Chapter 4 Metal nanoparticles stabilized by chiral ligands with carbohydrate backbone
... molecular species. Diphosphite chiral ligands were also used as stabilisers of palladium nanoparticles.[79] These palladium nanoparticles were active in the ...
... molecular species. Diphosphite chiral ligands were also used as stabilisers of palladium nanoparticles.[79] These palladium nanoparticles were active in the ...
Evaluated kinetic and photochemical data for atmospheric chemistry
... which is also reflected in the present evaluation. This evaluation is also constrained to studies that have quantified gas– liquid uptake kinetics rather than a complete evaluation of aqueous phase kinetics. Further systems will be evaluated in the future and published in future volumes if warranted ...
... which is also reflected in the present evaluation. This evaluation is also constrained to studies that have quantified gas– liquid uptake kinetics rather than a complete evaluation of aqueous phase kinetics. Further systems will be evaluated in the future and published in future volumes if warranted ...
Synthesis of Inorganic Nanostructures in Reverse Micelles
... not be valid any more for the particle formation in these dynamic compartmentalized media. However, if the involved reaction is very slow (e.g., the hydrolysis of tetraethoxysilane to produce silica), the intermicellar exchange does not play an important role in the particle formation, and the react ...
... not be valid any more for the particle formation in these dynamic compartmentalized media. However, if the involved reaction is very slow (e.g., the hydrolysis of tetraethoxysilane to produce silica), the intermicellar exchange does not play an important role in the particle formation, and the react ...
ZnS Core-Shell Quantum Dots
... were used as the Zn and S precursors. The amounts of Zn and S precursors needed to grow a ZnS shell of desired thickness for each CdSe sample were determined as follows: First, the average radius of the CdSe dots was estimated from TEM or SAXS measurements. Next, the ratio of ZnS to CdSe necessary t ...
... were used as the Zn and S precursors. The amounts of Zn and S precursors needed to grow a ZnS shell of desired thickness for each CdSe sample were determined as follows: First, the average radius of the CdSe dots was estimated from TEM or SAXS measurements. Next, the ratio of ZnS to CdSe necessary t ...
2nd Semester Practice Chemistry Final 2009
... a. less effective in solids than in liquids. c. equally effective in gases and in liquids. b. more effective in gases than in solids. d. more effective in liquids than in gases. 44. Which term best describes the process by which particles escape from the surface of a nonboiling liquid and enter the ...
... a. less effective in solids than in liquids. c. equally effective in gases and in liquids. b. more effective in gases than in solids. d. more effective in liquids than in gases. 44. Which term best describes the process by which particles escape from the surface of a nonboiling liquid and enter the ...
Chemical Transport Model - Technical Description
... reactive species, here written for a single species, and in a scaled flux form (Toon et al. 1988), in which geometric scaling factors are introduced into the governing equation to enable the CTM to be coupled to meteorological models which are formulated in a variety of coordinate systems. ...
... reactive species, here written for a single species, and in a scaled flux form (Toon et al. 1988), in which geometric scaling factors are introduced into the governing equation to enable the CTM to be coupled to meteorological models which are formulated in a variety of coordinate systems. ...
Sample pages 2 PDF
... rc, dissolve, concentration of the particles decreases slowly. Atoms formed during dissolution diffuse to coarser particles and absorb on their surface. On the whole, Ostwald ripening broadens standard deviation to the particle size distribution, i.e. it causes an increase in polydispersion, and the ...
... rc, dissolve, concentration of the particles decreases slowly. Atoms formed during dissolution diffuse to coarser particles and absorb on their surface. On the whole, Ostwald ripening broadens standard deviation to the particle size distribution, i.e. it causes an increase in polydispersion, and the ...
Transport and deposition of quantum dots and
... Table 2.1Zeta Potential of the quartz sand in the presence of electrolyte (KCl and CaCl2) used in the transport experiments. .............................................................................................................. 30 Table 2.2Physicochemical properties of the size-fractioned po ...
... Table 2.1Zeta Potential of the quartz sand in the presence of electrolyte (KCl and CaCl2) used in the transport experiments. .............................................................................................................. 30 Table 2.2Physicochemical properties of the size-fractioned po ...
Chemistry of Riming: The Retention of Organic and Inorganic
... In the present experiments single component systems were investigated so that the chemical properties were mainly determined by the substances themselves. This implies that possible interactions between various species present in the liquid phase are not considered (with the exception of CO2 ). As l ...
... In the present experiments single component systems were investigated so that the chemical properties were mainly determined by the substances themselves. This implies that possible interactions between various species present in the liquid phase are not considered (with the exception of CO2 ). As l ...
