* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Immunocompetence of Schwann Cells
Survey
Document related concepts
DNA vaccination wikipedia , lookup
Hygiene hypothesis wikipedia , lookup
Lymphopoiesis wikipedia , lookup
Immune system wikipedia , lookup
Sjögren syndrome wikipedia , lookup
Immunosuppressive drug wikipedia , lookup
Adaptive immune system wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Molecular mimicry wikipedia , lookup
Innate immune system wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Transcript
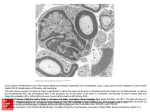
Im mmun nocom mpeten nce off Schw wann Cells G. Meyer Zu Hö örste, MD;; W. Hu, MD; M Hans-P P. Hartungg, MD; H. C C. Lehman nn, MD; B. C. C Kieseierr, MD No one iinvolved in the planning off this CME acttivity has any y relevant finaancial relation nships to discllose. Autthors/faculty have nothingg to disclose. CM ME is availab ble 1/15/2011 - 1/5/2014 Copy yright: 20008 an Associatio on of Neurom muscular and d Electrodiaggnostic Mediicine America 2621 Superior S Dr NW N Rochestter, MN 55901 deas and opin nions in this Jo ournal Review w are solely th those of the auuthor and do not necessariily The id represent th hose of the AA ANEM Produ uct: JR16 INVITED REVIEW ABSTRACT: Schwann cells are the myelinating glial cells of the peripheral nervous system that support and ensheath axons with myelin to enable rapid saltatory signal propagation in the axon. Immunocompetence, however, has only recently been recognized as an important feature of Schwann cells. An autoimmune response against components of the peripheral nervous system triggers disabling inflammatory neuropathies in patients and corresponding animal models. The immune system participates in nerve damage and disease manifestation even in non-inflammatory hereditary neuropathies. A growing body of evidence suggests that Schwann cells may modulate local immune responses by recognizing and presenting antigens and may also influence and terminate nerve inflammation by secreting cytokines. This review summarizes current knowledge on the interaction of Schwann cells with the immune system, which is involved in diseases of the peripheral nervous system. Muscle Nerve 37: 3–13, 2008 THE IMMUNOCOMPETENCE OF SCHWANN CELLS GERD MEYER ZU HÖRSTE, MD, WEI HU, MD, HANS-PETER HARTUNG, MD, HELMAR C. LEHMANN, MD, and BERND C. KIESEIER, MD Department of Neurology, Heinrich-Heine-University, Moorenstrasse 5, 40225 Düsseldorf, Germany Accepted 26 July 2007 Schwann cells myelinate and support axons of the peripheral nervous system (PNS) and they can become targets of a primary and secondary immune response. We review pathogenetic concepts and models of immune reactions in the PNS and elaborate on specific immune functions of Schwann cells in greater detail. Human inflammatory neuropathies are caused by an immune response mounted against still incompletely characterized autoantigens within the PNS. The clinical picture ranges from Guillain–Barré syndrome (GBS)35 to chronic inflammatory demyelinating polyneuropathy (CIDP).37 Different subforms and distinct regional variants have been defined. Available for Category 1 CME credit through the AANEM at www. aanem.org. Abbreviations: BDNF, brain-derived neurotrophic factor; CIDP, chronic inflammatory demyelinating polyneuropathy; CMT, Charcot–Marie–Tooth (disease); EAN, experimental autoimmune neuritis; FasL, Fas ligand; GBS, Guillain–Barré syndrome; IL, interleukin; LPS, lipopolysaccharide; MAG, myelinassociated glycoprotein; MBP, myelin basic protein; MHC, major histocompatibility complex; NF-B, nuclear transcription factor-B; P0, myelin protein zero; PNS, peripheral nervous system; Rag-1, recombination activating gene-1; TLR, toll-like receptors; TNF, tumor necrosis factor Key words: antigen presentation; Charcot–Marie–Tooth disease; experimental autoimmune neuritis; Guillain–Barré syndrome; inflammatory neuropathy; Schwann cells Correspondence to: B. C. Kieseier; e-mail: [email protected] © 2007 Wiley Periodicals, Inc. Published online 6 September 2007 in Wiley InterScience (www.interscience. wiley.com). DOI 10.1002/mus.20893 Immunocompetence of Schwann Cells Depending on the primary target of the autoimmune reaction, acute inflammatory neuropathies are classified either as the most common acute inflammatory demyelinating polyneuropathy (AIDP) or as the less frequent acute motor (and sensory) axonal neuropathies (AMAN and AMSAN, respectively). Miller Fisher syndrome (MFS) denotes the acute regional variant involving predominantly cranial nerves. Multifocal motor neuropathy (MMN) is a multifocal variant of CIDP involving mainly motor fibers.61 A consistent model of how these disorders develop has been established for certain subforms. Even under normal conditions, there may be autoreactive T lymphocytes recognizing peripheral nerve antigens in the systemic immune compartment, but their continuous suppression ensures self-tolerance. During a minor infectious disease these lymphocytes become activated upon encountering microbial epitopes. The hallmark finding that microbial epitopes can resemble endogenous peripheral nerve antigens has been termed “molecular mimicry.”133,134,136 Autoreactive T lymphocytes stimulate B cells to produce autoantibodies, which in turn can block nerve conduction,119,128 activate complement,102,115 and facilitate a macrophage attack in the peripheral nerve. Activated T cells transgress the blood–nerve barrier and provoke local inflammation by proinflammatory cytokines.54 Attracted macro- MUSCLE & NERVE January 2008 3 phages act as antigen-presenting cells, release toxic mediators, and directly damage myelinating Schwann cells and axons.52 Long-term disability is mainly determined by the degree of axonal degeneration. The immune response may eventually be terminated by increased T-cell apoptosis, as demonstrated in animal models of inflammatory neuropathies.31,32 PATHOMECHANISMS OF AUTOIMMUNE DISORDERS OF THE PNS The pathological hallmark of the demyelinating subtypes of inflammatory neuropathies is the infiltration of the PNS by lymphocytes and macrophages, which results in multifocal demyelination, predominantly around blood vessels.33 Macrophages actively strip off myelin lamellae from axons, induce vesicular disruption of the myelin sheath, and phagocytose both intact and damaged myelin, as shown by electron microscopy.34,52 Macrophages, numerous as resident cells in the endoneurium, represent the predominant cell population in the inflamed PNS, and they reside in spinal roots as well as in more distal segments of the affected nerves.51 Pathological studies suggest that the early invasion of the PNS by leukocytes is crucial in the pathogenesis of inflammatory demyelination. Circulating autoreactive T cells need to be activated in the periphery in order to cross the blood–nerve barrier and incite a local inflammatory response. Breakdown of the blood–nerve barrier is one of the earliest morphologically demonstrable events in lesion development in the animal model of demyelinating GBS.32 It remains elusive how the cascade of autoimmune responses targeting PNS structures is ignited. One pathogenic mechanism of special relevance to autoimmune neuropathies is “molecular mimicry.” In a proportion of patients with GBS, epitopes shared between the enteropathogen Campylobacter jejuni, cytomegalovirus, or Haemophilus influenzae and nerve fibers have been identified as targets for aberrant cross-reactive B-cell responses.53 In a recent study, antibody responses to the ganglioside GM1 were linked to axonal and motor injury, as seen in one clinical variant of GBS136 and in an experimental model induced in rabbits by active immunization with this glycoconjugate.135 This provides a conclusive pathomechanism of axonal subtypes of GBS. Despite long-term efforts, no such conclusive mechanistic models have been confirmed in the demyelinating GBS subtypes. Myelin protein–like structures have not been proven to exist in microbes. 