Unit 2: The Atom
... •Alpha decay is how elements greater than Bismuth try to become stable. •They will emit an alpha particle (2 neutrons and 2 protons) to try to become stable. •Alpha reactions will always have He on the right side! •To balance: write the upper and lower equations! ...
... •Alpha decay is how elements greater than Bismuth try to become stable. •They will emit an alpha particle (2 neutrons and 2 protons) to try to become stable. •Alpha reactions will always have He on the right side! •To balance: write the upper and lower equations! ...
3 main types of particle
... (the beta particle which is ejected) and a proton (which stays in the nucleus) During beta decay the mass number stays the same but the proton number goes up by 1. Remember the electron comes from the nucleus! ...
... (the beta particle which is ejected) and a proton (which stays in the nucleus) During beta decay the mass number stays the same but the proton number goes up by 1. Remember the electron comes from the nucleus! ...
Section G23: Atoms and Radioactivity
... (min), second (s). 7.2 describe the structure of an atom in terms of protons, neutrons and electrons and use symbols such as C 14 6 to describe particular nuclei 7.3 understand the terms atomic (proton) number, mass (nucleon) number and isotope 7.4 understand that alpha and beta particles and gamma ...
... (min), second (s). 7.2 describe the structure of an atom in terms of protons, neutrons and electrons and use symbols such as C 14 6 to describe particular nuclei 7.3 understand the terms atomic (proton) number, mass (nucleon) number and isotope 7.4 understand that alpha and beta particles and gamma ...
Chapter 2, section 4 Formation of Elements
... of the man-made isotopes are unstable. Unstable isotopes can become stable by releasing different types of particles. This process is called radioactive decay and the elements which undergo this process are called radioisotopes/. ...
... of the man-made isotopes are unstable. Unstable isotopes can become stable by releasing different types of particles. This process is called radioactive decay and the elements which undergo this process are called radioisotopes/. ...
on Nuclear Physics - Good Earth School
... PROPERTIES OF BETA PARTICLES negatively charged. So they are deflected by electric or magnetic fields ionizing power of beta particles is less than alpha but more than gamma particles penetrating power is more than alpha but less than gamma particles ...
... PROPERTIES OF BETA PARTICLES negatively charged. So they are deflected by electric or magnetic fields ionizing power of beta particles is less than alpha but more than gamma particles penetrating power is more than alpha but less than gamma particles ...
Rhenium isotopes in geochronology Stable isotope Relative atomic
... element (chemical element) – a species of atoms; all atoms with the same number of protons in the atomic nucleus. A pure chemical substance composed of atoms with the same number of protons in the atomic nucleus [703]. [return] gamma rays (gamma radiation) – a stream of high-energy electromagnetic r ...
... element (chemical element) – a species of atoms; all atoms with the same number of protons in the atomic nucleus. A pure chemical substance composed of atoms with the same number of protons in the atomic nucleus [703]. [return] gamma rays (gamma radiation) – a stream of high-energy electromagnetic r ...
Chapter 26
... Z X Z 1Y e ◦ is the symbol for the antineutrino To summarize, in beta decay, the following pairs of particles are emitted ◦ An electron and an antineutrino ◦ A positron and a neutrino ...
... Z X Z 1Y e ◦ is the symbol for the antineutrino To summarize, in beta decay, the following pairs of particles are emitted ◦ An electron and an antineutrino ◦ A positron and a neutrino ...
Chapter 10
... • Remember for section 2.2, this defines an isotope of boron. • In nuclear chemistry this is often called a nuclide. • This is not the only isotope (nuclide) of boron. – boron-10 also exists – How many protons and neutrons does boron-10 have? – 5 protons, 5 neutrons ...
... • Remember for section 2.2, this defines an isotope of boron. • In nuclear chemistry this is often called a nuclide. • This is not the only isotope (nuclide) of boron. – boron-10 also exists – How many protons and neutrons does boron-10 have? – 5 protons, 5 neutrons ...
Half Life
... TRUE/FALSE 19. An element that emits rays is said to be contaminated. 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The f ...
... TRUE/FALSE 19. An element that emits rays is said to be contaminated. 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The f ...
Grade 10S Physics T3W5 material
... 5‐ By measuring in the rocks the present rate of radioactivity or the concentration of the non radioactive products of radioactivity, the age of the rock can be determined. 6‐ a. Lower doses may cause Leukemia within 2years or other forms of cancer 15 to 20 years. b. Radiation can cause genetic a ...
... 5‐ By measuring in the rocks the present rate of radioactivity or the concentration of the non radioactive products of radioactivity, the age of the rock can be determined. 6‐ a. Lower doses may cause Leukemia within 2years or other forms of cancer 15 to 20 years. b. Radiation can cause genetic a ...
Nuc Chem PP - Liberty Union High School District
... energy that is released. • Gamma rays are electromagnetic waves. • They have no mass. • Gamma radiation has no charge. – Most Penetrating, can be stopped by 1m thick concrete or a several cm thick sheet of lead. ...
... energy that is released. • Gamma rays are electromagnetic waves. • They have no mass. • Gamma radiation has no charge. – Most Penetrating, can be stopped by 1m thick concrete or a several cm thick sheet of lead. ...
What is “Radiation”?
... nanometers can remove electrons from the outer atomic shells. • This process produces ions. • Ions can interact with living tissue to produce biological damage. • A major source of ionizing radiation is nuclear transformation. ...
... nanometers can remove electrons from the outer atomic shells. • This process produces ions. • Ions can interact with living tissue to produce biological damage. • A major source of ionizing radiation is nuclear transformation. ...
