* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lecture 17-PDF
Renormalization wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Renormalization group wikipedia , lookup
Magnetoreception wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Tight binding wikipedia , lookup
Nitrogen-vacancy center wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Spin (physics) wikipedia , lookup
Mössbauer spectroscopy wikipedia , lookup
Atomic orbital wikipedia , lookup
Electron paramagnetic resonance wikipedia , lookup
Electron scattering wikipedia , lookup
Atomic theory wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Electron configuration wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
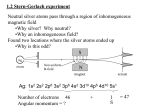
Lecture 17 Title :Fine structure : Spin-orbit coupling Page-0 In this lecture, we will concentrate on the fine structure of the one electron atoms. The cause of this fine structure is the interaction between the orbital angular momentum and the spin angular momentum. We will review the origin for this interaction and the effect of the coupling between orbital and spin on the spectral lines. We will discuss here mainly hydrogen atom and also the other alkali atoms. Page-1 It is observed in the sodium spectrum (shown in the figure below) that D-line (yellow emission) is split into two lines D1 = 5895.93 Å or 589.59 nm, D2 = 5889.9 Å or 588.99 nm The reason of observing this doublet is that the energy levels split into two for the terms except s-level (l = 0). Many lines of the other alkali atoms are also doublets. With high resolution spectrometer shows that even hydrogen atom lines are also having this doublet nature. The Coulomb interactions between the nucleus and electron in the outermost orbit for alkali atoms can not explain this observation Not only alkali atoms, but also in the other multielectron atoms the transitions between the terms split into more number of transitions. This is known as Fine structure. Emission from Sodium lamp 4000 3500 5895.93 Å 5889.96 Å 3000 2500 2000 Series1 1500 1000 500 0 580 585 590 Wavelength (nm) → 595 Page-2 In the previous lectures, we have discussed that the Hamiltonian for describing one electron atoms (Alkali atoms) is 2 2 − ∇ ψ ( r , θ , φ ) + V ( r )ψ ( r , θ , φ ) = Eψ ( r , θ , φ ) 2µ where V ( r ) is the Coulomb interaction between the nucleus and electron. We have also discussed the Hamiltonian needed to explain multielectron atoms = H H * + H1 2 N 2 N ∑ ∇i + ∑U ( ri ) 2me i 1 =i 1 = where H * =− where N Z e2 = − + U r ( ) ∑i = ∑ i ri i 1 e2 and= H1 ∑ − i < j rij N e2 ∑ i < j rij e2 ∑ i < j rij N N Non-Spherical part only However, these descriptions are not enough to explain the observed splitting. We need to include the spin-orbit coupling into the Hamiltonian. In the following we will understand the origin of this spin orbit interaction. Page-3 Orbital magnetic dipole moment: Let us consider that an electron is moving with velocity V in a circular Bohr orbit of radius r that produces a current. This current loop will produce a magnetic field with the magnetic moment, eω 2 1 µl = iA = πr = − − eω r 2 2π 2 eω Where i = − , ω is the angular velocity, A is the area. 2π µl v r -e l Magnitude of orbital angular momentum = l m= mω r 2 eVr Where, me is the mass of the electron. Substituting we get, the orbital magnetic moment µl = − e l 2m Now, the Bohr magneton is defined as the magnetic moment of the first Bohr orbital. So, the Bohr magneton= µB e = 9.27x10-24 J/T 2m The Orbital magnetic moment in terms of Bohr magneton is thus, in the vector form µl = − gl µ B µ l and putting gl = 1, µl = − B l Substituting the value of the angular momentum µ= l µB l (l + 1)= µ B l (l + 1) Along the Z-direction, the component of lZ = ml , so µ µ µl = − B lz = − B ml = − µ B ml z ml z l = l l (l + 1) Page-4 When this dipole moment is placed in an external magnetic field along the Z-direction, it experiences a torque which can be expressed as τ= µ × B The potential energy ∆E =− µˆ B ⋅ Bˆ For a