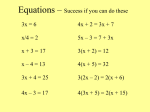* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download What are the general types of reactions?
Enantioselective synthesis wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Catalytic reforming wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Radical (chemistry) wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Artificial photosynthesis wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Organic chemistry wikipedia , lookup
History of chemistry wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Marcus theory wikipedia , lookup
Gaseous signaling molecules wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Asymmetric induction wikipedia , lookup
Process chemistry wikipedia , lookup
Isotopic labeling wikipedia , lookup
George S. Hammond wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Water splitting wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Electrolysis of water wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Electrochemistry wikipedia , lookup
Rate equation wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Atomic theory wikipedia , lookup
Transition state theory wikipedia , lookup
Click chemistry wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Chemical reaction wikipedia , lookup
S-133 • Write the formula for – Palladium (IV) Oxide – Calcium Fluoride – Cadmium (II) Nitride Write the name for - Al2S3 - Rb3P - CrO3 Unit 7 Chemical Reaction SPS2. Students will explore the nature of matter, its classifications, and its system for naming types of matter. d. Demonstrate the Law of Conservation of Matter in a chemical reaction. e. Apply the Law of Conservation of Matter by balancing the following types of chemical equations; - Synthesis Decomposition Single Replacement Double Replacement 7.1 Describing Reactions What is the law of conservation of mass? Why must chemical equations be balanced? Why do chemists use the mole? How can you calculate the mass of a reactant or product in a chemical reaction? 7.1 Describing Reactions What is the law of conservation of mass? • Chemical Equations – Reactants – the substances that are present before a reaction – Products – the substances present after a reaction is complete – Always given in the form Reactants Products – Example:Carbon + Oxygen Carbon Dioxide – Or C + O2 CO2 7.1 Describing Reactions What is the law of conservation of mass? • Writing Equations - Practice – Copper and Oxygen make Copper (II) Oxide – Magnesium and Hydrogen Chloride make Hydrogen and Magnesium Chloride – Ethylene (C2H4) burns with Oxygen to produce Carbon Dioxide and Water. – Hydrogen and Chlorine combine to make Hydrogen Chloride 7.1 Describing Reactions What is the law of conservation of mass? • Conservation of Mass – Mass is not created or destroyed in a chemical reaction – For practical purposes • Same types of atoms before and after a reaction • Same number of each type of atom before and after – Equations must show this – They are called balanced equations S-134 • Write the formula for – Dinitrogen Trioxide – Copper (II) oxide – Dinitrogen Pentaoxide Write the name for - CCl4 - CrBr3 - Mo2O5 S-135 • Write the equation for a reaction of hydrogen sulfide with aluminum oxide to make aluminum sulfide and water. 7.1 Describing Reactions Why must chemical equations be balanced? • Balancing Equations – If an equation does not have the same elements on both sides, it is a false equation • Can not actually occur – Coefficient – a number placed in front of a substance in a chemical equation • Used to balance equations 7.1 Describing Reactions Why must chemical equations be balanced? • Balancing Equations - Steps – First write out the equation • Hydrogen and Oxygen make water becomes • H2 + O2 H2O – List the elements on each side H-2 O-2 H-2 O-1 – Add substances until both sides are equal 7.1 Describing Reactions Why must chemical equations be balanced? • Balancing Equations - Practice – Copper and Oxygen make Copper (II) Oxide – Magnesium and Hydrogen Chloride make Hydrogen and Magnesium Chloride – Ethylene (C2H4) burns with Oxygen to produce Carbon Dioxide and Water. S-137 • Balance the following equations H2SO4 + Al(OH)3 Al2(SO4)3 + H2O 7.1 Describing Reactions Why do chemists use the mole? • Counting With Moles 18 Ar 39.95 – A unit of measurement Argon 23 – Equals 6.02 x 10 of anything – Used only to count atoms, molecules, formula unit – One mole of an element is equal to its atomic mass converted to grams (put a g beside the number) 7.1 Describing Reactions How can you calculate the mass of a reactant or product in a reaction? • Molar Mass – For an element equal to its atomic mass – For a compound • For each element – multiply the mass x the number of that element in the compound • Add the total 7.1 Describing Reactions How can you calculate the mass of a reactant or product in a reaction? • Molar Mass-Example – C 2 H4 – Carbon – Hydrogen – 12.01g x 2 = 24.02g 1.01g x 4 = 4.04g Total 28.06 7.1 Describing Reactions How can you calculate the mass of a reactant or product in a reaction? • Molar Mass-Example – Pb(OH)4 S-137 • What is the molar mass of Al2(SO4)3 7.2 Types of Reactions What are the general types of chemical reactions? Types of Reactions What are the general types of reactions? • Classifying Reactions – – Describe how reactants interact to form products Help to predict the products of reactions Types of Reactions What are the general types of reactions? • Synthesis – – – – Two or more substances react to form a single substance Pattern A + B AB Always has one product Examples • • 2Na + Cl2 2NaCl 2H2 + O2 2H2O Types of Reactions What are the general types of reactions? • Decomposition – – – – One substance breaks apart into two or more products Pattern AB A + B Always has one reactant Examples • • CaCO3 CaO + CO2 2H2O 2H2 + O2 Types of Reactions What are the general types of reactions? • Single Replacement (Displacement) – – – – A compound switches parts with an element Pattern A + BC B + AC Always has one element and one compound on each side Examples • • Cu + 2AgNO3 2Ag + Cu(NO3)2 2K + 2H2O H2 + KOH Types of Reactions What are the general types of reactions? • Double Replacement (Displacement) – – – – – Two compounds switch parts Often results in the formation of a precipitate Pattern AB + CD AD + CB Always has two compounds on each side Examples • • Pb(NO3)2 + 2KI PbI2 + 2KNO3 CaCO3 + 2HCl CaCl2 + H2CO3 Types of Reactions What are the general types of reactions? • Combustion – – – – – Reacts rapidly with oxygen Everything combines with oxygen Pattern AB + O2 AO + BO Always has oxygen as a reactant Examples • • CH4 + O2 CO2 + 2H2O 2Ca + O2 2CaO S-138 • Balance the following reactions and tell what type(s) they are. 1. Pb(NO3)2 + HCl PbCl2 + HNO3 2. Ca + HCl CaCl2 + H2 S-139 • Write out the reaction, then balance it and tell what type it is. 1. Mercury reacts with oxygen to form Mercury (II) Oxide S-140 • Write out the reaction, then balance it, tell what type it is and calculate the molar mass of the compound 1. Iron (III) Chloride is produced by the reaction of Iron (III) Oxide and Lead (II) Chloride. Lead (II) Oxide is also produced.