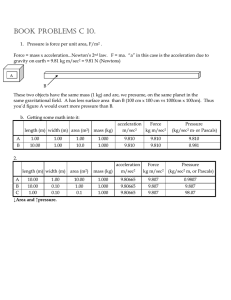
book problems c 10.
... BOOK PROBLEMS C 10. 1. Pressure is force per unit area, F/m2 . Force = mass x acceleration...Newton’s 2nd law. F = ma. “a” in this case is the acceleration due to gravity on earth = 9.81 kg m/sec2 = 9.81 N (Newtons) A B These two objects have the same mass (1 kg) and are, we presume, on the same pla ...
... BOOK PROBLEMS C 10. 1. Pressure is force per unit area, F/m2 . Force = mass x acceleration...Newton’s 2nd law. F = ma. “a” in this case is the acceleration due to gravity on earth = 9.81 kg m/sec2 = 9.81 N (Newtons) A B These two objects have the same mass (1 kg) and are, we presume, on the same pla ...
The shock tube as wave reactor for kinetic studies and material
... Rate expressions are in the form of A Tn exp 2E=RT in units of s 21, cm 3 mol ±1 s 21, cm 6 mol 22 s 21 depending on the reaction order. Reaction order is dependent on number of molecules on left side of equation. ...
... Rate expressions are in the form of A Tn exp 2E=RT in units of s 21, cm 3 mol ±1 s 21, cm 6 mol 22 s 21 depending on the reaction order. Reaction order is dependent on number of molecules on left side of equation. ...
Cold spray coating: review of material systems and future perspectives
... Particles/substrate interaction during the CS deposition process and the resultant bonding, is of great importance in CS technology because of the effect on coating characteristics. Experimental studies and computational modelings have been performed to have a better understanding of the bonding pro ...
... Particles/substrate interaction during the CS deposition process and the resultant bonding, is of great importance in CS technology because of the effect on coating characteristics. Experimental studies and computational modelings have been performed to have a better understanding of the bonding pro ...
Stoichiometric relationships
... Antoine-Laurent Lavoisier (1743–1794) is often called the ‘father of chemistry’. His many accomplishments include the naming of oxygen and hydrogen, the early development of the metric system, and a standardization of chemical nomenclature. Most importantly, he established an understanding of combus ...
... Antoine-Laurent Lavoisier (1743–1794) is often called the ‘father of chemistry’. His many accomplishments include the naming of oxygen and hydrogen, the early development of the metric system, and a standardization of chemical nomenclature. Most importantly, he established an understanding of combus ...
Ice nucleation efficiency of AgI: review and new insights
... at nucleating ice when they are exposed to relative humidity at or even above water saturation. For AgI particles that are completely immersed in water, the freezing temperature increases with increasing AgI surface area. Higher threshold freezing temperatures seem to correlate with improved lattice ...
... at nucleating ice when they are exposed to relative humidity at or even above water saturation. For AgI particles that are completely immersed in water, the freezing temperature increases with increasing AgI surface area. Higher threshold freezing temperatures seem to correlate with improved lattice ...
Topic 1 - Coral Gables Senior High
... The term stoichiometry is derived from two Greek words – stoicheion for element and metron for measure. Stoichiometry describes the relationships between the amounts of reactants and products during chemical reactions. As it is known that matter is conserved during chemical change, stoichiometry is ...
... The term stoichiometry is derived from two Greek words – stoicheion for element and metron for measure. Stoichiometry describes the relationships between the amounts of reactants and products during chemical reactions. As it is known that matter is conserved during chemical change, stoichiometry is ...
topic: chemical formula, chemical equations and stoichiometry
... [Relative Atomic Mass; H,7; N,14;Mg, 24; S, 32; O,16] a. Relative molecule Mass of Ammonia, NH3 =Mass of one nitrogen atom + mass of three hydrogen atoms =RAM of nitrogen x number of nitrogen atom + RAM of hydrogen x number of hydrogen atom Molar Mass of ammonia, NH3 = 14 x1 + 1x3 = 17 b. The relati ...
... [Relative Atomic Mass; H,7; N,14;Mg, 24; S, 32; O,16] a. Relative molecule Mass of Ammonia, NH3 =Mass of one nitrogen atom + mass of three hydrogen atoms =RAM of nitrogen x number of nitrogen atom + RAM of hydrogen x number of hydrogen atom Molar Mass of ammonia, NH3 = 14 x1 + 1x3 = 17 b. The relati ...