static magnetic moment, this torque tends to line up the magnetic moment with the magnetic field B, so that it reaches its lowest energy configuration. Here, the magnetic moment arises from the motion of an electron in orbit around a nucleus and the magnetic moment is proportional to the angular momentum of the electron. The torque exerted then produces a change in angular momentum. This change is perpendicular to that angular momentum, causing the magnetic moment to precess around the direction of the magnetic field rather than settle down in the direction of the magnetic field. This precession is known as Larmor precession. ∆φ l Sinθ Z BZ l θ µl = − Figure-17.1 e l 2me Page-5 When a torque is exerted perpendicular to the angular momentum l, it produces a change in angular momentum ∆l which is perpendicular to l. Referring to figure-17.1, the torque is given by, τ = ∆l l sin θ∆φ = = lω sin θ ………………………….(17.1) ∆t ∆t And also τ = µ × B = µl B sin θ = µB lB sin θ ……………………(17.2) µB So equating equations 17.1 and 17.2 we get lB sin θ = lω sin θ µ = > ω =B B This is known as Larmor frequency. Spin magnetic moment : Similar to orbital dipole moment, electron also produces the magnetic moment due to the spin angular momentum. Z The spin dipole moment, in terms of spin Lande g-factor g s . gs µB s µ sz = − g s µ B ms µs = − Where, µ SZ the component in the Z-direction µS s Page-6 As we discussed above, that the fine-structure in atomic spectra cannot be explained by Coulomb interaction between nucleus and electron. We have to consider magnetic interaction between orbital magnetic moment and the intrinsic spin magnetic moment. This is known as Spin-Orbit interaction. Let us understand how this interaction takes place. If we consider the reference frame of electron then nucleus moves about electron. This current j = − ZeV produces magnetic field at the electron. According to Ampere’s Law, the magnetic field at electron due to nucleus is µ0 j × r − Zeµ0 V × r B = = r3 4π r 3 4π Since l = r × mV = −mV × r − Zeµ0 mV × r Zeµ0 l B = = r3 4π m 4π m r 3 If we take the average field, then Zeµ0 1 B= l ……………..(17.3) 4π m r 3 + Ze r v −e Page-7 Now, the orientation potential energy of magnetic dipole moment is ∆ESpin −Orbit = −µs ⋅ B gµ We know that µs = − s B s gs µB ∆ESpin −Orbit = s ⋅B Transforming back to reference frame with nucleus, must include the factor of 2 due to Thomas precession (Reference : Eisberg & Resnick): 1 gs µB s ⋅B 2 This is the spin-orbit interaction energy. Substituting the value of B from equation 17.3, we get ∆= Eso ∆Eso = g s µ B Zeµ0 1 ⋅ = s l A l SO ⋅ s …………………………..(17.4) 2 4π m r 3 Page-8 Let us first discuss about the alkali atoms. The Hamiltonian needed for calculating the energy levels is 2 2 ∇ + V ( r ) + H Spin −Orbit H =− 2µ = H 0 + H Spin −Orbit We already know the energies calculated for H0 . We can treat H Spin −Orbit as perturbation on the energies of H0. But first let us see the effect of this interaction. This interaction couples the spin and orbital angular momentum to form the total angular momentum J. The figure 17.2 represents the vector diagram and accordingly we write j= l + s (for one electron atoms). Because of this coupling, the one electron wavefunctions l ml s ms which are the eigenfunctions of H0 are no more the eigenfunctions of the total Hamiltonian H. The eigenfunctions of the H will be J mJ which are the coupled wavefunctions and can be derived from the uncoupled wavefunctions l ml s ms , as described in previous lectures. So the perturbation energy ∆ESO = J mJ ASO l .s J mJ Now, j= l + s j 2 = l 2 + s 2 + 2l .s 1 2 2 2 j − l − s l= .s 2 j= l + s l s Figure-17.2 And thus, the energy correction due to the spin-orbit interaction is ASO = ∆ESO J mJ ASO l .s J= mJ [ j ( j + 1) − l (l + 1) − s( s + 1)] ………………….