* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download ORGANIC CHEMISTRY
Physical organic chemistry wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Hydroformylation wikipedia , lookup
Homoaromaticity wikipedia , lookup
Organosulfur compounds wikipedia , lookup
Aromatization wikipedia , lookup
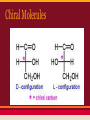
ORGANIC CHEMISTRY Classes of Organic Compounds Classification of Hydrocarbons 1. Aromatic Hydrocarbons-contain one or more benzene rings 2. Aliphatic Hydrocarbons - all other hydrocarbons Aliphatic Hydrocarbons Carbon atoms can be bonded in straight chains, branched chains, or rings. Chains may contain single, double, or triple bonds. A saturated compound contains only single bond between carbons. These are alkanes. ALKANES Simplest hydrocarbon Each differs from the next by a -CH2group. Compounds whose structures differ from each other by a specific structural unit is called a homologous series. ALKANES Simplest alkane is methane. CH4 General formula of alkane is CnH2n+2 All end in -ane Soluble in nonpolar solvents, C to C bonds have sp3 hybridization. Hydrocarbon Prefixes ALKANES When a hydrogen along with its associated electrons is removed from a hydrocarbon molecule, a radical is left. CH3 is a radical. It is named methyl. Radicals end in -yl. Alkanes form alkyl groups. Branched Chain Alkanes 1. Find longest continuous chain or parent chain. 2. Add the name of the branches (substituents) attached in alphabetical order using prefixes. 3. Number Carbon atoms in parent 4. Insert position # of each substituent group 5. Punctuate name - separate position # with hyphens, more than one # is separated by commas. Cycloalkanes Alkane rings General formula is CnH2n Example cyclopentane C5H10 Isomers Isomerism is the existence of two or more substances with the same molecular formula, but different molecular structures. Important because structure determines properties and function. There are two main types of isomers. Structural Isomers Structural Isomers Also called constitutional isomers. Atoms are bonded in different arrangements--thus different properties. Longer chains have higher boiling points (BP) and melting points (MP) Having branches on the chain serves to lower the BP but it raises the melting point. Stereoisomers All atoms are bonded in the same order but are arranged differently in space. There are two types: a. Geometric isomers b. Optical isomers Geometric Isomers Structural isomers have a different arrangement of groups around a double bond. In alkenes with double bonds, the carbon is locked in place and cannot rotate freely as it could in alkanes with single bonds. Pi bonds Geometric isomers Optical Isomers Optical Isomerism (Chiralty) occurs when there is an asymmetric carbon (C has 4 different atoms or groups attached) Chiral Molecules Chirality is important in drugs and enzymecatalyyzed rxns. Only L-form of ascorbic acid functions as vitamin C. Geometric Isomers Properties and Uses of Alkanes 1. C-H bonds are nonpolar 2. Strength of London dispersion forces increases as mass increases--# of C determines state of matter. 3. Longer the chain, higher the boiling point. Properties and uses of alkanes 4. Alkanes less reactive than other hydrocarbons due to stability of single covalent bonds. 5. Most important rxn is combustion. Fractional Distillation Gasoline is a component of fossil fuel. Is can be extracted by fractional distillation. Combustion Straight-chain alkanes ignite more spontaneously than branched chains but cause knocking in autos. Octane rating is a measure of burning efficiency and antiknock properties. 2,2,4-trimethyl pentane (isooctane) resists knocking C8H18 2,2,4-trimethylpentane octane Unsaturated Hydrocarbons Contain double and triple carbon to carbon bonds. Include Alkenes and Alkynes ALKENES Olefin is another name. Contain double bonds. General formula CnH2n Double bond is shorter than a single bond and it is also stronger and more reactive. Double C bonds have sp2 hybridization. Naming Alkenes End in -ene. The double bond location takes precedence in naming. If there is more than one double bond, the location is numbered and a prefix is used 2 = adiene 3 = atriene Alkenes are numbered so that the lowest position number is assigned to the first carbon atom to which the double bond is attached. 1,4-pentadiene Ethene #1 organic chemical in industry Product of petroleum refining Makes plastics and synthetics ALKYNES Contain triple bonds. General formula is CnH2n-2 End in -yne Ethyne or acetylene Naming Alkynes The numbering system follows the same pattern used for naming alkenes. 4,4-dimethyl-2-pentyne If a compound has both double and triple bonds, the double bonds take precedencein numbering. The double bond is given the lowest number and named first. Benzene Ring Kekule’s snake AROMATIC HYDROCARBONS Contain Benzene ring Most have distinctive odors. Thousands of compounds are derived from benzene. Ways to diagram benzene Important in making plastics and fibers Naphthalene 2 benzene rings Phenyl Radical Methyl Benzene or Toluene improves quality of gasoline used in production of explosives TNT Functional Groups A substituent group that is not made up of only carbon and hydrogen is a functional group. -OH is hydroxide