(neutron/proton ratio is 1).
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
NUCLEAR CHEMISTRY: INTRO
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
Chapter 32 Nuclear Physics
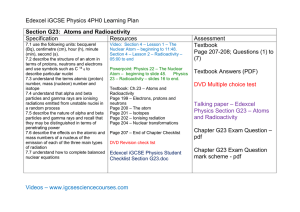
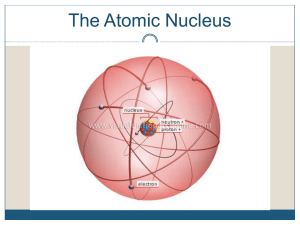
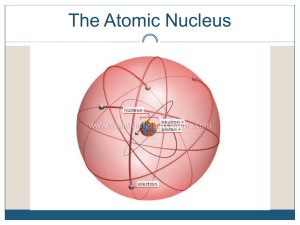
... The nucleus of an atom is incredibly small. The diameter of the nucleus is about 100,000 times smaller than the diameter of the atom. Although small, the nucleus contains more than 99.9% of the mass of an atom, because each of its constitutes (protons and neutrons) is about 1800 times more massive t ...
... The nucleus of an atom is incredibly small. The diameter of the nucleus is about 100,000 times smaller than the diameter of the atom. Although small, the nucleus contains more than 99.9% of the mass of an atom, because each of its constitutes (protons and neutrons) is about 1800 times more massive t ...
radioactivity-ppt
... unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. There are numerous types of radioactive decay. The ...
... unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. There are numerous types of radioactive decay. The ...
Radioactivity
... unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. There are numerous types of radioactive decay. The ...
... unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. There are numerous types of radioactive decay. The ...
Unit 3 – Atomic Structure
... Alpha Particles are very massive, but travel slower (low penetrating power) Can cause significant tissue damage if not shielded Shielding can be accomplished with clothing or paper ...
... Alpha Particles are very massive, but travel slower (low penetrating power) Can cause significant tissue damage if not shielded Shielding can be accomplished with clothing or paper ...
Isotopes of an atom have the same number of protons, but a different
... ● Gamma rays are waves, not particles. ● This means that they have no mass and no charge. ● In Gamma decay: atomic number unchanged, atomic mass unchanged. ● Gamma rays have a high penetrating power - it takes a thick sheet of metal such as lead to reduce them. ● Gamma rays do not directly ionize ot ...
... ● Gamma rays are waves, not particles. ● This means that they have no mass and no charge. ● In Gamma decay: atomic number unchanged, atomic mass unchanged. ● Gamma rays have a high penetrating power - it takes a thick sheet of metal such as lead to reduce them. ● Gamma rays do not directly ionize ot ...
cps ch 10 notes
... passing through cracks or holes in their foundation. • Most areas – amount of uranium in rocks and soil is very small. ...
... passing through cracks or holes in their foundation. • Most areas – amount of uranium in rocks and soil is very small. ...
AP Chem
... even number of neutrons. The least stable situation is when both numbers are odd. There are only four (or five) stable odd/odd nuclei. Nuclides with a mass number over 200 usually undergo alpha decay. They emit a particle consisting of two protons and two neutrons. Nuclides with too many neutrons un ...
... even number of neutrons. The least stable situation is when both numbers are odd. There are only four (or five) stable odd/odd nuclei. Nuclides with a mass number over 200 usually undergo alpha decay. They emit a particle consisting of two protons and two neutrons. Nuclides with too many neutrons un ...
Gamma ray
Gamma radiation, also known as gamma rays, and denoted by the Greek letter γ, refers to electromagnetic radiation of an extremely high frequency and therefore consists of high-energy photons. Gamma rays are ionizing radiation, and are thus biologically hazardous. They are classically produced by the decay of atomic nuclei as they transition from a high energy state to a lower state known as gamma decay, but may also be produced by other processes. Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900, while studying radiation emitted from radium. Villard's radiation was named ""gamma rays"" by Ernest Rutherford in 1903.Natural sources of gamma rays on Earth include gamma decay from naturally occurring radioisotopes, and secondary radiation from atmospheric interactions with cosmic ray particles. Rare terrestrial natural sources produce gamma rays that are not of a nuclear origin, such as lightning strikes and terrestrial gamma-ray flashes. Additionally, gamma rays are produced by a number of astronomical processes in which very high-energy electrons are produced, that in turn cause secondary gamma rays via bremsstrahlung, inverse Compton scattering, and synchrotron radiation. However, a large fraction of such astronomical gamma rays are screened by Earth's atmosphere and can only be detected by spacecraft.Gamma rays typically have frequencies above 10 exahertz (or >1019 Hz), and therefore have energies above 100 keV and wavelengths less than 10 picometers (10−12 meter), which is less than the diameter of an atom. However, this is not a hard and fast definition, but rather only a rule-of-thumb description for natural processes. Electromagnetic radiation from radioactive decay of atomic nuclei is referred to as ""gamma rays"" no matter its energy, so that there is no lower limit to gamma energy derived from radioactive decay. This radiation commonly has energy of a few hundred keV, and almost always less than 10 MeV. In astronomy, gamma rays are defined by their energy, and no production process needs to be specified. The energies of gamma rays from astronomical sources range to over 10 TeV, an energy far too large to result from radioactive decay. A notable example is extremely powerful bursts of high-energy radiation referred to as long duration gamma-ray bursts, of energies higher than can be produced by radioactive decay. These bursts of gamma rays, thought to be due to the collapse of stars called hypernovae, are the most powerful events so far discovered in the cosmos.