* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download The Mole - cloudfront.net
Survey
Document related concepts
Debye–Hückel equation wikipedia , lookup
Fluorescence correlation spectroscopy wikipedia , lookup
Particle-size distribution wikipedia , lookup
History of molecular theory wikipedia , lookup
Elementary particle wikipedia , lookup
Depletion force wikipedia , lookup
Dimensional analysis wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Stoichiometry wikipedia , lookup
Aerosol mass spectrometry wikipedia , lookup
Transcript
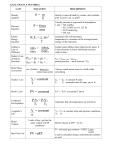
Chemical Quantities Chapter 10 Introduction • Counting and measuring quantities of atoms and molecules • Convert between units of volume, mass, and quantities of molecules, atoms, etc. • Calculate the percent composition of compounds and derive the empirical and molecular formulas and masses The Mole: A Measurement of Matter (Section 10.1) • Measuring matter • What is a mole? • The mass of a mole of an element • The mass of a mole of a compound I. Measuring Matter • Measure matter in three ways: 1. Mass 2. Volume 3. Counting • Recall the SI Units for mass and volume 1. Mass: kg 2. Volume: L • Remember how to use the prefixes Matter: Anything that has Mass and occupies space. (Ch. 1) Units that indicate a specific number of objects. • Dozen = 12 • Pair = 2 • Ream = 500 • Gross = 144 A relationship can be established between count units and mass or volume. • Let’s say 1 dozen apples = 2.0 kg and 1 dozen apples = 0.20 bushels • We can use these relationships to set up 3 conversion factors: 1 dozen 1dozen 1 dozen 12 apples 2.0 kg apples 0.20 bushels Let’s quickly review dimensional analysis. • Dimensional analysis is a tool for solving conversion problems (going from one unit to a different unit) • A conversion factor (a ratio relating the two units involved in the conversion) is used. • Simple multiplication of the ratios will yield the desired conversion. Three basic questions to ask when doing dimensional analysis: 1. What unit do I start out with? 2. What unit do I want to end up with? 3. What conversion factor relates those two units? Let’s try a problem. What is the mass of 90 average-sized apples if one dozen of the apples has a mass of 2.0 kg? II. What is a Mole? A mole (mol) of a substance is 6.02 x 1023 representative particles of that substance and is the SI unit for measuring the amount of a substance. • The mole makes counting atoms and molecules more feasible. • 6.02 x 1023 is known as Avogadro’s number. • Representative particle: the species present in a substance (i.e. atoms, molecules). What would a mole of stuff look like? 1 mole of atoms would cover the entire Earth to a depth of 50 miles. A mole of dollars would guarantee every person in the world (~ 6 billion people) an income of over $3 million dollars each second for 100 years. And how about a mole of moles? • The mass of a mole of moles would be 60 times greater than the mass of all the water in all the oceans. • A mole of moles lined up end-to-end would stretch from here to the nearest star more than 2 million times. What are these “representative particles?” • Most Elements • Some Elements • Molecular Compounds • Ionic Compounds Atoms (Fe, Na, P) Diatomic Molecules (O2, N2, Cl2) Molecules (CH4, H2O) Formula Unit (NaCl, CaCl2) We can use the mole to particle relationship as a conversion factor. • The equivalence is: 1 mole = 6.02 x 1023 representative particle • Which gives us the following conversion factors: 1 mole 6.02 x 1023 particles 6.02 x 1023 particles 1 mole • We can use these conversion factors to go from moles to particles and vice versa. # of Particles to Moles • What is the “unit” that we start out with? # of particles • What “unit do we want to end up with? mole • Which conversion factor would we use? 1 mole 6.02 x 1023 particles Let’s try a problem. Magnesium is a light metal used in the manufacture of aircraft, automobile wheels, tools, and garden furniture. How many moles of magnesium is 1.25 x 1023 atoms of magnesium? Moles to # of Particles • What is the “unit” that we start out with? mole • What “unit do we want to end up with? # of particles • Which conversion factor would we use? 6.02 x 1023 particles 1 mole Let’s try a problem. Propane gas is used for cooking and heating. How many atoms are in 2.12 mol of propane (C3H8)? III. The Mass of a Mole of an Element • Atomic masses are expressed in atomic mass units (amu). – Remember these are “weighted averages.” • By converting this relative scale to the more convenient mass scale in grams, we get values that are easier to measure out. • Thus, the atomic mass of an element expressed in grams is the mass of one mole