Change in the morphology of ZnO nanoparticles upon changing the
... Different synthesis methods for the preparation of ZnO nanostructures have been employed in the literature including sol–gel (Tokumoto et al. 2003; Hohenberger et al. 1992), micro-emulsion (Singhal et al. 1997), mechanochemical processing (Tsuzuki and McCormick 2001), vapour phase transport (Zhang e ...
... Different synthesis methods for the preparation of ZnO nanostructures have been employed in the literature including sol–gel (Tokumoto et al. 2003; Hohenberger et al. 1992), micro-emulsion (Singhal et al. 1997), mechanochemical processing (Tsuzuki and McCormick 2001), vapour phase transport (Zhang e ...
Techniques for the Preparation of Solid Lipid Nano and Microparticles
... Practically, SLN can be derived from the emulsions for parenteral nutrition just by replacing the liquid lipid (oil) of the emulsion droplets by a solid lipid. In contrast to emulsions for parenteral nutrition which are normally stabilised only by lecithin, the SLN are stabilised also by other surfa ...
... Practically, SLN can be derived from the emulsions for parenteral nutrition just by replacing the liquid lipid (oil) of the emulsion droplets by a solid lipid. In contrast to emulsions for parenteral nutrition which are normally stabilised only by lecithin, the SLN are stabilised also by other surfa ...
Ch 12 Solutions
... - An aerosol is composed of liquid droplets or solid particles dispersed in a gas, like hair spray and smoke. - An emulsion is composed of liquid droplets dispersed in another liquid, like oil and vinegar salad dressing. - A sol is composed of solid particles dispersed in a liquid, like silt in a ri ...
... - An aerosol is composed of liquid droplets or solid particles dispersed in a gas, like hair spray and smoke. - An emulsion is composed of liquid droplets dispersed in another liquid, like oil and vinegar salad dressing. - A sol is composed of solid particles dispersed in a liquid, like silt in a ri ...
Model for acid-base chemistry in nanoparticle growth (MABNAG)
... compound i (as a result of composition change due to condensation) for aqueous solution is τa,i = mw /Ki × τs,i (Seinfeld and Pandis, 2006) where mw is the total mass of liquid water and Ki is the equilibrium constant. The characteristic time for uptake of compound i from gas phase to particles by d ...
... compound i (as a result of composition change due to condensation) for aqueous solution is τa,i = mw /Ki × τs,i (Seinfeld and Pandis, 2006) where mw is the total mass of liquid water and Ki is the equilibrium constant. The characteristic time for uptake of compound i from gas phase to particles by d ...
Stoichiometry notes 1
... 2. Label your given and target substances. 3. Convert your given unit(s) to moles of given substance using the appropriate conversion factor. 4. Convert moles of given substance to moles of target substance using the mole ratio from the balanced equation. 5. Convert moles of target substance to the ...
... 2. Label your given and target substances. 3. Convert your given unit(s) to moles of given substance using the appropriate conversion factor. 4. Convert moles of given substance to moles of target substance using the mole ratio from the balanced equation. 5. Convert moles of target substance to the ...
(Ir)reversibility and entropy
... exclude a priori the possibility of triple collisions, simultaneous collisions between particles and the boundary, or again an infinity of collisions occurring in a finite time. However, such events are of probability zero if the initial conditions are drawn at random with respect to Lebesgue measur ...
... exclude a priori the possibility of triple collisions, simultaneous collisions between particles and the boundary, or again an infinity of collisions occurring in a finite time. However, such events are of probability zero if the initial conditions are drawn at random with respect to Lebesgue measur ...
The role of aqueous-phase oxidation in the A
... Atmospheric particulate matter (or "aerosol") is known to have important implications for cli- ...
... Atmospheric particulate matter (or "aerosol") is known to have important implications for cli- ...
Aerosol

An aerosol is a colloid of fine solid particles or liquid droplets, in air or another gas. Aerosols can be natural or artificial. Examples of natural aerosols are fog, forest exudates and geyser steam. Examples of artificial aerosols are haze, dust, particulate air pollutants and smoke. The liquid or solid particles have diameter mostly smaller than 1 μm or so; larger particles with a significant settling speed make the mixture a suspension, but the distinction is not clear-cut. In general conversation, aerosol usually refers to an aerosol spray that delivers a consumer product from a can or similar container. Other technological applications of aerosols include dispersal of pesticides, medical treatment of respiratory illnesses, and combustion technology. Diseases can also spread by means of small droplets in the breath, also called aerosols.Aerosol science covers generation and removal of aerosols, technological application of aerosols, effects of aerosols on the environment and people, and a wide variety of other topics.