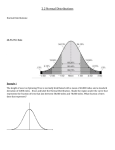
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download biochem_lec3_27-4
Peptide synthesis wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Butyric acid wikipedia , lookup
Carbon sink wikipedia , lookup
Citric acid cycle wikipedia , lookup
Biosequestration wikipedia , lookup
Human digestive system wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Biosynthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Biochemistry wikipedia , lookup
Biochemistry lec# 3 Dr. faisal al-khateeb 27/4/2011 Wednesday Chapter 18 *Slide (1) Chole/ster/OL, from its name ster it’s a steroid contain steroid nucleus, OL alcohol, chole gall bladder (bile) because it was first isolated from the gall bladder almost 200 years ago so it was named according to the gall bladder, its an alcohol steroid from the gall bladder. *Slide (2) This is a steroid nucleus consists of 4 rings, 3 six-member rings and the fourth is a five-member ring. Total number of carbon is 17C. (We have to know the numbering of carbons in the steroid nucleus and how to draw it). *Slide (3) This is the complete structure of cholesterol. That’s consisting of steroid nucleus and a hydrocarbon tail. Features of the cholesterol structure:1) It contains the hydroxyl group at carbon number 3, that’s why it's named cholesterol. (we have to know its location) 2) Double bond between carbon number 5 and carbon number 6. 3) 2 methyl groups attached to carbon 10 (carbon 19 of the first methyl group) and carbon 13 (carbon 18 of the second methyl group). 4) Side chain, that is consist of 8 carbons attached to carbon 17 * This makes the total number of carbon atoms in cholesterol equal to 27. 1 *Slide (4) These are the gall bladder stones, they are made of cholesterol so cholesterol may precipitate in the gall bladder forming cholesterol stones that’s because its is poorly or insoluble in water and its secreted in the bile from where it was isolated first. It should be in the soluble form, if the process of making it soluble is defected, stones will be formed. * Note cholesterol ester; fatty acid is esterifies to cholesterol by eliminating the hydroxyl group at carbon 3 making the cholesterol ester non polar compound. *Slide (5) Sources of cholesterol:1) Cholesterol is synthesized in the liver, small intestine, adrenal cortex and number of tissues at a rate of 1000 mg/day. ** Cholesterol is needed in all animal cells and all animal cells are capable of producing cholesterol, which shows its importance for the cells. **As cholesterol is mainly produced by the liver and small intestine it's then transported to different cells. Although cells have the ability to make it they get it ready made. 2) The dietary cholesterol present about 300 mg/day in the case of low cholesterol diet, it may be more than that, even if we want to reduce the amount of cholesterol with normal diet, we cant reduce it below 300 mg/day because we are consuming meat, egg, milk or any product that contain cholesterol. **Cholesterol is found in animal product because all animal cells contain cholesterol. On the other hand plant cells don’t produce cholesterol, any food derived from plants or vegetable oil doesn’t contain cholesterol. **Although cholesterol is produced by all cells, it's not degraded by these cells. **Cholesterol is eliminated from the body either as it's in the bile (cholesterol) or it is converted to bile salts and bile acids. Little amount of cholesterol is converted to steroid hormones. 2 *Slide (6) This is an Ergosterol (which differs a little bit from cholesterol) -It's found in plant cells, so plant cells produce sterols other than cholesterol. -Plant steroids are poorly absorbed by humans, if they are absorbed they are excreted again into the small intestine along with cholesterol. So plan sterols are helpful to reduce the amount of cholesterol in the body because their elimination or excretion is accompanied with cholesterol. *Slide (7) Homeostasis of cholesterol; These are the three major roots representing the source of cholesterol in the liver. It's obtained from 1) diet through chylomicron remnants then cholesterol is transferred to the liver, 2) other part is synthesized by the liver, 3) cholesterol synthesized and transported from the tissues to the liver through HDL. **cholesterol is distributed from the liver to 1) tissues in the form of VLDL, 2) free