* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Effect of Ig transgene on B cell activation
Survey
Document related concepts
Transcript
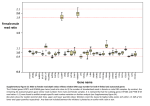
Explanatory text for supplementary figures, “How self-tolerance prevents B cell mitogenesis: contrasting effects of FK506 provide a rationale for improved immunosuppressants”. Corresponding author: Chris Goodnow Effect of purification method We used two purification methods to isolate naïve and tolerant cells: a negative depletion protocol that depletes splenocytes of T cells and macrophages and a positive selection for follicular B cells using FACS. Negative depletion has the advantages of speed, low cost and lack of engagement of B cell surface proteins but the purity is about 90% and follicular and mantle zone B cells are both purified. FACS selection of B cells allows higher purity and selection of follicular cells away from cells with a mantle zone phenotype but takes longer and requires engagement of cell surface molecules. Therefore, these two protocols are somewhat complementary and we reported genes that were significantly changed in the negative depletion samples between tolerant and naïve cells and which were consistently altered in the FACS sorted preparations. The effect of purification protocol on the expression levels of transcripts reported in the manuscript figure 1 can be seen in supplementary figures 1a and 1b. In figures 1c and 1d examples of some myeloid and erythoid transcripts are shown that would have been called as regulated in tolerance if not for the evidence that they are depleted in the FACS purified samples. Effect of Ig transgene on B cell activation In the activation experiments, we assayed HyHEL10 transgenic cells (IgHEL cells) which are monoclonal for an antigen receptor reactive to HEL, stimulated with HEL. We also assayed B cell that were non-transgenic (B6 cells) and activated with an antibody raised against the antigen receptor (anti-mu) which is predicted to stimulate all cells irrespective of receptor specificity. A multivariate analysis of the response of B6 cells to anti-Ig and IgHEL cells to HEL was performed and is presented in the supplementary data. In brief, there is only one significant change between B6 and IgHEL cells: expression of Vk23, the V region used by the HyHEL10 light chain transgene, was increased in IgHEL cells (1.8 fold change, p<0.0001). The two cell types differ slightly in their response to stimulation: MIP1a and MIP1b appear to be induced to 2 fold higher levels by HEL stimulation of Ig cells than by anti-Ig stimulation of B6 cells (p<0.002 and 0.0005 respectively). Other than this difference (which is only strictly significant for MIP1b), the cells appeared to respond to each stimulation identically and the data are presented together for clarity in manuscript figures 2 and 3. The data are presented in different colours for each cell type in supplementary figures 2 and 3. Effect of in vitro incubation A strength of this model is that the tolerance response is measured by comparing anergic cells stimulated chronically by self HEL and naïve unstimulated cells, both purified and lysed immediately ex vivo. While B cell activation is most reliably and synchronously studied in vitro, assays for tolerance responses in vitro are difficult to interpret because of the tendency of B cells to die in vitro if not stimulated mitogenically. It is not possible to isolate and purify RNA from B cells quickly enough to study early responses to antigen administered in vivo: the time taken in purification would be significant relative to the time of antigen exposure. Moreover, we know that for the first several hours after acute antigen stimulation in vivo it is difficult to purify the cells from spleen cell suspensions because of strong adhesion to stromal cells. Effects of the drug inhibitors and calcium chelation are likewise best defined in vitro. Therefore, for defining activation responses we decided to purify the B cells first and then to study the activation response in vitro, allowing aliquots or the same cell preparations to be used as controls that were mock stimulated by identical culture in the absence of antigenic stimulus. Thus in these comparisons, the only systematic differences between the activated or tolerant cell populations and their respective controls are the antigen stimuli. Within our dataset the IgHEL cells have been purified identically for defining genes in the activation and tolerance response: the only other difference is that for the former case the cells were incubated in vitro for one hour whereas in the latter case cells were lysed directly after purification. The average expression profiles of unstimulated IgHEL cells that were or were not incubated in vitro are quite similar. Using a multivariate analysis we found that in vitro incubation caused significant changes of 1.8 fold or more in 299 transcripts. This in vitro culture/in vivo withdrawal set did not resemble a partial activation response triggered by antigen receptor stimulation: in fact, some of the genes that were upregulated by antigen are downregulated by in vitro incubation and vice versa (supplementary figure 4a, b). While it is clear from this analysis that there are gene expression changes resulting from in vitro culture, the chief point is that genes which are triggered by antigen in vitro compared with in vitro incubation alone are unequivocally assigned to the B cell activation response. Some activation response genes may be missed if they are also induced simply by in vitro culture, as appears to be the case for B7.2, but this does not alter the conclusions drawn from the activation set presented in figure 2. Exemplar accession numbers The 6519 data points for each sample consist of: i) 172 controls ii) 2780 ESTs iii) 3074 genes iv) 493 where no Entrez definition line is given (blanks) An automated Entrez search was performed for the 6347 transcripts that were not controls. Of the 2780 transcripts in which the exemplar accession number given referred to an EST, Entrez matches were found for all but 39. These 39 all came from the same library: the absence of these from the current database is likely to reflect withdrawal from the database of sequences from that library. The remaining 2741 EST transcripts had an Entrez match. The results of this search are in the spreadsheet “EST entrez hits” in the supplementary information. Of the 493 “blanks”, 336 gave no Entrez match. 289 of these were from a library that was withdrawn from the database between the design of these arrays and the experiments. The explanation foe the absence of a hit for the remaining 47 is unclear. The results of the 493 “blanks” are in the spreadsheet “blank entrez hits” in the supplementary information. All the exemplar accession numbers for the 3074 genes had a match with Entrez and this data is in the spreadsheet “gene entrez hits”, supplementary information. In total, it is not possible to determine the source sequence for 375 transcripts tiled in these arrays, without access to the sequences that were used on the arrays (information that is propietary to Affymetrix). This represents about 6% of the genes on these arrays. Among the genes discussed in this manuscript, only Ebi2 came from a withdrawn EST. The identity of this probeset was confirmed by “one-off” disclosure of the tiled oligonucleotide sequences by Affymetrix. Control transcripts tiled on all 4 arrays of the set 43 tilings representing 15 controls are tiled on each of the 4 arrays. These controls include bacterial transcripts, added to the hybridisation solutions as “spikes” of known concentration, GAPDH, ribosomal RNA and mouse repeat sequences. The software used to compile the data presented in the supplementary information spreadsheet only recognised the probesetID for uniqueness. The result of this is that the four datapoints for each tiling control (one datapoint per chip) were all set to the hybridisation intensity of the first chip. The datapoints for the non-control transcripts, which are analysed and presented in the manuscript, are not affected by this problem. The bug was fixed during the course of the experiments so that for later experiments (for example the FACS purified B cells) the true data for all four chips is shown and the chip: chip variability measuring the same transcript is reflected.