4 Immunocompetence of Schwann Cells Antibodies against various components of the myelin sheath are found in only a fraction of demyelinating GBS patients and even in some healthy subjects. There may be several explanations for this observation. First, myelin components may not be the target of the immune response in demyelinating GBS. Second, antibodies against myelin components are important, but may not be the main orchestrators of the immune response in demyelinating GBS. A primarily cellular immune response may not be controlled sufficiently by screening for antibodies. Thus, it is impossible to prove that an animal model of demyelinating GBS actually reflects human disease, because the definite immunopathogenetic mechanisms and target antigens of this disease in humans still have not been identified. From a pathological, electrophysiological, and clinical point of view, myelin-induced experimental autoimmune neuritis (EAN) is a good model of acute demyelinating GBS. Furthermore, plasma exchange and intravenous immunoglobulins, the clinical mainstays of human GBS therapy, are effective in myelin-induced EAN. GBS should be defined as an organ-specific immune-mediated disorder emerging from a synergistic interaction of cell-mediated and humoral immune responses to still incompletely characterized peripheral nerve antigens.36,106 Schwann cells represent one of the major targets in immune-mediated disorders of the PNS. ANIMAL MODELS OF INFLAMMATORY NEUROPATHIES Many features of human inflammatory neuropathies are accurately recapitulated in EAN, an animal model of GBS that has greatly extended our knowledge of the underlying pathological mechanisms. Table 1 provides an overview of the available animal models. These models are explained in what follows. A variety of antigens can induce immunological responses against peripheral nerves in different species, including rats, mice, rabbits, monkeys, and guinea pigs (Table 1). Immunization with Schwanncell or myelin components generates demyelinating neuropathy models, whereas axonal components elicit axonal damage representing axonal GBS subtypes (Table 1). In the most widely used GBS model, Lewis rats are immunized with peripheral myelin homogenates, myelin proteins, or derived peptides to develop EAN (Table 1).76 Like entire myelin homogenates, the major myelin adhesion molecule P0,82 and the fatty acid– binding protein P2,47 and their immunogenic peptides, elicit marked im- MUSCLE & NERVE January 2008 Table 1. Animal models of inflammatory neuropathies. Reference nos. Animal model Transfer Possible antigens Description Rat Lewis Active PNS myelin, P0, P2, P0(180–199), P2(53–78), P2(57–81) PMP22 PNS myelin ⫹ cyclosporine PNS myelin PNS myelin P0, P2, P0(180–199), P2(61–70), MAG Frequently used EAN models 1, 23, 76, 83 Mild course EAN CIDP-like chronic relapsing, not robust Less severe EAN course CIDP-like relapsing Rapid-onset EAN, CIDP-like relapsing if transferred repeatedly 28 78 41 46 76, 64 Varying effectiveness reported Mild course EAN Severe course EAN Peripheral and central demyelination Spontaneous autoimmune neuropathy CIDP-like 81, 137 121, 14 AT Spontaneous P0(180–199), P0(106–125) ⫹ PTx P2 Myelin ⫹ PTx (⫹IL-12) MBP — Active PNS myelin, P2 galactocerebroside First EAN model Chronic inflammation Active Active GM1 GD1b Acute motor axonal neuropathy Ataxic sensory neuronopathy Active Active PNS myelin PNS myelin, P2 BN, SD, BUF, Wistar Dark Agouti Lewis Mouse C57/B6 SJL BALB/c B7-2⫺/⫺ NOD Rabbit Rabbit Rabbit (axonal) Other Guinea pig Monkey Active Active Active Active AT Active Active 2 100 76 48 114 135 63 109 24 PNS, peripheral nervous system; EAN, experimental autoimmune neuritis; CIDP, chronic inflammatory neuropathy; P0, myelin protein zero; P2, myelin protein 2; PMP22, peripheral myelin protein of 22 kDa; BN, Brown–Norway rats; SD, Sprague–Dawley rats; BUF, Buffalo rats; MAG, myelin-associated glycoprotein; MBP, myelin basic protein; PTx, pertussis toxin; AT, adoptive transfer. munological responses. Less severe forms of rat EAN are triggered by peripheral myelin protein of 22 kDa.28,59 Alternatively, transfer of stimulated T lymphocytes that are reactive toward various myelin antigens, including myelin proteins P2,98 P0,68 and derived peptides, evokes adoptive transfer EAN in host animals (Table 1). Adoptive transfer of lymphocytes that are reactive against myelin-associated glycoprotein (MAG) generates mild peripheral nerve inflammation.129 Unlike rats, mice were generally thought not to be susceptible to immunization with peripheral myelin, but recent studies have challenged this view. Although myelin protein P2 induced only subclinical EAN in SJL mice, extended immunization protocols have allowed the induction of severe EAN by myelin homogenates in this strain (Table 1).14 Adoptive transfer EAN in BALB/c mice identified myelin basic protein (MBP) as an alternative neuritogenic antigen. The number of regulatory T cells differs between mouse strains,17 which might explain their differential autoimmune susceptibility, and a similar concept might be relevant for human autoimmune diseases.22 Generation of active EAN by two different Immunocompetence of Schwann Cells P0 peptides, P0(180 –199)137 and P0(106 –125),81 together with pertussis toxin adjuvant in C57/B6 mice, has recently been reported. This protocol might enable the study of peripheral nerve inflammation in genetically modified animals, which are mostly generated on this background, allowing us to further extend our knowledge of inflammatory neuropathies. The present data clearly highlight the fact that immunization with myelin components derived from Schwann cells as the cellular source can be used to induce an immune reponse against the PNS. SECONDARY IMMUNE REACTIONS IN NONINFLAMMATORY HEREDITARY NEUROPATHIES Even hereditary disorders of the peripheral nervous system trigger immune responses in the peripheral nerve. These responses have been suggested to be an important determinant of disease manifestation. Charcot–Marie–Tooth (CMT) disease, clinically termed hereditary motor and sensory neuropathy (HMSN), is the most common inherited peripheral neuropathy. Animal models of many subtypes of MUSCLE & NERVE January 2008 5 CMT have been generated and have vastly extended our knowledge of the underlying disease mechanisms.80 In the more frequent demyelinating CMT forms, a genetic defect results in progressive destruction of myelin sheaths and secondary axonal loss.117 One may easily conceive that such genetically determined defects of myelination can trigger a secondary immune response. Indeed, infiltration of lymphocytes and macrophages has been demonstrated in peripheral nerves of some human CMT