cholesterol is secreted in the bile,3) half of the cholesterol is converted to bile salts or bile acids to be excreted in the small intestine. **Eggs are the major source of dietary cholesterol on daily bases, one egg contains 200 mg of cholesterol which is almost the daily requirement so if we want to reduce cholesterol intake one egg is sufficient ( low cholesterol diet). *Slide (8) Cholesterol synthesis requires:1) Carbon source: acetyl CoA (produced from Boxidation, pyruvate dehydrogenase), all the 27 carbon atoms of cholesterol are produced from acetyl CoA. The binding occur by labeling the acetyl groups in acetyl CoA (like using C 13 ) , it was found that all cholesterol atoms (27) comes from the two carbon atoms of acetyl CoA. 2) Energy: in the form of ATP (in order to join these molecules together). 3) Reducing power: NADPH (as in the case of fatty acid synthesis). 3 **Acetyl group is much more oxidized than the final product cholesterol which is reduced so that’s why we need a reducing agent. 4) One atom of oxygen (O2) for producing the oxygen of cholesterol. *Slide (9) Stages of cholesterol synthesis. - Cholesterol synthesis pathway is a long path consists of more than 20 steps. But we will study specific stages of the pathway. - Over view of cholesterol synthesis starts with Acetyl coA (two carbons compound) then it is converted to Mevalonate (6 carbon compound) which is decarboxylated to Isoprene units (hydrocarbon units of 5 carbon atoms " isopentenyl") six of these isoprene units condense together in several steps to produce hydrocarbon compound called Squalene (30 carbon compound) then its converted to sterole, Lanosterole( 30 carbon compound) after many steps its converted to Cholesterol (27 carbon compound). *Slide (10) First step of cholesterol synthesis: acetyl CoA condensation. Two Acetyl CoA gives Acetoacetyl CoA then the third acetyl CoA condenses to produce HMG CoA (hydroxymethylglutaryl CoA) an intermediate in ketone bodies synthesis. The enzyme required to catalyze this step is HMG CoA synthase. These two steps are similar to steps of ketone bodies synthesis but ketone bodies synthesis occurs in the mitochondria while cholesterol synthesis occurs in the cytosol or cytoplasm. *Slide (11) The third step is reduction of carboxyl group linked to coenzyme A. First reduction step produces an aldehyde then the second reduction step produces an alcohol so two reduction steps will convert HMG CoA into Mevalonate or 4 mevalonic acid ( it’s a 6 carbon compound with a hydroxyl group attached to carbon 3 and a carboxyl group). *Slide (12) Now how Mevalonic acid is condensed to produce hydrocarbon compound squalene? -Hydroxyl group at carbon number 1 in mevalonic acid is phosphorylated, two steps of phosphorylation using two ATP molecules produces 5-Pyrophosphomevalonic acid (2 phosphate groups are added to activate and produce isoprene units). The next step is decarboxilation of the hydroxyl group at carbon 3 and elimination of H2O by the usage of ATP gives up Isopentenyl pyrophosphate, then isopentenyl pyrophosphate is isomerized into 3,3-Dimethylallylpyrophosphate. Both isopentenyl pyrophosphate and its isomer condensate to form (10 carbon) Geranyl pyrophosphate, one more condensation gives Farnesyl pyrophosphate (15 carbon compound). ** The idea is activation then several condensations, to convert isopentenyl group into 10 carbon compound then 15 carbon compound. *Slide (13) Two farnesyl pyrophosphate condense head to head so two pyrophosphate groups will be removed producing squalene( hydrocarbon branched compound consist of hydrogen and carbon, contain 30 carbon) ** Pathway of squalene synthesis is similar to many compounds that are formed from number of isoprene units like coenzyme Q, some vitamins, and rubber in plants. *Slide (14) A hydrocarbon chain formed from 30 carbons: squalene. It's drawn in this way to show the similarity to cholesterol structure. *Slide (15) squalene is converted to squalene 2, 3 epoxide. Oxygen now is needed and it is added to carbon 2, 3 in the form of epoxide which is considered as a very unstable compound that rapidly change to form lanosterol (first sterole, has about 30 carbon), next steps contain several modification to convert lanosterol into cholesterol. 