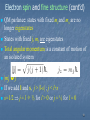* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Optically polarized atoms_ch_2_old
Elementary particle wikipedia , lookup
Dirac equation wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Particle in a box wikipedia , lookup
Renormalization wikipedia , lookup
Renormalization group wikipedia , lookup
Bell's theorem wikipedia , lookup
Canonical quantization wikipedia , lookup
Quantum state wikipedia , lookup
Nitrogen-vacancy center wikipedia , lookup
EPR paradox wikipedia , lookup
Tight binding wikipedia , lookup
Ferromagnetism wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Electron paramagnetic resonance wikipedia , lookup
Atomic orbital wikipedia , lookup
Electron scattering wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Spin (physics) wikipedia , lookup
Atomic theory wikipedia , lookup
Electron configuration wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Optically polarized atoms Marcis Auzinsh, University of Latvia Dmitry Budker, UC Berkeley and LBNL Simon M. Rochester, UC Berkeley 1 Chapter 2: Atomic states A brief summary of atomic structure Begin with hydrogen atom The Schrödinger Eqn: Image from Wikipedia In this approximation (ignoring spin and relativity): Principal quant. Number n=1,2,3,… 2 Could have guessed me 4/2 from dimensions me 4/2 = 1 Hartree me 4/22 = 1 Rydberg E does not depend on l or m degeneracy i.e. different wavefunction have same E We will see that the degeneracy is n2 3 Angular momentum of the electron in the hydrogen atom Orbital-angular-momentum quantum number l = 0,1,2,… This can be obtained, e.g., from the Schrödinger Eqn., or straight from QM commutation relations The Bohr model: classical orbits quantized by requiring angular momentum to be integer multiple of There is kinetic energy associated with orbital motion an upper bound on l for a given value of En Turns out: l = 0,1,2, …, n-1 4 Angular momentum of the electron in the hydrogen atom (cont’d) In classical physics, to fully specify orbital angular momentum, one needs two more parameters (e.g., to angles) in addition to the magnitude In QM, if we know projection on one axis (quantization axis), projections on other two axes are uncertain Choosing z as quantization axis: Note: this is reasonable as we expect projection magnitude not to exceed 5 Angular momentum of the electron in the hydrogen atom (cont’d) m – magnetic quantum number because B-field can be used to define quantization axis Can also define the axis with E (static or oscillating), other fields (e.g., gravitational), or nothing Let’s count states: m = -l,…,l i. e. 2l+1 states l = 0,…,n-1 n 1 1 2( n 1) 1 2 (2 l 1) n n 2 l 0 As advertised ! 6 Angular momentum of the electron in the hydrogen atom (cont’d) Degeneracy w.r.t. m expected from isotropy of space Degeneracy w.r.t. l, in contrast, is a special feature of 1/r (Coulomb) potential How can one understand why only one projection of the angular momentum at a time can be determined? In analogy with write an uncertainty relation between lz and φ (angle in the x-y plane of the projection of the angular momentum w.r.t. x axis): 7 Angular momentum of the electron in the hydrogen atom (cont’d) How can one understand why only one projection of the angular momentum at a time can be determined? In analogy with (*) write an uncertainty relation between lz and φ (angle in the x-y plane of the projection of the angular momentum w.r.t. x axis): This is a bit more complex than (*) because φ is cyclic With definite lz , φ is completely uncertain… 8 Wavefunctions of the H atom A specific wavefunction is labeled with n l m : In polar coordinates : i.e. separation of radial and angular parts Spherical functions (Harmonics) Further separation: 9 Wavefunctions of the H atom (cont’d) Legendre Polynomials Separation into radial and angular part is possible for any central potential ! Things get nontrivial for multielectron atoms 10 Electron