* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download MIKROGEN recomBlot EBV IgG recomBlot EBV IgM/IgA Epstein
Anaerobic infection wikipedia , lookup
2015–16 Zika virus epidemic wikipedia , lookup
Sarcocystis wikipedia , lookup
Leptospirosis wikipedia , lookup
Brucellosis wikipedia , lookup
African trypanosomiasis wikipedia , lookup
Marburg virus disease wikipedia , lookup
Trichinosis wikipedia , lookup
Herpes simplex wikipedia , lookup
Oesophagostomum wikipedia , lookup
Schistosomiasis wikipedia , lookup
Herpes simplex virus wikipedia , lookup
Middle East respiratory syndrome wikipedia , lookup
Toxoplasmosis wikipedia , lookup
West Nile fever wikipedia , lookup
Dirofilaria immitis wikipedia , lookup
Hepatitis C wikipedia , lookup
Neonatal infection wikipedia , lookup
Henipavirus wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Diagnosis of HIV/AIDS wikipedia , lookup
Human cytomegalovirus wikipedia , lookup
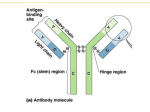
E MIKROGEN Western blot Epstein-Barr Virus molekularbiologische Entwicklungs-GmbH recomBlot EBV IgG recomBlot EBV IgM/IgA Immunoblot test with antigens produced by recombinant techniques for the detection of IgG, IgM and IgA antibodies against the Epstein-Barr virus (EBV). The Epstein-Barr virus, an ubiquitously occurring herpes virus, can cause the symptoms of infectious mononucleosis (Pfeiffer´s disease) on primary infection. Moreover, as a result of the lifelong persistence of this pathogen, reactivations can occur, especially in immuno-incompetent persons. Due to the diversity of symptoms caused by primary infection or reactivation and their correspondence with the symptoms of other diseases, one of the main tasks in routine diagnosis is the serological detection of a primary infection, past infection or possible reactivation. For this purpose, a series of individual determinations (EIA and IFT) are generally carried out for the particular class of antigen and type of antibody. The Western blot technique allows, at a glance, the detection and identification of IgG, IgM, and IgA antibodies against various classes of antigens. The application of highly specific and characteristic EBV proteins is made possible by the use of antigens produced by genetic engineering. Here the antigens EBNA-1 and p18 are of major importance: Due to the fact, that antiEBNA-1 and/or anti-p18-IgG antibodies are detected only in the case of postacute or past EBV infections (s. Evaluation), more than 95 % of the past EBV infections can be correctly identified with the recomBlot EBV IgG strip only. „The combination of p18 and EBNA-1 (in IgG detection) represents a so far unrivalled degree of certainty in the exclusion of primary infections ...“ React. Control gp 250/350 (MA) p54 (EA) p72 (EBNA-1) p138 (EA) p23 (VCA) p18 (VCA) Prof. Dr. G. Bauer, Freiburg ‘99 n Product Advantages • • • • • • • Recombinant antigens, therefore: ➣ High sensitivity and specificity ➣ Easy and clear interpretation due to easy to read bands ➣ No interference by anticellular antibodies Easy test procedure; automation possible Safe evaluation due to control sera and control strips Separate detection of IgG, IgM and IgA antibodies Screening of different anti-EBV antibodies in a single approach CE label: The recomBlot EBV tests meet the high standard of the EC directive 98/79/EC on in vitro diagnostic medical devices More than 95 % of the past EBV infections are correctly identified with the recomBlot EBV IgG strip only n Recombinant EBV Antigens used in the Test EBV antigen group Abbreviation Recombinant antigen Size of rec. antigen Membrane antigen MA gp250/350 70 kDal "Early antigens" EA p54 p138 54 kDal 40 kDal Virus capsid / structural antigen VCA p23 p18 23 kDal 18 kDal Nuclear antigen EBNA-1 p72 45 kDal n Test Principle and Procedure 1st. Incubation: A test strip loaded with EBV antigens is incubated with diluted serum or plasma in a dish for 3 h. Wash 4 times E E E 2nd. Incubation: Peroxidase conjugated anti-human antibodies (IgG, IgM or IgA specific) are added. Incubate for 1 h. Wash 4 times 3rd. Incubation: 5 - 10 minutes after addition of the coloring solution, insoluble colored bands develop at the sites on the test strips occupied by antibodies. n Evaluation The diagnostic value of the recomBlot EBV, supplemented by the recombinant antigen p18 (VCA), was investigated in two different studies on two different collectives of sera (Table1 + Table 2). The test results supply evidence for the fact, that the addition of the p18 antigen makes it possible to estimate the EBV status of a patient in most cases with the recomBlot IgG strip only. Table 1: Normal collective (healthy students, n = 272) Serum group Number Interpretation with recomBlot EBV IgG I - anti-EBNA-1 and/or anti-p18-IgG antibodies 234 (86 %) II - no anti-EBNA-1 or anti-p18-IgG antibodies 13 (5 %) possible primary EBV infection (detection of anti-EBV-IgM antibodies recommended!) III - no anti-EBV-IgG antibodies 25 (9 %) no EBV antibodies detectable (second serum probe recommended!) Group I postacute or past EBV infection gp250/350 (MA) p54 (EA) p72 (EBNA-1) p138 (EA) p23 (VCA) p18 (VCA) 53 (23 %) 66 (28 %) 225 (97 %) 97 (42 %) 227 (98 %) 228 (98 %) IgG Table 2: Sera suspicious for a primary EBV infection because of ELISA results (n = 199) Serum group Number Interpretation with recomBlot EBV IgG, IgM I - no anti-EBNA-1 or anti-p18-IgG antibodies - anti-EA-IgG and/or IgM antibodies 154 (77 %) primar y EBV infection II - anti-EBNA-1 and/or anti-p18-IgG antibodies 38 (19 %) postacute or past EBV infection III - no anti-EBNA-1 or anti-p18-IgG antibodies - no anti-EA-IgG and/or IgM antibodies - no anti-EBV-IgG and IgM antibodies 7 (4 %) Group I borderline or atypic band pattern or no EBV antibodies detectable (second serum probe recommended!) gp250/350 (MA) p54 (EA) p72 (EBNA-1) p138 (EA) p23 (VCA) p18 (VCA) IgG 5 (3 %) 111 (72 %) 0 137 (89 %) 83 (54 %) 0 IgM 99 (64 %) 97 (63 %) 2 (1 %) 118 (77 %) 54 (35 %) 91 (59 %) n Storage and Shelf Life n Commercial Product At 4 °C 18 months from the time of production Article No. 4502 recomBlot EBV IgG Reagents for 20 determinations Article No. 4503 recomBlot EBV IgM/IgA Reagents for 20 determinations pirbebe004a MIKROGEN GmbH Floriansbogen 2-4 · D-82061 Neuried · Germany · Tel.: +49 (0)89 54801-0 · Fax: +49 (0)89 54801-100 Internet: www.mikrogen.de · eMail: [email protected]