patients,27,125 and corresponding animal models.7,57,104 Apart from this secondary immune response, however, a series of experimental studies have suggested immune cells as a primary cause for disease manifestation in genetically determined neuropathies. Mouse models of CMT subtypes CMT1B72 and CMTX5,80 were crossbred with mouse strains lacking functional T lymphocytes or with impaired macrophage function. Surprisingly, immune deficiency ameliorated mild, chronic demyelination in these models of inherited neuropathies. Impairment of immune function was achieved by abolishing functional T lymphocytes in mouse strains carrying null mutations of the recombination activating gene-1 (Rag-1)56,104 or the T-cell-receptor ␣-subunit gene.77 Macrophage function was impeded in mice lacking functional genes of macrophage colony stimulating factor16 and adhesion protein sialoadhesin preferentially expressed in macrophages.58 Demyelination in the Rag-1– deficient CMT1B model could be reconstituted by transfer of wild-type bone marrow,75 which excludes Schwann-cell intrinsic effects of Rag-1 deficiency. This effect, however, has not been shown in the most common CMT subtype, 1A, and, in contrast to its effect on mild chronic demyelination, immune deficiency was found to aggravate early-onset severe dysmyelination.7 Furthermore, myelinating cell cultures generated from a CMT1A animal model develop defects of myelination despite the obvious absence of immune cells in such a model.88 The efficacy of an anti-inflammatory treatment in CMT remains controversial.73 One possible explanation for the increased immune reaction in the CMT1B animal model may be reduced intrathymic induction of tolerance. A heterozygous knockout of the myelin protein zero (P0) gene determines this CMT subtype and this reduction in gene dose was demonstrated to increase Tcell reactivity to a P0 peptide.84 Genetically determined P0 deficiency in mice abolishes tolerance and causes the P0 protein to be recognized as a foreign antigen,124 which further supports autoimmune mechanisms in hereditary neuropathies. 6 Immunocompetence of Schwann Cells Finally, we have learned that macrophages and lymphocytes are required for myelin loss in models of at least certain forms of hereditary neuropathies. This raises the question of how immune cells “sense” the defectiveness of the myelin sheath and why they participate in its gradual destruction. Increased immunogenity due to altered protein composition or mechanical instability of the myelin sheath has been suggested. Molecules required for antigen presentation could communicate this information from Schwann cells to immune cells and one may speculate that a myelin protein mutant Schwann cell would not demyelinate if it lacked the molecules required for antigen processing and presentation. We have described the intimate interaction between myelinating Schwann cells and immune cells and its importance for various peripheral nerve disorders. In what follows, we specifically elaborate on the expanding recognition of Schwann cells as immunocompetent cells that form part of the local immune circuitry within the peripheral nerve. The present data suggest that the entire spectrum of an immune response can be displayed by Schwann cells, which includes recognition of antigens; presentation of antigens; mounting of an immune response; and, finally, terminating the immune response within the inflamed peripheral nerve (Fig. 1). ANTIGEN RECOGNITION BY SCHWANN CELLS Two types of responses to invading organisms can take place in the mammalian immune system: an acute response launched within hours and a delayed response occurring within days. The immediately responding system is called the innate immune system, and it evolves stereotypically and at the same intensity regardless of how often the infectious agent is encountered. The strategy of the innate immune response is not to recognize every possible antigen, but rather to focus on a few highly conserved structures present in large groups of microorganisms. These structures are referred to as pathogen-associated molecular patterns, and the corresponding receptors of the innate immune system are called patternrecognition receptors.45 Examples of pathogen-associated molecular patterns are bacterial lipopolysaccharide (LPS), peptidoglycan, and bacterial DNA. Although chemically quite distinct, these molecules display common features. They are only produced by microbial pathogens and not by their host. They generally represent invariant structures shared by large classes of pathogens, and they are usually relevant for the survival or pathogenicity of microorganisms.79 MUSCLE & NERVE January 2008 FIGURE 1. Immunocompetence of Schwann cells. (a) Facultative antigen presentation: Schwann cells are able to process and present endogenous antigens to immune cells. Schwann cells can express the major histocompatibility (MHC) class I molecules as well as MHC class II molecules; in addition, co-stimulatory molecules are expressed on the cell surface. (b) Facultative antigen recognition: via toll-like receptors, Schwann cells can recognize antigens, such as LPS, activating the nuclear transcription factor NF-B. (c) Regulation of an immune response: Schwann cells, after stimulation, secrete various soluble factors, such as cytokines, growth factors, and other immune mediators. (d) Termination of an immune response: via the interaction of Fas and FasL, Schwann cells can drive inflammatory T cells into apoptosis. LPS, lipopolysaccharide; GDNF, glial cell line– derived neurotrophic factor; PGE2, prostaglandin E2; IL, interleukin; TNF, tumor necrosis factor; TGF, transforming growth factor. The family of toll-like receptors (TLRs) belongs to the group of pattern-recognition receptors that recognize specific conserved components of microbes,118 such as LPS, and that have been implicated to play a critical role in various inflammatory disorders.20,40 To date, 11 TLRs have been identified in humans and 7 in rats.43 LPS, a major component of the outer membrane of gram-negative bacteria, which appears to be a relevant antigen triggering immune-mediated demyelination of the PNS,53,60 is recognized by TLR-4. TLRs are usually found on antigen-presenting cells, such as dendritic cells. TLR-2 has been shown to be expressed constitutively on primary human and rat Schwann cells and has been invoked as a target receptor for Mycobacterium leprae.38,89 Under inflammatory conditions, expression of various TLRs, especially TLR-4, is inducible on rat Schwann cells in vitro. Selective stimulation of TLR-4 on Immunocompetence of Schwann Cells Schwann cells with LPS has been shown to elicit the production of various inflammatory mediators such as chemokines, protease inhibitors, and growth factors.42 Thus, in aggregate, these findings suggest that Schwann cells can detect LPS fragments and may act as a link between innate and acquired immunity via TLR-4 activation in the inflamed PNS. Recent studies have also revealed that inflammatory Schwann-cell activation can be induced via TLR-2 and TLR-3.65 SCHWANN CELLS AS FACULTATIVE ANTIGENPRESENTING CELLS The task of displaying the antigens of cell-associated microbes for recognition by T lymphocytes is performed by specific molecules that are encoded by genes comprising the major histocompatibility complex (MHC). The physiological