Half-life and Radioactive Decay guided notes
... cloth and paper. Beta particles can penetrate deeply into skin and potentially However, these particles cannot penetrate thin layers of ...
... cloth and paper. Beta particles can penetrate deeply into skin and potentially However, these particles cannot penetrate thin layers of ...
Nuclear Chemistry - Mona Shores Blogs
... – After this point, there is too little carbon-14 left to be measurable ...
... – After this point, there is too little carbon-14 left to be measurable ...
notes ch 39 1st half Atomic Nucleus and Radioactivity
... • Alpha rays are streams of particles that are made of two protons and two neutrons, and are identical to the nuclei of a helium atom. • These particles are called alpha particles. • They can be stopped by a few sheets of thin paper or a sheet of heavy paper. • They quickly grab some random loose el ...
... • Alpha rays are streams of particles that are made of two protons and two neutrons, and are identical to the nuclei of a helium atom. • These particles are called alpha particles. • They can be stopped by a few sheets of thin paper or a sheet of heavy paper. • They quickly grab some random loose el ...
File
... are too many protons in a nucleus. In this case the element will emit radiation in the form of positively charged particles called alpha particles. Alpha particles are considered not dangerous as it can be stopped by a sheet of paper Beta decay - Beta decay is caused when there are too many neutrons ...
... are too many protons in a nucleus. In this case the element will emit radiation in the form of positively charged particles called alpha particles. Alpha particles are considered not dangerous as it can be stopped by a sheet of paper Beta decay - Beta decay is caused when there are too many neutrons ...
Isotopes of an atom have the same number of protons, but a different
... ● Gamma rays are waves, not particles. ● This means that they have no mass and no charge. ● In Gamma decay: atomic number unchanged, atomic mass unchanged. ● Gamma rays have a high penetrating power - it takes a thick sheet of metal such as lead to reduce them. ● Gamma rays do not directly ionize ot ...
... ● Gamma rays are waves, not particles. ● This means that they have no mass and no charge. ● In Gamma decay: atomic number unchanged, atomic mass unchanged. ● Gamma rays have a high penetrating power - it takes a thick sheet of metal such as lead to reduce them. ● Gamma rays do not directly ionize ot ...
Topic 6 – Benefits and drawbacks of using radioactive materials
... The risk of harm decreases with distance from the sourceradioactive substances are always handled with tongs, and kept away from other people Protective clothing is worn in case the radioactive source happens to come into contact with the skin The most penetrating radiation (gamma rays) can be stop ...
... The risk of harm decreases with distance from the sourceradioactive substances are always handled with tongs, and kept away from other people Protective clothing is worn in case the radioactive source happens to come into contact with the skin The most penetrating radiation (gamma rays) can be stop ...
NUCLEAR CHEMISTRY PACKET - Student
... Each radioactive isotope has a specific mode and rate of decay (half-life). (4.4a) A change in the nucleus of an atom that converts it from one element to another is called transmutation. This can occur naturally or can be induced by the bombardment of the nucleus by high-energy particles. (5.3a) Sp ...
... Each radioactive isotope has a specific mode and rate of decay (half-life). (4.4a) A change in the nucleus of an atom that converts it from one element to another is called transmutation. This can occur naturally or can be induced by the bombardment of the nucleus by high-energy particles. (5.3a) Sp ...
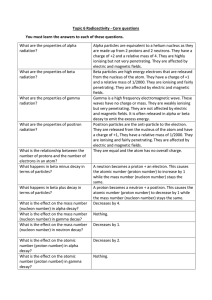
Topic 6 Radioactivity Core Questions
... How many protons there are in the nucleus of an atom, number (proton number) tell you? ion or isotope and so what type of atom it is. What information does the mass The total number of protons + neutrons in the nucleus of number (nucleon number) tell you? an atom. What happens to an atom when an Ele ...
... How many protons there are in the nucleus of an atom, number (proton number) tell you? ion or isotope and so what type of atom it is. What information does the mass The total number of protons + neutrons in the nucleus of number (nucleon number) tell you? an atom. What happens to an atom when an Ele ...
By what process do most stars release energy? A. Electromagnetic
... Nuclear energy can be generated by ssion or fusion. Fusion is not currently being used in reactors as an energy source. Why is this? A. ...
... Nuclear energy can be generated by ssion or fusion. Fusion is not currently being used in reactors as an energy source. Why is this? A. ...
particle - Uplift North Hills
... α and β - particles have energies measured in MeV. To ionized an atom requires about 10 eV, so each particle can ionize 105 atoms before they have run out of energy. When radiation ionizes atoms that are part of a living cell, it can effect the ability of the cell to carry out its function or even c ...
... α and β - particles have energies measured in MeV. To ionized an atom requires about 10 eV, so each particle can ionize 105 atoms before they have run out of energy. When radiation ionizes atoms that are part of a living cell, it can effect the ability of the cell to carry out its function or even c ...
Radioactivity - Williamstown Independent Schools
... number +1) with the same mass and a beta particle. • A beta particle (β) is the same as an ...
... number +1) with the same mass and a beta particle. • A beta particle (β) is the same as an ...
Revision of Atomic Structure and Nuclide Notations Nuclide
... Gamma emission often happens at the same time but has no effect on the mass number or atomic number. It is not included in nuclear equations. Balancing the Numbers! In nuclear equations the top numbers (mass numbers) must add up to the same number on both sides of the equation. The bottom numbers, t ...
... Gamma emission often happens at the same time but has no effect on the mass number or atomic number. It is not included in nuclear equations. Balancing the Numbers! In nuclear equations the top numbers (mass numbers) must add up to the same number on both sides of the equation. The bottom numbers, t ...