of the element. Mass of 1 mole of marbles (if 1 marble = 1 g.) 6.02 x 1023 g. Molar Mass The mass of 1 mole of an element. • These masses are essentially the same as the average atomic masses given in the periodic table. • If you take one mole of any element and weigh it, the mass will be equal to the molar mass. IV. The Mass of a Mole of a Compound • In order to find the mass of a mole of a compound, we must know the formula of the compound. • Calculate the mass of a compound by adding the atomic masses of the atoms that make up the compound. • The molar mass of any compound is the mass in grams of 1 mole of that compound. Let’s try a problem. The decomposition of hydrogen peroxide (H2O2) provides sufficient energy to launch a rocket. What is the molar mass of hydrogen peroxide? Mole-Mass and Mole-Volume Relationships (Section 10.2) Representative Particles • The MoleMass Relationship • The MoleVolume Relationship Mole Mass Volume I. The Mole-Mass Relationship • The mole-mass relationship is essentially the relationship between # of particles to mass they have. • In section 10.1 we saw how the mole is related to mass by the molar mass of a substance (element or compound). • We can use the molar mass to convert between the moles of a substance and its mass (more dimensional analysis!). Let’s see how this is done. What is the mass of 3.00 mol of NaCl? 1. Calculate the molar mass of NaCl. 2. Use this as the conversion factor. 3. Solve for the answer using dimensional analysis. Let’s convert from mass to moles. How many moles is 10.0 g. of sodium sulfate? II. The Mole-Volume Relationship • The mole-volume relationship is essentially a relationship between # of particles and the volume they occupy. • For gases the mole-volume relationship is not as straight forward as the mole-mass relationship. • For gases, volume is dependent upon temperature and pressure. The mole-volume relationship in gases is straight forward if we define the specific conditions the gases are in. • This is because gases occupy different volumes at different temperatures and pressures. • To compare the mole-volume relationships of different gases we must compare at the same temperature and pressure (“STP”) Avogadro’s Hypothesis Equal volumes of gases at the same temperature and pressure contain the same number of particles. • Though gas molecules are not the same sizes, the spaces between gas molecules, for any gas is so large, it makes the sizes of the individual gas molecules insignificant. • All three gases occupy the same volume (22.4 L) and have the same number of particles (1 mol) because they are all at the same temperature and pressure. What is STP? • STP = “Standard Temperature and Pressure” – Temperature = 0O C. – Pressure = 101.3 kPa or 1 atm. • At STP the following relationship exists between moles and volume: I mole = 22.4 L (“Molar Volume”) or 6.02 x 1023 particles = 22.4 L Calculating the volume of a gas at STP. • The molar volume is used to convert a known number of moles of gas to the volume of the gas at STP. • The conversion factors are: 22.4 L 1 mol 1 mol 22.4 L • The choice of conversion factor depends on whether you start with mol or L. Let’s try a problem. If you have 0.375 mol of O2, what volume would it occupy at STP? Calculating the molar mass of a gas from its density. • Different gases have different densities (d= m/v). • For gases the density is measured in grams per liter (g/L) at a specific temperature. • We can use this density of a gas to calculate its molar mass. • molar mass = density (STP) x molar vol. Let’s analyze the units and see how we can determine molar mass from density and molar volume. 1. What are the units of density? 2. What are the units of molar volume? 3. What happens when we multiply them? 4. What do these units describe? Let’s try a problem. The density of a gaseous compound containing carbon and oxygen is found to be 1.964 g/L at STP. What is the molar mass of the compound? A Summary of the Mole • A unit equivalent to 6.02 x 1023 particles. • The “particles” are atoms, ions, molecules, and formula units. • It’s relationships to mass and volume allow us to convert between mass, volume, and number of particles. The Mole and Conversions 1. Four types of conversion problems: • mol particles (atoms,etc) – 1 mol = 6.02 x 1023 particles • mol mass – molar mass (g/mol) • mol volume – molar volume (1 mol = 22.4 L @ STP) • density – molar volume molar mass Percent Composition and Chemical Formulas (Section 10.3) • The Percent Composition of a Compound • Empirical Formulas 26.8% Cr 40.3% K 32.9% O • Molecular Formulas Potassium Chromate, K2CrO4 I. The Percent Composition of a Compound The relative amounts of the elements in a compound. • In other words, the % composition is the percent by mass of each element in a compound. • This consists of the percent value for each element of a compound. Let’s look at an example. • Each percentage represents the percent by mass of each element. • The total has to equal 100. 