLa cells were transfected with control siRNA (si cnt) or siRNA to SMS2 (si SMS2) and further incubated for 72 hours. E left, Western blot on 50 g protein extracts using anti-V5 or anti--actin antibodies. Cells were further incubated for 8 hours in the presence or absence of FasL (125 ng/mL). E right, The proportion of cells exhibiting apoptotic nuclei was evaluated by microscopic analysis. A-E, data are means s.e.m. of 3-4 independent experiments. 5