function of MHC molecules is the presentation of peptides to T cells. MUSCLE & NERVE January 2008 7 Two types of MHC gene products can be distinguished, class I MHC molecules and class II MHC molecules, which differ functionally. Each MHC molecule consists of an extracellular peptide-binding cleft or groove and a pair of immunoglobulinlike domains, containing binding sites for the T-cell surface markers CD4 and CD8.71,122 By transmembrane domains, MHC molecules are anchored to the cell surface. MHC molecules exhibit a broad specificity for peptide binding, whereas the fine specificity of antigen recognition resides mostly in the T-cell receptor.13 In general, endogenous cytosolic peptides are presented via MHC class I molecules to CD8⫹ T cells, whereas mainly exogenously derived peptides generated in vesicles are bound to MHC class II molecules and recognized by CD4⫹ T lymphocytes.97,127 The two classes of MHC molecules are expressed differentially on cells. All nucleated cells exhibit MHC class I molecules, although hematopoietic cells express them at the highest densities. MHC class II molecules, in contrast, are normally only expressed on professional antigen-presenting cells, such as macrophages, dendritic cells, and B lymphocytes. The levels of both class I and II molecules can be markedly upregulated by cytokines, particularly interferon-␥ and tumor necrosis factor (TNF)-␣. Macrophages are the predominant professional antigen-presenting cells in EAN. These macrophages in EAN shift from resident endoneural in the early disease stages to a hematopoietic origin in later stages.85 Apart from such professional antigen-presenting cells, other cell types may acquire the ability to process and present antigens in inflammatory conditions, thus representing facultative antigen-presenting cells. Human and rat Schwann cells in vitro constitutively express low levels of MHC class I but not MHC class II.6,9,30,66,101 Significant numbers of MHC class II molecules can be detected on Schwann cells in the presence of activated T lymphocytes upon stimulation with the proinflammatory cytokine interferon-␥, which can be synergistically increased by the addition of TNF-␣.6,30,55,66,123 Moreover, a number of molecules that are prominent in the intracellular processing cascade of peptides prone to MHC presentation can be visualized in Schwann cells in vitro and in vivo (Meyer zu Hörste and Kieseier, unpublished observations). In human nerve biopsies from patients with GBS and its chronic variant CIDP, Schwann cells stained positive for MHC class II, suggesting that these cells may indeed act as facultative antigen-presenting cells in immune-mediated disorders of the PNS.92,93 Antigen-presenting cells are characterized by their ability 8 Immunocompetence of Schwann Cells to phagocytose exogenous antigen and its degradation to antigenic peptides, which can be expressed in the cleft of the MHC molecule.15,122 Schwann cells in vitro have been shown to present foreign and exogenous antigens, such as MBP, to antigen-specific syngeneic T-cell lines.10,130 Moreover, the presentation of endogenous antigen, such as the myelin component P2, by Schwann cells via MHC class II can restimulate resting antigen-specific CD4⫹ T-cell lines.67 Non-endogenous antigen can also be presented by Schwann cells, which may be of special relevance in leprosy. On a global scale, leprosy is one of the major causes of peripheral neuropathy, with sensory loss being its foremost symptom. Neuropathy in leprosy mainly affects pain and temperature sensation from small cutaneous nerves, resulting in painless injury and mutilation.99 The causal agent of the disease, Mycobacterium leprae, shows a remarkable preference for invading and proliferating in nonmyelinating Schwann cells.96 Schwann cells present M. leprae– derived antigens to lymphocytes in an MHC II– dependent manner,26 whereas CD8⫹ lymphocytes lyse M. leprae–infected Schwann cells in vitro.112 A different study showed M. leprae– derived antigen presentation by Schwann cells to CD4⫹ lymphocytes.110 This MHC II–mediated interaction resulted in Schwann-cell destruction. Thus, apart from other mechanisms described,95,120 antigen presentation by Schwann cells evolves as an important mechanism of nerve damage in this infectious neuropathy. For optimal T-cell activation and differentiation to occur, at least two distinct signals delivered during the interaction with an antigen-presenting cell are required. These include antigen-specific signaling via MHC and signaling through costimulatory molecules. If the T cell does not receive adequate costimulation, it is rendered anergic or undergoes apoptosis. Thus, the costimulation signal is central to T-cell activation and survival.21,105 Recent data suggest that BB-1, a member of the family of costimulatory molecules, can be detected on non-myelinating Schwann cells and appears to be upregulated on myelinating Schwann cells in nerve biopsies from CIDP patients. This further indicates that Schwann cells possess the cellular components required to act as facultative antigen-presenting cells in the inflamed PNS.86,94 The extent to which these cells may propagate a T-cell response via antigen presentation in situ needs to be elucidated in greater detail. It needs to be investigated whether Schwann cells can suppress such a response in vivo as well as in vitro.70 In any case, it is an appealing concept that Schwann MUSCLE & NERVE January 2008 cells actively control the local T-cell response within the peripheral nerve by acting as facultative antigenpresenting cells. SCHWANN CELLS AS REGULATORS OF AN AUTOIMMUNE RESPONSE For a long time, cytokines, as mediators of an immune response within the peripheral nerve, were considered to be the exclusive product of inflammatory cells. Nowadays, there is a large body of evidence implying that Schwann cells can produce and secrete a wide variety of cytokines, which could act as immunomodulators.70 Interleukin (IL)-1, a cytokine relevant in the initiation of an immune response, can be produced by cultured Schwann cells.8 Other proinflammatory cytokines, such as IL-6,8,12,29,87 TNF-␣,8,12,29,87,116,126 and transforming growth factor,103,113 are generated and released by Schwann cells under certain conditions, some in vitro and others in vivo.50,74,108 Schwann cells are able to regulate the production of proinflammatory cytokines, at least in part, in a specific autocrine manner, as shown for IL-1.107,108 The specific receptors for some of the cytokines, such as TNF receptor, are constitutively expressed on Schwann cells, rendering these cells susceptible to, for example, a TNF-␣ response.11,90 Other proinflammatory and immunoregulatory mediators, such as prostaglandin E2, thromboxane A2, and leukotriene C4, are synthesized in large amounts by Schwann cells, and may regulate the immune cascade within the inflamed PNS.18,19 Further mediators produced by Schwann cells include osteopontin, which is constitutively expressed and can be easily induced in Schwann cells,3 as well as glial cell line– derived neurotrophic factor, a known survival factor for neurons, and glial cell line– derived neurotrophic factor family receptor ␣-1, displayed on the surface of Schwann cells.39,44 Nuclear transcription factor-B (NF-B) plays a pivotal role in the regulation of the host innate antimicrobial