5 *Slide (16) The last step in the synthesis pathway 7-dehydrocholesterol is the substrate for production of vitamin D in the skin by the exposure to sun light (vitamin D3). *Slide (17) Overall steps for cholesterol synthesis with the intermediate. (We should be familiar with it!) *Slide (18) Synthesis of bile acid. The conversion of cholesterol to cholic acid (intermediate in the TAG digestion) involve hydroxylation at carbon 7 and 12, this first step in bile acids synthesis is catalyzed by an enzyme that adds hydroxyl group to carbon 7 called 7-a-hydroxylose. It is a rate-limiting step and its regulated, activated by cholesterol and inhibition by cholic acid. So it’s a feed-back inhibition, bile acids inhibit their own synthesis. If we decrease the amount of cholic acid in the liver, this will help in converting more cholesterol into bile acids and getting rid of cholesterol (stimulating the conversion). *Slide (19) Synthesis of bile acids and their circulation between the liver and the small intestine. First step: cholesterol is converted to primary bile acids (cholic acids) then these cholic acids are conjugated with glycine to produce conjugated bile acids, these are secreted by the liver to the bile then to small intestine, in the small intestine they are conjugated Again and the primary bile acids are converted to secondary bile acids by removing some hydroxyl groups. -Primary: - they are produced in the liver -Secondary:-they are modified in the small intestine and reabsorbed after doing their job (which is making the fat soluble), then they are added again to the liver to be execrated again .So everyday 30 g of bile salt are secreted, 90% are reabsorb and execrated. This enterohepatic cycle occur many times per day. Some cholesterol or bile acids escape from reabsorbing. So there is net loss about of 0.5 g per day of bile acids and this represent the amount that should be synthesized by the liver per day. ** Everyday 0.5g cholesterol is lost in fesses and 0.5 g produced by the liver. 6 **Any substance that binds to bile acids and prevent reabsorption will help in decreasing the amount of bile acids absorbed in the liver so it will increase the amount of cholesterol converted to bile acids. If we cut this circle we are forcing more cholesterol to be converted into bile acid and this is away to reduce the amount of cholesterol in the body. *Slide (20) Lowering cholesterol level. - That's in order to prevent and protect our bodies from atheroscterosis. - it can be achieved; 1. Dietary:* first step- is to reduce the cholesterol intake to less than 300 mg per day. But unfortunately even if the cholesterol intake is reduced the synthesis will increase. If we reduce the cholesterol intake into zero, the cholesterol percentage will decrease only 10% not more than that. *Second step-increase polyunsaturated fatty acid to saturated fatty acid (like w-3/ w-6/w-9) will decease the cholesterol level. *Third step- increasing the fiber in the food (dietary fiber) achieve 5-10% in lowering cholesterol level but we can't make all our food fibers. *Forth step-ingestion of plant steroid esters. 2. Inhibition of synthesis may be the only possible way to reduce the cholesterol into the target level. 3. Decrease enterohypatic circulation of bile acid by some drugs that bind to bile acid and prevent their reabsorbtion. *Slide (21) This is the structure of HMG. Conversion of HMG to mevalonate is a rate limiting step in cholesterol synthesis. The enzyme HMG coA reductase can be inhibited by drugs so the rate of synthesis will be decreased. Simvastatis is a very common drug; it has a structure (ring) that is similar to HMG. It binds to enzyme HMG CoA reductase and inhibits it. *Slide (22) A very common drug (lipitor) - Is used to reduce cholesterol synthesis. - HMG CoA reductase inhibitor. 7 *Slides (23) Esterefication of cholesterol in the cells into cholesterol ester occur by the addition of fatty acid to hydroxyl group at carbon 3. Source of fatty acid is fatty acyl CoA - type of enzymes acyl transferase ( transfer fatty acid from acyl CoA to the cholesterol ), its whole name acyl CoA : cholesterol acyltranferase (ACAT). *Slide (24) Esterefication of cholesterol in the plasma -There is NO acyl CoA in the plasma. Cholesterol in the plasma is found in the lipoproteins and HDL. In the lipoproteins there is phospholipids lecithin which is the donor of fatty acid, after donating fatty acid it becomes lycolycitin, the enzyme required is lecithin: cholesterol acyl transferase (LCAT). 8