* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Macromolecule Notes
Nucleic acid analogue wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Photosynthesis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
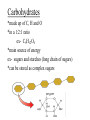
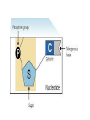
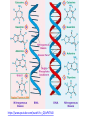
Buffers- weak acids or bases that resist pH changes *aid in keeping pH regulated in cells Vocabulary activity CUT AND GLUE TERM & MATCHING DEFINITION IN YOUR PDN BOOK Carbon Compounds Organic chemistry- the study of compounds that contain bonds between carbon (C) atoms *C has four valence e- (e- in outermost energy level) *can form four covalent bonds *can have single, double or triple bonds *C bonds with H, O, N, P and S *C can form long chains and complex molecules Carbon Compounds Macromolecules -giant molecules -made of many smaller molecules -formed by polymerization- large molecules are formed by joining smaller ones together monomers- smaller units polymers- larger units formed -Monomers are joined together by dehydration synthesis -Removes a water molecule to form a polymer Hydrolysis- water is added to separate a polymer into its monomers. Groups of organic compounds in living things: 1. carbohydrates 2. lipids 3. nucleic acids 4. proteins Carbohydrates *made up of C, H and O *in a 1:2:1 ratio ex- C6H12O6 *main source of energy ex- sugars and starches (long chain of sugars) *can be stored as complex sugars monomers = monosaccharide- single sugar molecules ex- glucose, galactose and fructose polymers = polysaccharides- larger molecule formed from many monosaccharides ex- glycogen (excess stored sugar) and cellulose (found in wood) https://www.youtube.com/watch?v=4bXkdny-wjM Lipids *made up of C and H and some O *not soluble in water (do not dissolve) *store energy and form membranes monomers = glycerol and fatty acids polymer = fats, oils and waxes page 46 fig 2-14: LIPIDS Saturated vs Unsaturated Fatty Acids saturated- all single bonds which leads to maximum # of H unsaturated- at least one double bond between two carbon atoms -not loaded with Hydrogen https://www.youtube.com/watch?v=0kWI2datvFY Nucleic acids *macromolecules containing H, O, N,C and P *store and transmit hereditary or genetic info monomers = nucleotides -consists of 5-C sugar, a phosphate group and a nitrogenous base polymer = nucleic acid (DNA/RNA) https://www.youtube.com/watch?v=_QDxPV7Si6I Proteins *contain C, N, H and O *have an amino group and a carboxyl group *most diverse macromolecule because of Rgroups *regulates cell processes, control reaction rates, transport substances and fight disease monomers = amino acids polymer = protein Amino Acids and Proteins general structure alanine https://www.youtube.com/watch?v=-WFWyRIb4Yo serine Chemical Reactions and Enzymes chemical reaction- process that changes one set of chemicals into another -atoms are rearranged -some reactions are fast ex: something blowing up -some are slow ex: rust forming ex: H2CO3 CO2 + H2O reactants- elements or compounds that enter a reaction (in ex. CO2 and H2O) products- elements or compounds formed during reaction (in ex. H2CO3) -chemical reactions involve the breaking and making of bonds -the number of each element you begin with must be the same as you end with ex: C O H CO2 + H2O H2CO3 reactants products 1 1 3 3 2 2 -Energy is either released or absorbed when bonds are made or broken ex: we release energy when we metabolize our food Activation energy- energy needed to get a reaction started -If the activation energy cannot be reached there will not be a reaction activation energy products reactant s activation energy reactant s products Catalysts- substances that speed up the rate of a chemical reaction -lowers activation energy Enzymes- proteins that speed up reactions in cells *biological catalysts reaction pathway without enzyme activation energy without enzyme reactant s reaction pathway with enzyme activatio n energy with enzyme products substrates- reactants of enzymecatalyzed reactions -substrates bind with enzymes -enzymes are specific- can only bond with one type of substrate -like a lock and key Enzymes jobs include: 1.regulating chemical pathways 2.making materials that cells need 3.releasing energy 4. transferring info enzy me gluco se substra tes produc ADP ts glucose -6phosph ate products are released ATP active site enzymesubstrate complex substrates are converted into products substra tes bind to enzyme