response. It governs the expression of many immunological mediators, including cytokines, their receptors, and components of their signal transduction. Recent studies have suggested that two NF-B complexes, p65/p50 and p50/p50, can be activated and regulated in human Schwann cells under certain conditions.91 Interestingly, the natural inhibitor of NF-B, IB, can be detected in large amounts in Schwann cells.4 These observations further point to an active rather than a passive role for Schwann cells in the inflamed PNS. Apart from engaging such cellular factors, Schwann cells can modulate local immune reactivity by humoral mecha- Immunocompetence of Schwann Cells nisms. One pathway may operate through the production and release of nitric oxide (NO), a multipotential mediator with neurotoxic and immunosuppressive properties. Schwann cells are endowed with inducible nitrite oxide synthase, which is upregulated after the stimulation with proinflammatory cytokines.131 Taken together, a large number of pro- and antiinflammatory mediators can be induced and released by Schwann cells. The extent to which these mediators have a significant impact on the clinical course of an immune-mediated disorder in the PNS clearly warrants further investigation. In animal models of inflammatory diseases of the central nervous system, neurotrophic cytokines and neurotrophins have been suggested as important regulators of axonal degeneration and of resulting clinical impairment.69 Most interestingly, brainderived neurotrophic factor (BDNF), originating from inflammatory cells, has been demonstrated to mediate neuroprotective effects,49,111 which emphasizes the concept of neuroprotective autoimmunity. Few studies, however, support a comparable concept in inflammatory diseases of the peripheral nervous system. Except for reduced urinary bladder weight as a possible indirect sign of reduced autonomic dysregulation, BDNF treatment did not significantly influence clinical disease manifestation in rat EAN.25 SCHWANN CELLS AS TERMINATORS OF THE AUTOIMMUNE RESPONSE In order to control the massive expansion of cellular and soluble immune mediators within the target tissue, certain mechanisms must operate with high fidelity to regulate the immune response. Once the target antigen has been eliminated or infection abated, the activated effector cells are no longer needed. When the antigenic stimulus is no longer present, the cells will succumb to programmed cell death or apoptosis.32 The survival of lymphocytes depends on a delicate balance between death-promoting and deathinhibiting factors. Various mechanisms can induce apoptosis. It can, for example, be affected through the interaction of the cell surface receptor Fas on T cells with its ligand, FasL, a member of the TNF family.62 Schwann cells reveal surface expression of FasL after stimulation with proinflammatory cytokines in vitro. Functional analysis indicates that the interaction between Fas on T cells and FasL on Schwann cells promotes apoptosis of T lymphocytes. This raises the possibility that Schwann cells are MUSCLE & NERVE January 2008 9 important in terminating the immune response in the inflamed PNS.132 11. SCHWANN CELLS AS IMMUNOCOMPETENT CELLS In summary, our current knowledge of the potential immunocompetence of Schwann cells suggests that these cells can induce an immune response within the peripheral nerve via pattern-recognition receptors, but can also trigger a T-cell response via the presentation of antigen fragments on MHC class II molecules in the context of costimulatory molecules. Through the release of immunomodulators, Schwann cells could regulate the immune reaction in situ and, by inducing apoptosis, appear even to terminate an ongoing immune response. This evidence collected in recent years indicates that the many functions of Schwann cells go far beyond forming the myelin sheath. This work was supported by grants from the German Research Foundation (GRK320) and the Research Commission of the Heinrich-Heine-University, Düsseldorf (FoKo) (to G.M.z.H, H.L., and B.C.K.). 12. 13. 14. 15. 16. 17. 18. REFERENCES 1. Abbas N, Zou LP, Pelidou SH, Winblad B, Zhu J. Protective effect of Rolipram in experimental autoimmune neuritis: protection is associated with down-regulation of IFN-gamma and inflammatory chemokines as well as up-regulation of IL-4 in peripheral nervous system. Autoimmunity 2000;32: 93–99. 2. Abromson-Leeman S, Bronson R, Dorf ME. Experimental autoimmune peripheral neuritis induced in BALB/c mice by myelin basic protein-specific T cell clones. J Exp Med 1995; 182:587–592. 3. Ahn M, Lee Y, Moon C, Jin JK, Matsumoto Y, Koh CS, et al. Upregulation of osteopontin in Schwann cells of the sciatic nerves of Lewis rats with experimental autoimmune neuritis. Neurosci Lett 2004;372:137–141. 4. Andorfer B, Kieseier BC, Mathey E, Armati P, Pollard J, Oka N, et al. Expression and distribution of transcription factor NF-kappaB and inhibitor IkappaB in the inflamed peripheral nervous system. J Neuroimmunol 2001;116:226 –232. 5. Anzini P, Neuberg DH, Schachner M, Nelles E, Willecke K, Zielasek J, et al. Structural abnormalities and deficient maintenance of peripheral nerve myelin in mice lacking the gap junction protein connexin 32. J Neurosci 1997;17:4545– 4551. 6. Armati PJ, Pollard JD, Gatenby P. Rat and human Schwann cells in vitro can synthesize and express MHC molecules. Muscle Nerve 1990;13:106 –116. 7. Berghoff M, Samsam M, Muller M, Kobsar I, Toyka KV, Kiefer R, et al. Neuroprotective effect of the immune system in a mouse model of severe dysmyelinating hereditary neuropathy: enhanced axonal degeneration following disruption of the RAG-1 gene. Mol Cell Neurosci 2005;28:118 –127. 8. Bergsteinsdottir K, Kingston A, Mirsky R, Jessen KR. Rat Schwann cells produce interleukin-1. J Neuroimmunol 1991; 34:15–23. 9. Bergsteinsdottir K, Kingston A, Jessen KR. Rat Schwann cells can be induced to express major histocompatibility complex class II molecules in vivo. J Neurocytol 1992;21:382–390. 10. Bigbee JW, Yoshino JE, DeVries GH. Morphological and proliferative responses of cultured Schwann cells following 10 Immunocompetence of Schwann Cells 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. rapid phagocytosis of a myelin-enriched fraction. J Neurocytol 1987;16:487– 496. Bonetti B, Valdo P, Stegagno C, Tanel R, Zanusso GL, Ramarli D, et al. Tumor necrosis factor alpha and human Schwann cells: signalling and phenotype modulation without cell death. J Neuropathol Exp Neurol 2000;59:74 – 84. Bourde O, Kiefer R, Toyka KV, Hartung HP. Quantification of interleukin-6 mRNA in wallerian degeneration by competitive reverse transcription polymerase chain reaction. J Neuroimmunol 1996;69:135–140. Bromley SK, Burack WR, Johnson KG, Somersalo K, Sims TN, Sumen C, et al. The immunological synapse. Annu Rev Immunol 2001;19:375–396. Calida DM, Kremlev SG, Fujioka T, Hilliard B, Ventura E, Constantinescu CS, et al. Experimental allergic neuritis in the SJL/J mouse: induction of severe and reproducible disease with bovine peripheral nerve myelin and pertussis toxin with or without interleukin-12. J Neuroimmunol 2000;107: 1–7. Cambier JC, Littman DR, Weiss A. Antigen presentation to T lymphocytes. In: Janeway JCA, Travers P, Walport M, Shlomchik MJ, editors. Immunobiology. New York: Garland Publishing; 2001. Carenini S, Maurer