Document
... EB = strong nuclear force binding -surface tension binding + spin pairing +shell binding-Coulomb repulsion 1) strong nuclear force -- the more nucleons the better 2) surface tension -- the less surface/volume the better (U better than He) 3) spin pairing -- neutrons and protons have + and - spins, p ...
... EB = strong nuclear force binding -surface tension binding + spin pairing +shell binding-Coulomb repulsion 1) strong nuclear force -- the more nucleons the better 2) surface tension -- the less surface/volume the better (U better than He) 3) spin pairing -- neutrons and protons have + and - spins, p ...
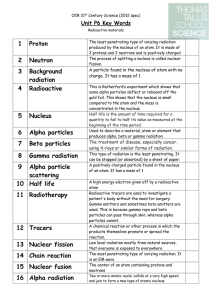
3 Background radiation
... Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. A chemical reaction or other process in which the produ ...
... Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. A chemical reaction or other process in which the produ ...
Activity 3.1
... 1. Shortly after the discovery of X-rays, the French scientist Henri Becquerel tried to find out whether any elements spontaneously emit X-rays. Becquerel was conducting experiments, which started with the exposure of a uranium-bearing crystal to sunlight. Once the crystal had v=been in the sunshine ...
... 1. Shortly after the discovery of X-rays, the French scientist Henri Becquerel tried to find out whether any elements spontaneously emit X-rays. Becquerel was conducting experiments, which started with the exposure of a uranium-bearing crystal to sunlight. Once the crystal had v=been in the sunshine ...
What do these food items have in common?
... are exposed briefly to highenergy radiation to control insects, mold, bacteria, and spoilage. ...
... are exposed briefly to highenergy radiation to control insects, mold, bacteria, and spoilage. ...
What do I know about……
... radiations emitted from unstable nuclei in a random process describe the nature of alpha and beta particles and gamma rays and recall that they may be distinguished in terms of penetrating power describe the effects on the atomic and mass numbers of a nucleus of the emission of each of the three mai ...
... radiations emitted from unstable nuclei in a random process describe the nature of alpha and beta particles and gamma rays and recall that they may be distinguished in terms of penetrating power describe the effects on the atomic and mass numbers of a nucleus of the emission of each of the three mai ...
Notes: Nuclear Chemistry
... Gamma Rays: As the name implies, these are not particles but high-energy, short-wavelength, electromagnetic radiation waves emitted from the nucleus of an atom. They are very similar to X-rays but have a shorter wavelength and therefore more energy. The penetrating ability of gamma rays is much grea ...
... Gamma Rays: As the name implies, these are not particles but high-energy, short-wavelength, electromagnetic radiation waves emitted from the nucleus of an atom. They are very similar to X-rays but have a shorter wavelength and therefore more energy. The penetrating ability of gamma rays is much grea ...
Alpha
... Neutrons have no charge. Electrons are small and are ------------------ charged. Protons & neutrons are in an atom’s -----------------Electrons are found in ------------------ around an atom’s nucleus. The mass number is equal to an atom’s number of protons and ----------------- added together. 7. T ...
... Neutrons have no charge. Electrons are small and are ------------------ charged. Protons & neutrons are in an atom’s -----------------Electrons are found in ------------------ around an atom’s nucleus. The mass number is equal to an atom’s number of protons and ----------------- added together. 7. T ...
Nuclear Radiation and Decay File
... • He hypothesized that the uranium salt had emitted some unknown invisible rays, or radiation, that had darkened the film. ...
... • He hypothesized that the uranium salt had emitted some unknown invisible rays, or radiation, that had darkened the film. ...
Alpha Decay
... excitation and ionization of surrounding atoms. Alpha particles are the least penetrating radiation featuring a relatively straight path over a short distance (several cm in air). The specific ionization of alpha particles is very high. Could barely pass through a single sheet of paper. Deflec ...
... excitation and ionization of surrounding atoms. Alpha particles are the least penetrating radiation featuring a relatively straight path over a short distance (several cm in air). The specific ionization of alpha particles is very high. Could barely pass through a single sheet of paper. Deflec ...
Powerpoint Slides
... Radioactive Decay If you start out with a sample of parent atoms (No), after some time there will be fewer because of radioactive decay into the daughter atoms. ...
... Radioactive Decay If you start out with a sample of parent atoms (No), after some time there will be fewer because of radioactive decay into the daughter atoms. ...
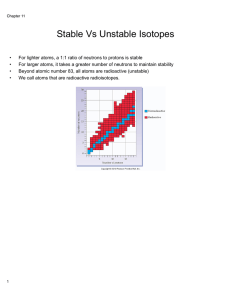
The Band of Stability
... Radioactivity is the spontaneous emission of radiation by nuclei. Radioactive decay changes the nature and identity of an atom’s nucleus. This occurs for a specific reason. Elements from hydrogen to lead (atomic numbers 1-82) have stable isotopes in which the tendency of protons to repel one another ...
... Radioactivity is the spontaneous emission of radiation by nuclei. Radioactive decay changes the nature and identity of an atom’s nucleus. This occurs for a specific reason. Elements from hydrogen to lead (atomic numbers 1-82) have stable isotopes in which the tendency of protons to repel one another ...
Nuclear Decay
... Gamma Decay - after a nuclear reaction such as alpha or beta decay has occurred, the daughter nucleus is in high-energy, or excited state. As a result the nucleus spontaneously releases energy in the form of a gamma ray to return to a lower more stable energy state. gamma ray - a highly energetic fo ...