6.2 - Hockerill Students
... gamma rays are not deflected by the field. Alpha and beta particles are also affected by an electric field - in other words, there is a force on them if they pass between oppositely charged plates. ...
... gamma rays are not deflected by the field. Alpha and beta particles are also affected by an electric field - in other words, there is a force on them if they pass between oppositely charged plates. ...
Ch 10 Nuclear Chemistry
... • Radioisotopes spontaneously change into other isotopes over time and is said to undergo nuclear ...
... • Radioisotopes spontaneously change into other isotopes over time and is said to undergo nuclear ...
Nuclear Chemistry
... e. Students know alpha, beta, and gamma radiation produce different amounts and kinds of damage in matter and have different penetrations. ...
... e. Students know alpha, beta, and gamma radiation produce different amounts and kinds of damage in matter and have different penetrations. ...
Gamma Decay - UNLV Radiochemistry
... Emission of electromagnetic radiation ( radiation) internal-conversion electrons newly created electron and positron (higher energy) Internal conversion from interaction between nucleus and extranuclear electrons leading to emission of atomic electron kinetic energy equal to difference bet ...
... Emission of electromagnetic radiation ( radiation) internal-conversion electrons newly created electron and positron (higher energy) Internal conversion from interaction between nucleus and extranuclear electrons leading to emission of atomic electron kinetic energy equal to difference bet ...
Quarter 1 Unit 3 Radioactivitypptx
... gamma decay only energy is released! ◦ Gamma decay does not generally occur alone, it occurs with other modes of decay. (alpha or beta) ...
... gamma decay only energy is released! ◦ Gamma decay does not generally occur alone, it occurs with other modes of decay. (alpha or beta) ...
Multiple Choice Questions
... (3) both mass and charge (4) neither mass nor charge 2. Which of the following particles has the greatest mass? (1) an alpha particle (2) a beta particle (3) a proton (4) an electron 3. Which nuclear emission is negatively charged? (1) an alpha particle (2) a beta particle (3) a neutron (4) an posit ...
... (3) both mass and charge (4) neither mass nor charge 2. Which of the following particles has the greatest mass? (1) an alpha particle (2) a beta particle (3) a proton (4) an electron 3. Which nuclear emission is negatively charged? (1) an alpha particle (2) a beta particle (3) a neutron (4) an posit ...
Radioactivity
... As nuclei in a radioactive sample decay, the activity gets less, till eventually there are no more nuclei left to decay. The half-life of a radioactive source is the time for the activity to fall by half Or, since the activity of a source is directly proportional to the number of undecayed nuclei, t ...
... As nuclei in a radioactive sample decay, the activity gets less, till eventually there are no more nuclei left to decay. The half-life of a radioactive source is the time for the activity to fall by half Or, since the activity of a source is directly proportional to the number of undecayed nuclei, t ...
Nuclear Reactions
... penetrating than Alpha particles • Can be stopped by a sheet of aluminum foil. ...
... penetrating than Alpha particles • Can be stopped by a sheet of aluminum foil. ...
10380 radioactivity - The Described and Captioned Media Program
... Nuclear chemistry is the study of those elements that are radioactive. Radiation is the spontaneous emission of particles or rays from the nucleus of a radioactive element. Not all nuclei decay spontaneously and so some, therefore, are stable. Others, however, are unstable. There are common types of ...
... Nuclear chemistry is the study of those elements that are radioactive. Radiation is the spontaneous emission of particles or rays from the nucleus of a radioactive element. Not all nuclei decay spontaneously and so some, therefore, are stable. Others, however, are unstable. There are common types of ...
Gamma ray
Gamma radiation, also known as gamma rays, and denoted by the Greek letter γ, refers to electromagnetic radiation of an extremely high frequency and therefore consists of high-energy photons. Gamma rays are ionizing radiation, and are thus biologically hazardous. They are classically produced by the decay of atomic nuclei as they transition from a high energy state to a lower state known as gamma decay, but may also be produced by other processes. Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900, while studying radiation emitted from radium. Villard's radiation was named ""gamma rays"" by Ernest Rutherford in 1903.Natural sources of gamma rays on Earth include gamma decay from naturally occurring radioisotopes, and secondary radiation from atmospheric interactions with cosmic ray particles. Rare terrestrial natural sources produce gamma rays that are not of a nuclear origin, such as lightning strikes and terrestrial gamma-ray flashes. Additionally, gamma rays are produced by a number of astronomical processes in which very high-energy electrons are produced, that in turn cause secondary gamma rays via bremsstrahlung, inverse Compton scattering, and synchrotron radiation. However, a large fraction of such astronomical gamma rays are screened by Earth's atmosphere and can only be detected by spacecraft.Gamma rays typically have frequencies above 10 exahertz (or >1019 Hz), and therefore have energies above 100 keV and wavelengths less than 10 picometers (10−12 meter), which is less than the diameter of an atom. However, this is not a hard and fast definition, but rather only a rule-of-thumb description for natural processes. Electromagnetic radiation from radioactive decay of atomic nuclei is referred to as ""gamma rays"" no matter its energy, so that there is no lower limit to gamma energy derived from radioactive decay. This radiation commonly has energy of a few hundred keV, and almost always less than 10 MeV. In astronomy, gamma rays are defined by their energy, and no production process needs to be specified. The energies of gamma rays from astronomical sources range to over 10 TeV, an energy far too large to result from radioactive decay. A notable example is extremely powerful bursts of high-energy radiation referred to as long duration gamma-ray bursts, of energies higher than can be produced by radioactive decay. These bursts of gamma rays, thought to be due to the collapse of stars called hypernovae, are the most powerful events so far discovered in the cosmos.