M, Werner A, Blazyca H, Toyka KV, Schmid CD, et al. The role of macrophages in demyelinating peripheral nervous system of mice heterozygously deficient in p0. J Cell Biol 2001;152:301–308. Chen X, Oppenheim JJ, Howard OM. BALB/c mice have more CD4⫹CD25⫹ T regulatory cells and show greater susceptibility to suppression of their CD4⫹CD25- responder T cells than C57BL/6 mice. J Leukoc Biol 2005;78:114 –121. Constable AL, Armati PJ, Toyka KV, Hartung HP. Production of prostanoids by Lewis rat Schwann cells in vitro. Brain Res 1994;635:75– 80. Constable AL, Armati PJ, Hartung HP. DMSO induction of the leukotriene LTC4 by Lewis rat Schwann cells. J Neurol Sci 1999;162:120 –126. Cook DN, Pisetsky DS, Schwartz DA. Toll-like receptors in the pathogenesis of human disease. Nat Immunol 2004;5: 975–979. Coyle AJ, Gutierrez-Ramos JC. The expanding B7 superfamily: increasing complexity in costimulatory signals regulating T cell function. Nat Immunol 2001;2:203–209. Dejaco C, Duftner C, Grubeck-Loebenstein B, Schirmer M. Imbalance of regulatory T cells in human autoimmune diseases. Immunology 2006;117:289 –300. Deretzi G, Zou LP, Pelidou SH, Nennesmo I, Levi M, Wahren B, et al. Nasal administration of recombinant rat IL-4 ameliorates ongoing experimental autoimmune neuritis and inhibits demyelination. J Autoimmun 1999;12:81– 89. Eylar EH, Toro-Goyco E, Kessler MJ, Szymanska I. Induction of allergic neuritis in rhesus monkeys. J Neuroimmunol 1982;3:91–98. Felts PA, Smith KJ, Gregson NA, Hughes RA. Brain-derived neurotrophic factor in experimental autoimmune neuritis. J Neuroimmunol 2002;124:62– 69. Ford AL, Britton WJ, Armati PJ. Schwann cells are able to present exogenous mycobacterial hsp70 to antigen-specific T lymphocytes. J Neuroimmunol 1993;43:151–159. Gabreels-Festen AA, Joosten EM, Gabreels FJ, Jennekens FG, Janssen-van Kempen TW. Early morphological features in dominantly inherited demyelinating motor and sensory neuropathy (HMSN type I). J Neurol Sci 1992;107:145–154. Gabriel CM, Hughes RA, Moore SE, Smith KJ, Walsh FS. Induction of experimental autoimmune neuritis with peripheral myelin protein-22. Brain 1998;121:1895–1902. Gadient RA, Otten U. Postnatal expression of interleukin-6 (IL-6) and IL-6 receptor (IL-6R) mRNAs in rat sympathetic and sensory ganglia. Brain Res 1996;724:41– 46. Gold R, Toyka KV, Hartung HP. Synergistic effect of IFNgamma and TNF-alpha on expression of immune molecules MUSCLE & NERVE January 2008 31. 32. 33. 34. 35. 36. 37. 38. 39. 40. 41. 42. 43. 44. 45. 46. 47. 48. 49. 50. 51. 52. and antigen presentation by Schwann cells. Cell Immunol 1995;165:65–70. Gold R, Hartung HP, Lassmann H. T-cell apoptosis in autoimmune diseases: termination of inflammation in the nervous system and other sites with specialized immune-defense mechanisms. Trends Neurosci 1997;20:399 – 404. Gold R, Archelos JJ, Hartung H-P. Mechanisms of immune regulation in the peripheral nervous system. Brain Pathol 1999;9:343–360. Gold R, Stoll G, Kieseier BC, Hartung HP, Toyka KV. Experimental autoimmune neuritis. In: Dyck PJ, Thomas PK, editors. Peripheral neuropathy, 4th ed. (vol. 2). Philadelphia: Elsevier Saunders; 2005. Griffin JW, George R, Ho T. Macrophage systems in peripheral nerves. A review. J Neuropathol Exp Neurol 1993;52: 553–560. Griffin JW, Sheikh K. The Guillain–Barré syndromes. In: Dyck PJ, Thomas PK, editors. Peripheral neuropathy, 4th ed. (vol. 2). Philadelphia: Elsevier Saunders; 2005. p 2197–2220. Hahn AF. Guillain–Barré syndrome. Lancet 1998;352:635– 641. Hahn AF, Hartung HP, Dyck PJ. Chronic inflammatory demyelinating polyradiculoneuropathy. In: Dyck PJ, Thomas PK, editors. Peripheral neuropathy, 4th ed. (vol. 2). Philadelphia: Elsevier Saunders; 2005. p 2221–2254. Harboe M, Aseffa A, Leekassa R. Challenges presented by nerve damage in leprosy. Lepr Rev 2005;76:5–13. Hase A, Saito F, Yamada H, Arai K, Shimizu T, Matsumura K. Characterization of glial cell line-derived neurotrophic factor family receptor alpha-1 in peripheral nerve Schwann cells. J Neurochem 2005;95:537–543. Hoebe K, Janssen E, Beutler B. The interface between innate and adaptive immunity. Nat Immunol 2004;5:971–974. Hoffman PM, Powers JM, Weise MJ, Brostoff SW. Experimental allergic neuritis. I. Rat strain differences in the response to bovine myelin antigens. Brain Res 1980;195:355– 362. Hu W, Ramacher M, Hartung H-P, Kieseier BC. Schwann cells express Toll-like receptors. J Neuroimmunol 2004;154: 48. Iwasaki A, Medzhitov R. Toll-like receptor control of the adaptive immune responses. Nat Immunol 2004;5:987–995. Iwase T, Jung CG, Bae H, Zhang M, Soliven B. Glial cell line-derived neurotrophic factor-induced signaling in Schwann cells. J Neurochem 2005;94:1488 –1499. Janeway CA Jr, Medzhitov R. Innate immune recognition. Annu Rev Immunol 2002;20:197–216. Jung S, Gaupp S, Korn T, Kollner G, Hartung HP, Toyka KV. Biphasic form of experimental autoimmune neuritis in dark Agouti rats and its oral therapy by antigen-specific tolerization. J Neurosci Res 2004;75:524 –535. Kadlubowski M, Hughes RA. Identification of the neuritogen for experimental allergic neuritis. Nature 1979;277:140 – 141. Kadlubowski M, Hughes RA. The neuritogenicity and encephalitogenicity of P2 in the rat, guinea-pig and rabbit. J Neurol Sci 1980;48:171–178. Kerschensteiner M, Gallmeier E, Behrens L, Leal VV, Misgeld T, Klinkert WE, et al. Activated human T cells, B cells, and monocytes produce brain-derived neurotrophic factor in vitro and in inflammatory brain lesions: a neuroprotective role of inflammation? J Exp Med 1999;189:865– 870. Kiefer R, Funa K, Schweitzer T, Jung S, Bourde O, Toyka KV, et al. Transforming growth factor-beta 1 in experimental autoimmune neuritis. Cellular localization and time course. Am J Pathol 1996;148:211–223. Kiefer R, Kieseier BC, Bruck W, Hartung HP, Toyka KV. Macrophage differentiation antigens in acute and chronic autoimmune polyneuropathies. Brain 1998;121:469 – 479. Kiefer R, Kieseier BC, Stoll G, Hartung HP. The role of macrophages in immune-mediated damage to the peripheral nervous system. Prog Neurobiol 2001;64:109 –127. Immunocompetence of Schwann Cells 53. Kieseier BC, Kiefer R, Gold R, Hemmer B, Willison HJ, Hartung HP. Advances in understanding and treatment of immune-mediated disorders of the peripheral nervous system. Muscle Nerve 2004;30:131–156. 54. Kieseier BC, Hartung HP, Wiendl H. Immune circuitry in the peripheral nervous system. Curr Opin Neurol 2006;19: 437– 445. 55. Kingston AE, Bergsteinsdottir K, Jessen KR, Van der Meide PH, Colston MJ, Mirsky R. Schwann cells co-cultured with stimulated T cells and antigen express major histocompatibility complex (MHC) class II determinants without interferon-gamma pretreatment: synergistic effects of interferongamma and tumor necrosis factor on MHC class II induction. Eur J Immunol 1989;19:177–183. 56. Kobsar I, Berghoff M, Samsam M, Wessig C, Maurer M, Toyka KV, et al. Preserved myelin integrity and reduced axonopathy in connexin32-deficient mice lacking the recombination activating gene-1. Brain 2003;126:804 – 813. 57. Kobsar I, Hasenpusch-Theil K, Wessig C, Muller HW, Martini R. Evidence for macrophage-mediated myelin disruption in an animal model for Charcot–Marie–Tooth neuropathy type 1A. J Neurosci Res 2005;81:857– 864. 