(17.5) 2 Page-9 Let us first look at the Sodium energy levels. The terms arising from the ground state configuration is 2 S . So= l 0,= s 1 . According to coupling of angular momenta j = 1 2 2 2 s +1 The notation we will use is LJ According to this the ground state energy level is 2 S 1 . Using the relation given in 2 equation 17.5, we get A1 1 1 2 ( + 1) − 0(0 + 1) − 1 ( 1 + 1) ∆ESO= S1 2 2 2 2 2 2 =0 ( ) Now for the first excited state, terms is 2 P So= l 1,= s 1 . According to coupling of angular momenta j = 1 , 3 2 2 2 2 2 Now this terms will split into two energy levels P1 and P3 2 ( ) 2 ∆ESO = P1 2 A2 1 1 ( + 1) − 1(1 + 1) − 1 ( 1 + 1) 2 2 2 2 2 = − A2 And ( ) 2 ∆ESO = P3 2 A2 3 3 ( + 1) − 1(1 + 1) − 1 ( 1 + 1) 2 2 2 2 2 A = 2 2 2 Page-10 So the construction of the energy levels is 2 2 A2 P A2 2 S 2 2 2 E0 P3 P1 2 5895.93 Å (2) 2 S 5889.96 Å (1) 1 2 Now let us calculate the spin orbit constant for 2P level. For (1) the transition energy : ν 1 (cm −1 ) = E0 + A2 108 16978.04 cm −1 = = 2 5889.96 For (2) the transition energy : ν 2 (cm −1 ) = E0 − A2 = 108 = 16960.85 cm −1 5895.93 Solving these two we get A2 = 11.46 cm −1 and E0 = 16972.31 cm −1 Page-11 Similar to sodium atom, Hydrogen atom also shows doublet. Spectral lines of H found to be composed of closely spaced doublets. Splitting is due to interactions between electron spin s and the orbital angular momentum l Hα line is single line according to the Bohr or Schrödinger theory. occurs at 656.47 nm for Hydrogen and 656.29 nm for Deuterium (isotope shift, λ∆~0.2 nm). Spin-orbit coupling produces fine-structure splitting of ~0.016 nm corresponds to an internal magnetic field on the electron of about 0.4 Tesla. Orbital and spin angular momenta couple together via the spin-orbit interaction Internal magnetic field produces torque which results in precession of l and s about their sum, the total angular momentum: This kind of coupling is called L-S coupling or Russell-Saunders coupling 2 2 P3 P 2 10.2 eV 121.6 nm 2 2 P1 2 splitting 4.5×10-5 eV S 2 S1 2 Page-12 Relativistic kinetic energy correction : According to special relativity, the kinetic energy of an electron of mass m and velocity v is: p2 p4 where p is the momentum T≈ − 2m 8m3c 2 The first term is the standard non-relativistic expression for kinetic energy. The second term is the lowest-order relativistic correction to this energy. Using perturbation theory, it can be show that 3 Z 2α 4 1 ∆Erel = − 3 mc 2 − n 2l + 1 8n This energy correction does not split the energy levels, it only produces an energy shift comparable to spin-orbit effect. So the total energy correction for the fine structure ∆EFS = ∆Eso + ∆Erel As En = -Z2E0/n2, where E0 = 1/2α2mc2, we can write EH − atom Z 2 E0 Z 2α 2 1 3 = − 2 1 + − n n j + 1/ 2 4n Page-13 So we found out that Energy correction only depends on j, which is of the order of α2 ~ 10-4 times smaller that the principle energy splitting. All levels are shifted down from the Bohr energies. For every n>1 and l, there are two states corresponding to j = l ± 1/2. States with same n and j but different l, have the same energies i.e., they are degenerate. L=l = 0 L=l = 1 L=l = 2 n=3 3 2S 1 ∆EFS 2 n=2 2 2S 1 2 1 S1 2 P1 2 D3 2 2 2 ∆EFS 2 2 2 ∆EFS n =1 2 2 P3 P3 P1 2 2 2 D5 2 Page-14 Here, we learnt that due to the orbital motion of the electron, the charge of the nucleus creates a magnetic field at the electron and this interacts with the spin of the electron. This interaction essentially couples the orbital and the spin angular momenta and the effect is the splitting of energy levels. This transition pattern is known as fine structure. The coupled angular momentum J is the good quantum number when spin-orbit coupling is introduced for energy level calculation. Classical explanation of spin-orbit interaction is not enough for the level having l = 0. The quantum explanation reveals that for describing the l = 0 level, the Fermi contact term is needed which does not have any classical analogue. Relativistic correction is needed to predict the accurate energies.
















![NAME: Quiz #5: Phys142 1. [4pts] Find the resulting current through](http://s1.studyres.com/store/data/006404813_1-90fcf53f79a7b619eafe061618bfacc1-150x150.png)