* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Rotation Final Report
Immune system wikipedia , lookup
Lymphopoiesis wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Adaptive immune system wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Immunosuppressive drug wikipedia , lookup
Innate immune system wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Rotation Report:
An introduction to Immunology and Molecular Dynamics
Denise Chac
25 November 2014
Contents:
I.
II.
III.
IV.
I.
Expectations for Rotation
Immunology
Molecular Dynamics
Protocols for simulating HIV – gp120 in water.
EXPECTATIONS FOR ROTATION
At the start of this rotation, I had no background experience in immunology or
molecular dynamics. Entering Dr. Ha Youn Lee’s lab, I expected to learn the basics of
immunology with regards to HIV and gain an understanding of Molecular Dynamics. Due to
my lack of experience with a wet lab, I wanted to focus on the computational side of the lab.
While I only had basic knowledge of math and physics, I would like to further improve my
skills through learning molecular dynamics.
Goals:
II.
1) Gain a basic understanding of immunology
2) Acquire the skills to operate and run molecular dynamics
IMMUNOLOGY
Immunology is the study of how our bodies react to foreign pathogens such as
diseases and viruses. Our body responses using two different systems: the innate immune
system and the adaptive immune system. The innate immune system is known to be the
more system that is always available and can readily combat a wide range of pathogens.
While it is the more rapid system, it lacks specificity. The adaptive immune system, which
is the more specific and effective form of immunity, requires previous encounters with the
pathogens.
One way in which our immune system reacts to pathogens is through white blood
cells, specifically lymphocytes. Of the lymphocytes, there are the B cells, T cells, and natural
killer (NK) cells.
B Cells
B cells derive form the bone marrow and have membrane bound antibodies on their
cell surface to identify antigens. All B cells have antibodies on their cell surface but the
types of antibodies are highly variable. This variation of antibodies is essential for
identifying the different pathogens that invade the body. B cells are activated when they
encounter their matching antigen through the variable antibody. The antibodies on the B
cell bind to an area on the antigen called the epitope. Once the B cell binds to the antigen,
the B cell is activated and begins to engulf the pathogen; the pathogen gets digested and the
resulting peptides are presented on the cell surface using MHC (Major Histocompatibility
complex) class II proteins. These MHC proteins are important for the activation of other
immune responses. Before the B cell can begin replicating and differentiating, the MHC
attracts the help of mature helper T cells. Once it does, the naïve B cells differentiate into
memory cells and effector cells. Effector cells are plasma cells that produce free antibodies
that are able to recognize the original pathogen and tag them for further immune response.
Memory B cells are clones of the original B cell with the same variation of antibodies
capable of recognizing the original pathogen.
T Cells
T cells also derive from the bone marrow but they mature in the thymus. They differ
form B cells in that they have T-cell receptors that recognize different MHC classes. Of T
cells, there are two types: cytotoxic and helper T cells.
Cytotoxic T cells are capable of identifying class I MHC on any antigen-presenting
cell. They have a CD8 protein that further helps the binding of the T cell to the target cell.
Once the T cell binds to its target cell, it then begins differentiating into effector and
memory cells. Effector cells are capable of latching onto cancerous or “bad” cells trhough
the class I MHC protein presented on the cell surface. Once it is bound, the cell releases
proteins to lyse the cell membrane, resulting in cell death.
Helper T cells, on the other hand, are attracted to class II MHC found on B cells. They
have CD4 proteins that help the binding of the helper T cells to the target cell. Helper T cells
are attracted to professional antigen presenting cells. Once helper T cells attach to their
target cell, they release cytokines and differentiate into effector and memory cells.
Cytokines are a general group of proteins that help increase cell signaling and recruit other
proteins to fight the pathogens.
Viruses
Viruses are unique in that they utilize the cell’s own mechanism to reproduce and
proliferate. They act differently than bacteria and other pathogens because they are
capable of incorporating their own DNA into the host or target cell. The virus first fuses it’s
membrane with the target cell. Once the membrane is fussed, it releases its content into the
host cell and the viral RNA is incorporated into the host DNA through splicing mechanisms.