58. Kobsar I, Oetke C, Kroner A, Wessig C, Crocker P, Martini R. Attenuated demyelination in the absence of the macrophage-restricted adhesion molecule sialoadhesin (Siglec-1) in mice heterozygously deficient in P0. Mol Cell Neurosci 2006; 31:685– 691. 59. Koehler NK, Martin R, Wietholter H. The antibody repertoire in experimental allergic neuritis: evidence for PMP-22 as a novel neuritogen. J Neuroimmunol 1996;71:179 –189. 60. Koller H, Kieseier BC, Jander S, Hartung HP. Chronic inflammatory demyelinating polyneuropathy. N Engl J Med 2005;352:1343–1356. 61. Koller H, Schroeter M, Kieseier BC, Hartung HP. Chronic inflammatory demyelinating polyneuropathy— update on pathogenesis, diagnostic criteria and therapy. Curr Opin Neurol 2005;18:273–278. 62. Krammer PH. CD95⬘s deadly mission in the immune system. Nature 2000;407:789 –795. 63. Kusunoki S, Shimizu J, Chiba A, Ugawa Y, Hitoshi S, Kanazawa I. Experimental sensory neuropathy induced by sensitization with ganglioside GD1b. Ann Neurol 1996;39:424 – 431. 64. Lassmann H, Fierz W, Neuchrist C, Meyermann R. Chronic relapsing experimental allergic neuritis induced by repeated transfer of P2-protein reactive T cell lines. Brain 1991;114: 429 – 442. 65. Lee H, Jo EK, Choi SY, Oh SB, Park K, Kim JS, et al. Necrotic neuronal cells induce inflammatory Schwann cell activation via TLR2 and TLR3: implication in Wallerian degeneration. Biochem Biophys Res Commun 2006;350:742–747. 66. Lilje O, Armati PJ. The distribution and abundance of MHC and ICAM-1 on Schwann cells in vitro. J Neuroimmunol 1997;77:75– 84. 67. Lilje O. The processing and presentation of endogenous and exogenous antigen by Schwann cells in vitro. Cell Mol Life Sci 2002;59:2191–2198. 68. Linington C, Lassmann H, Ozawa K, Kosin S, Mongan L. Cell adhesion molecules of the immunoglobulin supergene family as tissue-specific autoantigens: induction of experimental allergic neuritis (EAN) by P0 protein-specific T cell lines. Eur J Immunol 1992;22:1813–1817. 69. Linker R, Lee DH, Siglienti I, Gold R. Is there a role for neurotrophins in the pathology of multiple sclerosis? J Neurol 2007;254(suppl I):I33–I40. 70. Lisak RP, Skundric D, Bealmear B, Ragheb S. The role of cytokines in Schwann cell damage, protection, and repair. J Infect Dis 1997;176(suppl 2):S173–179. 71. Maenaka K, Jones EY. MHC superfamily structure and the immune system. Curr Opin Struct Biol 1999;9:745–753. 72. Martini R, Zielasek J, Toyka KV, Giese KP, Schachner M. Protein zero (P0)-deficient mice show myelin degeneration MUSCLE & NERVE January 2008 11 73. 74. 75. 76. 77. 78. 79. 80. 81. 82. 83. 84. 85. 86. 87. 88. 89. 90. 12 in peripheral nerves characteristic of inherited human neuropathies. Nat Genet 1995;11:281–286. Martini R, Toyka KV. Immune-mediated components of hereditary demyelinating neuropathies: lessons from animal models and patients. Lancet Neurol 2004;3:457– 465. Mathey EK, Pollard JD, Armati PJ. TNF alpha, IFN gamma and IL-2 mRNA expression in CIDP sural nerve biopsies. J Neurol Sci 1999;163:47–52. Maurer M, Schmid CD, Bootz F, Zielasek J, Toyka KV, Oehen S, et al. Bone marrow transfer from wild-type mice reverts the beneficial effect of genetically mediated immune deficiency in myelin mutants. Mol Cell Neurosci 2001;17:1094 – 1101. Maurer M, Gold R. Animal models of immune-mediated neuropathies. Curr Opin Neurol 2002;15:617– 622. Maurer M, Muller M, Kobsar I, Leonhard C, Martini R, Kiefer R. Origin of pathogenic macrophages and endoneurial fibroblast-like cells in an animal model of inherited neuropathy. Mol Cell Neurosci 2003;23:351–359. McCombe PA, Harness J, Pender MP. Effects of cyclosporin A treatment on clinical course and inflammatory cell apoptosis in experimental autoimmune encephalomyelitis induced in Lewis rats by inoculation with myelin basic protein. J Neuroimmunol 1999;97:60 – 69. Medzhitov R, Janeway CJ. Innate immunity. N Engl J Med 2000;343:338 –344. Meyer zu Horste G, Nave KA. Animal models of inherited neuropathies. Curr Opin Neurol 2006;19:464 – 473. Miletic H, Utermohlen O, Wedekind C, Hermann M, Stenzel W, Lassmann H, et al. P0(106-125) is a neuritogenic epitope of the peripheral myelin protein P0 and induces autoimmune neuritis in C57BL/6 mice. J Neuropathol Exp Neurol 2005;64:66 –73. Milner P, Lovelidge CA, Taylor WA, Hughes RA. P0 myelin protein produces experimental allergic neuritis in Lewis rats. J Neurol Sci 1987;79:275–285. Miyamoto K, Oka N, Kawasaki T, Satoi H, Matsuo A, Akiguchi I. The action mechanism of cyclooxygenase-2 inhibitor for treatment of experimental allergic neuritis. Muscle Nerve 1999;22:1704 –1709. Miyamoto K, Miyake S, Schachner M, Yamamura T. Heterozygous null mutation of myelin P0 protein enhances susceptibility to autoimmune neuritis targeting P0 peptide. Eur J Immunol 2003;33:656 – 665. Muller M, Stenner M, Wacker K, Ringelstein EB, Hickey WF, Kiefer R. Contribution of resident endoneurial macrophages to the local cellular response in experimental autoimmune neuritis. J Neuropathol Exp Neurol 2006;65:499 – 507. Murata K, Dalakas MC. Expression of the co-stimulatory molecule BB-1, the ligands CTLA-4 and CD28 and their mRNAs in chronic inflammatory demyelinating polyneuropathy. Brain 2000;123:1660 –1666. Murwani R, Hodgkinson S, Armati P. Tumor necrosis factor alpha and interleukin-6 mRNA expression in neonatal Lewis rat Schwann cells and a neonatal rat Schwann cell line following interferon gamma stimulation. J Neuroimmunol 1996;71:65–71. Nobbio L, Gherardi G, Vigo T, Passalacqua M, Melloni E, Abbruzzese M, et al. Axonal damage and demyelination in long-term dorsal root ganglia cultures from a rat model of Charcot–Marie–Tooth type 1A disease. Eur J Neurosci 2006; 23:1445–1452. Oliveira RB, Ochoa MT, Sieling PA, Rea TH, Rambukkana A, Sarno EN, et al. Expression of Toll-like receptor 2 on human Schwann cells: a mechanism of nerve damage in leprosy. Infect Immun 2003;71:1427–1433. Oliveira RB, Sampaio EP, Aarestrup F, Teles RM, Silva TP, Oliveira AL, et al. Cytokines and Mycobacterium leprae induce apoptosis in human Schwann cells. J Neuropathol Exp Neurol 2005;64:882– 890. Immunocompetence of Schwann Cells 91. Pereira RM, Calegari-Silva TC, Hernandez MO, Saliba AM, Redner P, Pessolani MC, et al. Mycobacterium leprae induces NF-kappaB-dependent transcription repression in human Schwann cells. Biochem Biophys Res Commun 2005; 335:20 –26. 92. Pollard JD, McCombe PA, Baverstock J, Gatenby PA, McLeod JG. Class II antigen expression and T lymphocyte subsets in chronic inflammatory demyelinating polyneuropathy. J Neuroimmunol 1986;13:123–134. 93. Pollard JD, Baverstock J, McLeod JG. Class II antigen expression and inflammatory cells in the Guillain-Barré syndrome. Ann Neurol 1987;21:337–341. 94. Pollard JD. Chronic inflammatory demyelinating polyradiculoneuropathy. Curr Opin Neurol 2002;15:279 –283. 95. Rambukkana A, Yamada H, Zanazzi G, Mathus T, Salzer JL, Yurchenco PD, et al. Role of alpha-dystroglycan as a Schwann cell receptor for Mycobacterium leprae. Science 1998; 282:2076 –2079. 96. Rambukkana A, Zanazzi G, Tapinos N, Salzer JL. Contactdependent demyelination by Mycobacterium leprae in the absence of immune cells. Science 2002;296:927–931. 97. Rock KL, York IA, Goldberg AL. Post-proteasomal antigen processing for major histocompatibility complex class I presentation. Nat Immunol 2004;5:670 – 677. 98. Rostami A, Burns JB, Brown MJ, Rosen J, Zweiman B, Lisak RP, et al. Transfer of experimental allergic neuritis with P2-reactive T-cell lines. Cell Immunol 1985;91:354 –361. 