* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chapter 11 Carboxylic Anhydrides, Esters, and Amides
Ring-closing metathesis wikipedia , lookup
Bottromycin wikipedia , lookup
Hydroformylation wikipedia , lookup
Sulfuric acid wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Petasis reaction wikipedia , lookup
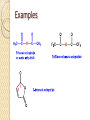
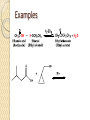
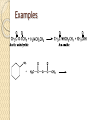
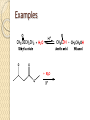
Chapter 11 Carboxylic Anhydrides, Esters, and Amides Carboxyl Derivatives In this chapter, we study three classes of compounds derived from carboxylic acids; anhydrides, esters, and amides. • Each is related to a carboxyl group by loss of H2O. Anhydrides The functional group of an anhydride is two carbonyl groups bonded to the same oxygen. ◦ The anhydride may be symmetrical (from two identical acyl groups), or mixed (from two different acyl groups). ◦ To name an anhydride, drop the word "acid" from the name of the carboxylic acid from which the anhydride is derived and add the word "anhydride”. Examples Esters The functional group of an ester is a carbonyl group bonded to an -OR group. R may be alkyl or aryl. • Both IUPAC and common names of esters are derived from the names of the parent carboxylic acids. • Name the alkyl or aryl group bonded to oxygen first, followed by the name of the acid; replace the suffix -ic acid by -ate. • A cyclic ester is called a lactone. Examples Amides The functional group of an amide is a carbonyl group bonded to a nitrogen atom. • To name an amide, drop the suffix -oic acid from the IUPAC name of the parent acid, or -ic acid from its common name, and add -amide. Examples If the amide nitrogen is also bonded to an alkyl or aryl group, name the group and show its location on nitrogen by N- ; two alkyl or aryl groups by N,N-di-. Amides A cyclic amide is called a lactam. Amides The penicillins are referred to as b-lactam antibiotics Amides • The cephalosporins are also b-lactam antibiotics. Fischer Esterification Fischer esterification is the most common method for the preparation of esters • In Fischer esterification, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, such as concentrated sulfuric acid. Examples Preparation of Amides • In principle, we can form an amide by treating a carboxylic acid with an amine and removing -OH from the acid and an -H from the amine. • In practice what occurs if the two are mixed is an acid-base reaction to form an ammonium salt. • If this salt is heated to a high enough temperature, water is eliminated and an amide forms. Examples Predict the amide product How to synthesize the following compound Preparation of Amides • It is much more common, however, to prepare amides by treating an amine with an anhydride. Examples Hydrolysis of Anhydrides Hydrolysis is a chemical decomposition involving breaking a bond and the addition of the elements of water. • Carboxylic anhydrides, particularly the low-molecular- weight ones, react readily with water (hydrolyze) to give two carboxylic acids. Example Hydrolysis of Esters • Esters hydrolyze only very slowly, even in boiling water. • Hydrolysis becomes considerably more rapid, however, when the ester is heated in aqueous acid or base. • Hydrolysis of esters in aqueous acid is the reverse of Fischer esterification. • A large excess of water drives the equilibrium to the right to form the carboxylic acid and alcohol (Le Châtelier's principle). Examples Hydrolysis of Esters • We can also hydrolyze an ester using a hot aqueous base, such as aqueous NaOH. • This reaction is often called saponification, a reference to its use in the manufacture of soaps. • The carboxylic acid formed in the hydrolysis reacts with hydroxide ion to form a carboxylic acid anion. • Each mole of ester hydrolyzed requires one mole of base. Examples Hydrolysis of Amides Amides require more vigorous conditions for hydrolysis in both acid and base than do esters. • Hydrolysis in hot aqueous acid gives a carboxylic acid and an ammonium ion. • Hydrolysis is driven to completion by the acid-base reaction between ammonia or the amine and the acid to form an ammonium ion. • Each mole of amide hydrolyzed requires one mole of acid. Hydrolysis of Amides • Hydrolysis of an amide in aqueous base gives a carboxylic acid salt and ammonia or an amine. • Hydrolysis is driven to completion by the acid-base reaction between the carboxylic acid and base to form a salt. • Each mole of amide hydrolyzed requires one mole of base. Examples Reaction with Alcohols Anhydrides react with alcohols and phenols to give an ester and a carboxylic acid. Reaction with Alcohols Aspirin is prepared by the reaction of salicylic acid with acetic anhydride. Reaction with Amines Anhydrides react with ammonia and with 1° and 2° amines to form amides. ◦ Two moles of amine are required; one to form the amide and one to neutralize the carboxylic acid by-product. Reaction with Amines Esters react with ammonia and with 1° and 2° amines to form amides. Thus, an amide can be prepared from a carboxylic acid by first converting the carboxylic acid to an ester by Fischer esterification and then reaction of the ester with an amine. Amides do not react with ammonia or with amines Phosphoric Anhydrides The functional group of a phosphoric anhydride is two phosphoryl (P=O) groups bonded to the same oxygen atom. Phosphoric Esters • Phosphoric acid forms mono-, di-, and triphosphoric esters. • In more complex phosphoric esters, it is common to name the organic molecule and then indicate the presence of the phosphoric ester by either the word "phosphate" or the prefix phospho-. • Dihydroxyacetone phosphate and pyridoxal phosphate are shown as they are ionized at pH 7.4, the pH of blood plasma. Step-Growth Polymerization Step-growth polymers are formed by reaction between two molecules, each of which contains two functional groups. Each new bond is created in a separate step. • in this section, we discuss three types of step-growth polymers; polyamides, polyesters, and polycarbonates. Polyamides Nylon-66 was the first purely synthetic fiber. • It is synthesized from two six-carbon monomers. Polyamides The polyaromatic amide known as Kevlar is made from an aromatic dicarboxylic acid and an aromatic diamine. Polyesters The first polyester involved polymerization of this diester and diol. Polycarbonates Lexan, the most familiar polycarbonate, is formed by reaction between the disodium salt of bisphenol A and phosgene.