spin and fine structure Experiment: electron has intrinsic angular momentum -spin (quantum number s) It is tempting to think of the spin classically as a spinning object. This might be useful, but to a point. L I mr 2 Presumably, we want finite The surface of the object has linear velocity r c If we have L , (1,2) r mc = c (1) (2) 3.9 1011 cm Experiment: electron is pointlike down to ~ 10-18 cm 11 Electron spin and fine structure (cont’d) Another issue for classical picture: it takes a 4π rotation to bring a half-integer spin to its original state. Amazingly, this does happen in classical world: from Feynman's 1986 Dirac Memorial Lecture (Elementary Particles and the Laws of Physics, CUP 1987) 12 Electron spin and fine structure (cont’d) Another amusing classical picture: spin angular momentum comes from the electromagnetic field of the electron: This leads to electron size Experiment: electron is pointlike down to ~ 10-18 cm 13 Electron spin and fine structure (cont’d) s=1/2 “Spin up” and “down” should be used with understanding that the length (modulus) of the spin vector is >/2 ! 14 Electron spin and fine structure (cont’d) Both orbital angular momentum and spin have associated magnetic moments μl and μs Classical estimate of μl : current loop For orbit of radius r, speed p/m, revolution rate is Gyromagnetic ratio 15 Electron spin and fine structure (cont’d) Bohr magneton In analogy, there is also spin magnetic moment : 16 Electron spin and fine structure (cont’d) The factor 2 is important ! Dirac equation for spin-1/2 predicts exactly 2 QED predicts deviations from 2 due to vacuum fluctuations of the E/M field One of the most precisely measured physical constants: 2=21.00115965218085(76)(0.8 parts per trillion) New Measurement of the Electron Magnetic Moment Using a One-Electron Quantum Cyclotron, B. Odom, D. Hanneke, B. D'Urso, and G. Gabrielse, Phys. Rev. Lett. 97, 030801 (2006) Prof. G. Gabrielse, Harvard 17 Electron spin and fine structure (cont’d) When both l and s are present, these are not conserved separately This is like planetary spin and orbital motion On a short time scale, conservation of individual angular momenta can be a good approximation l and s are coupled via spin-orbit interaction: interaction of the motional magnetic field in the electron’s frame with μs Energy shift depends on relative orientation of l and s, i.e., on 18 Electron spin and fine structure (cont’d) QM parlance: states with fixed ml and ms are no longer eigenstates States with fixed j, mj are eigenstates Total angular momentum is a constant of motion of an isolated system |mj| j If we add l and s, j > |l-s| ; j < l+s s=1/2 j = l ½ for l > 0 or j = ½ for l = 0 19 Electron spin and fine structure (cont’d) Spin-orbit interaction is a relativistic effect Including rel. effects : Correction to the Bohr formula 2 The energy now depends on n and j 20 Electron spin and fine structure (cont’d) 1/137 relativistic corrections are small ~ 10-5 Ry E 0.366 cm-1 or 10.9 GHz for 2P3/2 , 2P1/2 E 0.108 cm-1 or 3.24 GHz for 3P3/2 , 3P1/2 21 Electron spin and fine structure (cont’d) The Dirac formula : predicts that states of same n and j, but different l remain degenerate In reality, this degeneracy is also lifted by QED effects (Lamb shift) For 2S1/2 , 2P1/2: E 0.035 cm-1 or 1057 MHz 22 Vector model of the atom Some people really need pictures… jx j y 0; Recall: Expectation value of j2 is j( j 1) We can draw all of this as (j=3/2) mj = 3/2 mj = 1/2 23 Vector model of the atom (cont’d) These pictures are nice, but NOT problem-free Consider maximum-projection state mj = j mj = 3/2 Q: What is the maximal value of jx or jy that can be measured ? A: that might be inferred from the picture is wrong… 24 Vector model of the atom (cont’d) So how do we draw angular momenta and coupling ? Maybe as a vector of expectation values, e.g., Simple Has well defined QM meaning ? BUT Boring Non-illuminating Or stick with the cones ? 25