99. Sabin TD, Swift TR, Jacobson RR. Leprosy. In: Dyck PJ, Thomas PK, editors. Peripheral neuropathy, 4th ed. (vol. 2). Philadelphia: Elsevier Saunders; 2005. p 2081–2108. 100. Salomon B, Rhee L, Bour-Jordan H, Hsin H, Montag A, Soliven B, et al. Development of spontaneous autoimmune peripheral polyneuropathy in B7–2-deficient NOD mice. J Exp Med 2001;194:677– 684. 101. Samuel NM, Mirsky R, Grange JM, Jessen KR. Expression of major histocompatibility complex class I and class II antigens in human Schwann cell cultures and effects of infection with Mycobacterium leprae. Clin Exp Immunol 1987;68:500 –509. 102. Sawant-Mane S, Piddlesden SJ, Morgan BP, Holers VM, Koski CL. CD59 homologue regulates complement-dependent cytolysis of rat Schwann cells. J Neuroimmunol 1996;69:63– 71. 103. Scherer SS, Kamholz J, Jakowlew SB. Axons modulate the expression of transforming growth factor-betas in Schwann cells. Glia 1993;8:265–276. 104. Schmid CD, Stienekemeier M, Oehen S, Bootz F, Zielasek J, Gold R, et al. Immune deficiency in mouse models for inherited peripheral neuropathies leads to improved myelin maintenance. J Neurosci 2000;20:729 –735. 105. Sharpe AH, Freeman GJ. The B7-CD28 superfamily. Nat Rev Immunol 2002;2:116 –126. 106. Sheikh KA, Ho TW, Nachamkin I, Li CY, Cornblath DR, Asbury AK, et al. Molecular mimicry in Guillain–Barré syndrome. Ann NY Acad Sci 1998;845:307–321. 107. Skundric DS, Bealmear B, Lisak RP. Induced upregulation of IL-1, IL-1RA and IL-1R type I gene expression by Schwann cells. J Neuroimmunol 1997;74:9 –18. 108. Skundric DS, Lisak RP, Rouhi M, Kieseier BC, Jung S, Hartung HP. Schwann cell–specific regulation of IL-1 and IL1Ra during EAN: possible relevance for immune regulation at paranodal regions. J Neuroimmunol 2001;116:74 – 82. 109. Snyder DH, Stone SH, Raine CS. Attempts to induce chronic experimental allergic neuritis in strain 13 and Hartley guinea pigs. J Neuropathol Exp Neurol 1977;36:488 – 498. 110. Spierings E, de Boer T, Wieles B, Adams LB, Marani E, Ottenhoff TH. Mycobacterium leprae-specific, HLA class IIrestricted killing of human Schwann cells by CD4⫹ Th1 cells: a novel immunopathogenic mechanism of nerve damage in leprosy. J Immunol 2001;166:5883–5888. 111. Stadelmann C, Kerschensteiner M, Misgeld T, Bruck W, Hohlfeld R, Lassmann H. BDNF and gp145trkB in multiple MUSCLE & NERVE January 2008 112. 113. 114. 115. 116. 117. 118. 119. 120. 121. 122. 123. sclerosis brain lesions: neuroprotective interactions between immune and neuronal cells? Brain 2002;125:75– 85. Steinhoff U, Kaufmann SH. Specific lysis by CD8⫹ T cells of Schwann cells expressing Mycobacterium leprae antigens. Eur J Immunol 1988;18:969 –972. Stewart HJ, Rougon G, Dong Z, Dean C, Jessen KR, Mirsky R. TGF-betas upregulate NCAM and L1 expression in cultured Schwann cells, suppress cyclic AMP-induced expression of O4 and galactocerebroside, and are widely expressed in cells of the Schwann cell lineage in vivo. Glia 1995;15:419 – 436. Stoll G, Schwendemann G, Heininger K, Kohne W, Hartung HP, Seitz R, Toyka KV. Relation of clinical, serological, morphological, and electrophysiological findings in galactocerebroside-induced experimental allergic neuritis. J Neurol Neurosurg Psychiatry 1986;49:258 –264. Stoll G, Schmidt B, Jander S, Toyka KV, Hartung HP. Presence of the terminal complement complex (C5b-9) precedes myelin degradation in immune-mediated demyelination of the rat peripheral nervous system. Ann Neurol 1991;30:147– 155. Stoll G, Jung S, Jander S, van der Meide P, Hartung HP. Tumor necrosis factor-alpha in immune-mediated demyelination and Wallerian degeneration of the rat peripheral nervous system. J Neuroimmunol 1993;45:175–182. Suter U, Scherer SS. Disease mechanisms in inherited neuropathies. Nat Rev Neurosci 2003;4:714 –726. Takeda K, Kaisho T, Akira S. Toll-like receptors. Annu Rev Immunol 2003;21:335–376. Takigawa T, Yasuda H, Terada M, Haneda M, Kashiwagi A, Saito T, et al. The sera from GM1 ganglioside antibody positive patients with Guillain–Barré syndrome or chronic inflammatory demyelinating polyneuropathy blocks Na⫹ currents in rat single myelinated nerve fibers. Intern Med 2000;39:123–127. Tapinos N, Ohnishi M, Rambukkana A. ErbB2 receptor tyrosine kinase signaling mediates early demyelination induced by leprosy bacilli. Nat Med 2006;12:961–966. Taylor WA, Hughes RA. Experimental allergic neuritis induced in SJL mice by bovine P2. J Neuroimmunol 1985;8: 153–157. Trombetta ES, Mellman I. Cell biology of antigen processing in vitro and in vivo. Annu Rev Immunol 2005;23:975–1028. Tsai CP, Pollard JD, Armati PJ. Interferon-gamma inhibition suppresses experimental allergic neuritis: modulation of major histocompatibility complex expression of Schwann cells in vitro. J Neuroimmunol 1991;31:133–145. Immunocompetence of Schwann Cells 124. Visan L, Visan IA, Weishaupt A, Hofstetter HH, Toyka KV, Hunig T, et al. Tolerance induction by intrathymic expression of P0. J Immunol 2004;172:1364 –1370. 125. Vital A, Vital C, Julien J, Fontan D. Occurrence of active demyelinating lesions in children with hereditary motor and sensory neuropathy (HMSN) type I. Acta Neuropathol (Berl) 1992;84:433– 436. 126. Wagner R, Myers RR. Schwann cells produce tumor necrosis factor alpha: expression in injured and non-injured nerves. Neuroscience 1996;73:625– 629. 127. Watts C. The exogenous pathway for antigen presentation on major histocompatibility complex class II and CD1 molecules. Nat Immunol 2004;5:685– 692. 128. Weber F, Rudel R, Aulkemeyer P, Brinkmeier H. Anti-GM1 antibodies can block neuronal voltage-gated sodium channels. Muscle Nerve 2000;23:1414 –1420. 129. Weerth S, Berger T, Lassmann H, Linington C. Encephalitogenic and neuritogenic T cell responses to the myelinassociated glycoprotein (MAG) in the Lewis rat. J Neuroimmunol 1999;95:157–164. 130. Wekerle H, Schwab M, Linington C, Meyermann R. Antigen presentation in the peripheral nervous system: Schwann cells present endogenous myelin autoantigens to lymphocytes. Eur J Immunol 1986;16:1551–1557. 131. Wohlleben G, Hartung HP, Gold R. Humoral and cellular immune functions of cytokine-treated Schwann cells. Adv Exp Med Biol 1999;468:151–156. 132. Wohlleben G, Ibrahim SM, Schmidt J, Toyka KV, Hartung HP, Gold R. Regulation of Fas and FasL expression on rat Schwann cells. Glia 2000;30:373–381. 133. Yuki N, Taki T, Inagaki F, Kasama T, Takahashi M, Saito K, et al. A bacterium lipopolysaccharide that elicits Guillain– Barré syndrome has a GM1 ganglioside-like structure. J Exp Med 1993;178:1771–1775. 134. Yuki N, Taki T, Takahashi M, Saito K, Yoshino H, Tai T, et al. Molecular mimicry between GQ1b ganglioside and lipopolysaccharides of Campylobacter jejuni isolated from patients with Fisher’s syndrome. Ann Neurol 1994;36:791–793. 135. Yuki N, Yamada M, Koga M, Odaka M, Susuki K, Tagawa Y, et al. Animal model of axonal Guillain–Barré syndrome induced by sensitization with GM1 ganglioside. Ann Neurol 2001;49:712–720. 136. Yuki N. Carbohydrate mimicry: a new paradigm of autoimmune diseases. Curr Opin Immunol 2005;17:577–582. 137. Zou LP, Ljunggren HG, Levi M, Nennesmo I, Wahren B, Mix E, et al. P0 protein peptide 180 –199 together with pertussis toxin induces experimental autoimmune neuritis in resistant C57BL/6 mice. J Neurosci Res 2000;62:717–721. MUSCLE & NERVE January 2008 13