Once the viral DNA is combined with the host DNA, viral replication is possible when the
host cell replicates. As for the other remnants of the virus, viral peptides are transported
into the ER lumen. The viral peptide is then bound to MHC proteins and transported
through the Golgi apparatus. The MHC protein and that attached viral peptide is then
presented on the cell surface. Once the MHC 1 and antigen peptide are presented, it is
possible for cytotoxic T cells to recognize the infection.
HIV
The Human Immunodeficiency Virus (HIV) is a retrovirus that attacks key
components of the immune system. The HIV virion first binds to CD4 proteins on T cells
through the membrane envelope protein gp120. Once it is bound, another envelope
protein, gp41, helps the HIV envelope fuse with the target cell’s membrane. Once it is fused,
the HIV contents are spilled into the cytosol of the target cell. Through reverse
transcriptase, the HIV RNA is transcribed into DNA and then incorporated into the target
cell’s DNA with integrase. When the target cell (usually a T cell) becomes activated, the HIV
provirus (the portion of the virus DNA incorporated into the host genome) becomes
transcribed and replicated.
MORE BACKGROUND
III. MOLECULAR DYNAMICS
While in vitro and in vivo experiments are the conventional way to test hypotheses,
with technological advances, it is now possible to perform experiments in silico in biology.
Molecular dynamics is a powerful tool that allows biologists to observe the reactions of
molecules in a simulated environment. Through th
IV. CASE STUDY: GP120
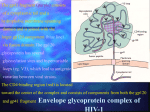
The envelope glycoprotein gp120 is an essential component in the HIV that
determines the docking of the HIV virion to the targeted T cell. On the cell surface, gp120
attaches to the CD4 protein on the T cell and enables the HIV virion to dock and then
continue on its life cycle of fusing its membrane and releasing the viral content into the
targeted cell.
Simulating gp120 using Molecular Dynamics
In this project, I (1) minimized the gp120 protein in water to obtain the lowest
energy conformation, (2) heated the structure from 0 K to 300 K, and then ran the (3)
simulation of gp120 with constant temperature 300 K and pressure 1 atm for 60 ps.
V.
PROTOCOLS
Necessary Programs:
o Terminal Box / Command Box
o X11 (for Mac)
o AMBER
tLEaP – basic model building
SANDER - Simulated annealing with NMR-derived energy restraints
Gedit – text editor
Gnuplot – program to graph data
1.
2.
Ptraj - program to analyze trajectories
VMD – Visual Molecular Dynamics
Loading gp120
To build the molecule, you need the LEaP program provided by the AMBER software
package. This program provides the tools to build the system/model and then
produce the Amber coordinates and parameter/topology input files.
After opening the LEaP program, the force field must be specified.
When building the molecule, it is possible to build the molecule from a sequence ($
foo = sequence { ACE ALAN ME } ) or it may be obtained from a pdf file ($ foo =
loadpdb file.pdb ).
Solvating the structure is also possible in LEaP. For this project, the gp120 protein
was solvated in a period box of water with a 10.0 Angstroms buffer (between the
protein and the periodic box wall).
After building the molecule, the files need to be saved in the appropriate files for
Amber. These files are prmtop and inpcrd.
o Prmtop = parameter and topology file
o Inpcrd = coordinate file
COMMANDS
o $ tleap
$ source leaprc.ff03
$ gp120 = loadpdb 1MEQ.pdb
$ solvatebox gp120 TIP3PBOX 10.0
$ saveamberparm gp120 prmtop incrd
$ quit
Scripting input files
o Once all start files are obtained (prmtop and inpcrd), the next step is to make the
input files that tell the MD program what to do to the system. To create these
script, the simple text editor program gedit was used.