26.8% Cr 40.3% K 32.9% O Potassium Chromate, K2CrO4 Obtaining % Composition from mass data. • We can obtain the percent composition of a compound if we have information on the masses of the elements that make up the compound and the total mass of the compound. • We can obtain these masses experimentally. In other words, plug numbers into the following equation. % mass = mass of the element x 100 of an element mass of compound Let’s try a problem. When a 13.60 g. sample of a compound containing only magnesium and oxygen is decomposed, 5.40 g. of oxygen is obtained. What is the percent composition of this compound? Obtaining the percent composition from the chemical formula. • The chemical formula allows us to determine the percent composition of a compound. • This is because the % composition of a compound stays the same regardless of how much of it we have. • The subscripts in the formula is used to calculate the mass of each element in 1 mole of a compound. Let’s look at an example. Water = H2O 11.1% H and 88.9% O by mass regardless of the volume of water We can use the following equation to obtain the percent composition from the chemical formula: % mass = mass of element in 1 mole of compound molar mass of compound x 100 Obtaining % Composition from the chemical formula. 1. Know what the chemical formula is. 2. Determine the molar mass of each element. 3. Calculate the total mass of the compound. 4. Divide the molar mass of each element by the total mass of the compound and divide by 100. Let’s try a problem. Propane (C3H8), the fuel commonly used in gas grills, is one of the compounds obtained from petroleum. Calculate the percent composition of propane. II. Empirical Formulas • Formulas for some molecular compounds show a basic ratio of elements. • Multiplying that ratio by any factor can produce formulas for other compounds. • This “basic ratio” is known as the empirical formula. Defining “empirical formula” A formula which gives the lowest whole-number ratio of the atoms of the elements in a compound. • The percent composition is required to calculate the basic ratio of the elements contained in a compound. • The empirical formula, like any chemical formula, may be interpreted in terms of number of atoms and in terms of moles of each atom. Interpreting the empirical formula. Ethyne (aka: acetylene) C2H2 Polystyrene (aka: packing material) C8H8 What is the empirical formula for ethyne and styrene? Determining empirical formulas 1. 2. 3. 4. 5. 6. 7. Identify the percent composition of each element in a compound. Assume 100 g. total molar mass for the compound. Convert percentages to masses. Use molar masses of the elements to convert masses to moles Divide all moles by the smallest number of moles. The numbers obtained will give us the smallest whole number ratio for each element in the compound. If a whole number is not obtained, multiply each ratio by the smallest whole number that will convert both subscripts to whole numbers. Let’s try a problem A compound is analyzed and found to contain 25.9% nitrogen and 74.1% oxygen. What is the empirical formula of the compound? III. Molecular Formulas • Many compounds share the same empirical formula, however they may have different molar masses. • The difference in the molar masses of these compounds are usually simple whole-number multiples of the molar mass of the empirical formula. Let’s look at some examples. Glucose C6H12O6 (180g/mol) Acetic acid C2H4O2 (60g/mol) Formaldehyde CH2O (30g/mol) What can we conclude about the molecular formula? The molecular formula of a compound is either the same as its experimentally determined empirical formula, or it is a simple whole-number multiple of its empirical formula. To determine the molecular formula from the empirical formula, we need to know the compounds molar mass. • We can obtain this molar mass experimentally using a machine known as a mass spectrometer. Determining the molecular formula. 1. Determine the empirical formula. 2. Calculate the molar mass of this formula. 3. Obtain the molar mass of the compound in question. 4. Divide this molar mass by the molar mass of the empirical formula. 5. This is the multiplier used to obtain the molecular formula from the empirical formula. Let’s try a problem. Calculate the molecular formula of a compound whose molar mass is 60.0g/mol and empirical formula is CH4N. Chemical Quantities Chapter 10 The End