Minimization
o This script is used to obtain the lowest energy structure.
o Contents:
Gedit Script – Minimization
imin =1 this dictates that it is a
minimization run
Minimize
ntx = 1 read the coordinates but
&cntrl
not the velocities from ASCII
imin=1,
formatted inpcrd coordinate file
ntx=1,
irest = 0 do not restart simulation
irest=0,
(not applicable to minimization)
maxcyc=2000,
maxcyc = 2000 maximum
ncyc=1000,
minimization cycles
ntpr=100,
ntwx=0,
cut=8.0,
/
ncyc = 1000 the steepest desecent algorithm for the first 0-ncyc
cycles, then switches to conjugate gradient algorithm for ncyc-maxcyc
cycles
ntpr = 100 print to the amber
mdout outfile every ntpr cycles
Gedit Script – Heat
ntwx = 0 no amber mdcrd
trajectory file written (not
Heat
applicable to minimization)
&cntrl
cut = 8.0 nonbounded cutoff
imin=0,
distance in Angstroms
ntx=1,
irest=0,
Heat
nstlim=10000,
o This script is used to heat the system from
dt=0.002,
0 K to 300 K while maintaining a constant
ntf=2,
number of particles and volume.
ntc=2,
o Contents
tempi=0.0,
imin = 0 dictates the type of
temp0=300.0,
molecular dynamics run (it is not a
ntpr=100,
minimization run)
ntwx=100,
nstilm = 10000 number of MD
cut=8.0,
steps in the run
ntb=1,
dt = 0.002 time step in
ntp=0,
picoseconds (ps)
ntt=3,
ntf = 2 setting to not calculate
gamma_ln=2.0,
force for SHAKE constrained bonds
nmropt=1,
ntc = 2 enable SHAKE to constrain
ig=-1,
all bonds involving hydrogen
/
tempi = 0.0 Initial thermostat
&wt type='TEMP0', istep1=0,
temperature in K
istep2=9000, value1=0.0,
temp0 = 300.0 Final thermostat
value2=300.0 /
temperature in K
&wt type='TEMP0',
ntwx = 1000 periodic boundaries
istep1=9001, istep2=10000,
for constant volume
value1=300.0, value2=300.0 /
ntb = 1 periodic boundaries for
&wt type='END' /
constant volume
ntp = 0 no pressure control
ntt = 3 temperature control with Langevin thermostat
gamma_ln = 2.0 Langevin thermostat collision frequency
nmropt = 1 NMR restrains and weight changes read
ig = -1 randomize the seed for the pseudo-random number
generator
Final steps For steps 0-9000, increase temperature from 0 K to
300. For steps 9001-10,000, keep constant temperature at 300 K.
3.
Production
o This script is used to run the simulation of gp120 in the periodic solvate box
at constant temperature 300 K and constant pressure 1 atm.
o Contents:
ntx = 5 read coordinates and
Gedit Script – Production
velocities from unformatted inpcrd
coordinate file
Production
rest = 1 restart previous MD run
&cntrl
velocities from inpcrd file will be used
imin=0,
fur initial atom velocities
ntx=5,
temp0 = 300.0 thermostat
irest=1,
temperature set at 300 K
nstlim=30000,
ntb = 2 use periodic boundary
dt=0.002,
conditions with constant pressure
ntf=2,
ntp = 1 use the Berendsen barostat
ntc=2,
for constant pressure simulation
temp0=300.0,
ntpr=100,
To calculate the run length, take the number of MD
ntwx=100,
steps (nstilm) and multiple it by the time step of MD
cut=8.0,
(dt).
ntb=2,
o Heat:
ntp=1,
Nstlim = 10,000 run simulation for
ntt=3,
10,000 MD time steps
gamma_ln=2.0,
Dt = 0.002 MD time step is 0.002 ps
ig=-1,
Length of simulation 10,000 * 0.002
/
= 20 ps
o Production:
Nstilm = 30,000 run simulation for 30,000 MD time steps
Dt = 0.002 MD time step is 0.002 ps
Length of simulation 30,000 * 0.002 = 60 ps
Sdf
Commands
o $ gedit Min.in
o $ gedit Heat.in
o $ gedit Prod.in
o Once the program opens up in a separate window, create these scripts by
copy, paste, and save.
MD Simulation