... Gamma Decay - after a nuclear reaction such as alpha or beta decay has occurred, the daughter nucleus is in high-energy, or excited state. As a result the nucleus spontaneously releases energy in the form of a gamma ray to return to a lower more stable energy state. gamma ray - a highly energetic fo ...
Nuclear - PEO Scarborough Chapter
... 1. Positive - release of positively charged particle called positron and neutrino 2. Negative - release of negatively charged particle called electron and antineutrino. For example, carbon-14 (an unstable isotope of carbon), when subjected to beta decay, forms nitrogen (stable) along with an electro ...
... 1. Positive - release of positively charged particle called positron and neutrino 2. Negative - release of negatively charged particle called electron and antineutrino. For example, carbon-14 (an unstable isotope of carbon), when subjected to beta decay, forms nitrogen (stable) along with an electro ...
NUCLEAR CHEMISTRY
... 1. Gamma rays () are high-energy electromagnetic waves emitted from a nucleus as it changes from an excited state to a ground energy state 2. Gamma emission usually follows other types of decay that leave the nucleus in an excited state ...
... 1. Gamma rays () are high-energy electromagnetic waves emitted from a nucleus as it changes from an excited state to a ground energy state 2. Gamma emission usually follows other types of decay that leave the nucleus in an excited state ...
radioactive decay - Aurora City Schools
... • An element with a different number of neutrons • Because has same number of protons, still that element and has all chem/phys properties • Write isotopes using atomic # & mass # ...
... • An element with a different number of neutrons • Because has same number of protons, still that element and has all chem/phys properties • Write isotopes using atomic # & mass # ...
isotope - Aurora City Schools
... • An element with a different number of neutrons • Because has same number of protons, still that element and has all chem/phys properties • Write isotopes using atomic # & mass # ...
... • An element with a different number of neutrons • Because has same number of protons, still that element and has all chem/phys properties • Write isotopes using atomic # & mass # ...
Isotopes and Radioactive Decay
... GAMMA RADIATION: Radiation that is made up of ________________________ rays. A gamma ray is high-energy and contains no _____________ and is represented by the symbol __________. Gamma rays usually accompany ___________________ and ___________________ radiation. Gamma rays also account for _________ ...
... GAMMA RADIATION: Radiation that is made up of ________________________ rays. A gamma ray is high-energy and contains no _____________ and is represented by the symbol __________. Gamma rays usually accompany ___________________ and ___________________ radiation. Gamma rays also account for _________ ...
Ernest Rutherford Essay Research Paper Rutherford was
... can be stopped by a single sheet of paper. Alpha particles electrically charge molecules in the air through which they travel. Beta particles consist of streams of electrons traveling at very high speeds, often approaching the speed of light. They have a mass of 0.000 55 atomic mass unit and a charg ...
... can be stopped by a single sheet of paper. Alpha particles electrically charge molecules in the air through which they travel. Beta particles consist of streams of electrons traveling at very high speeds, often approaching the speed of light. They have a mass of 0.000 55 atomic mass unit and a charg ...
Review and Radioactivity
... reduced by 2 and its mass number reduced by 4 (that is, by 2 protons and 2 neutrons). ...
... reduced by 2 and its mass number reduced by 4 (that is, by 2 protons and 2 neutrons). ...
Chapter 25
... 1. What causes a transmutation of the nucleus to occur? 2. How are nuclear decay reaction equations balanced? 3. Do all radionuclides decay at the same rate? ...
... 1. What causes a transmutation of the nucleus to occur? 2. How are nuclear decay reaction equations balanced? 3. Do all radionuclides decay at the same rate? ...
Exit Slip: Atomic Structure and Nuclear Chemistry-1
... A. both negatively charged and repel each other C. both positively charged and repel each other B. oppositely charged and attract each other D. oppositely charged and repel each other 5. Most atomic nuclei are stable, even though they contain positively charged protons that repel each other. Which f ...
... A. both negatively charged and repel each other C. both positively charged and repel each other B. oppositely charged and attract each other D. oppositely charged and repel each other 5. Most atomic nuclei are stable, even though they contain positively charged protons that repel each other. Which f ...
radioisotopes and radiotherapy - video
... 1. Isotopes are nuclides with the same number of protons but different number of neutrons. Isotopes have same atomic number but different mass number. 2. Radioisotopes are unstable isotopes, where their nuclei decay to a more stable state or type of nuclei, giving off matter and energy as radiation. ...
... 1. Isotopes are nuclides with the same number of protons but different number of neutrons. Isotopes have same atomic number but different mass number. 2. Radioisotopes are unstable isotopes, where their nuclei decay to a more stable state or type of nuclei, giving off matter and energy as radiation. ...
Untitled
... Cosmic rays are streams of electrically charged particles. Collisions between cosmic rays And Earth’s atmosphere shower the surface below with nuclear radiation. ...
... Cosmic rays are streams of electrically charged particles. Collisions between cosmic rays And Earth’s atmosphere shower the surface below with nuclear radiation. ...
Stable Vs Unstable Isotopes
... Which of the properties of radioisotopes make them useful as tracers in medical or agricultural applications? I. Their chemical behavior is the same as nonradioactive isotopes. II. They emit various types of radiation. III. The nuclear reaction is unaffected by the chemical state of the isotope. ...
... Which of the properties of radioisotopes make them useful as tracers in medical or agricultural applications? I. Their chemical behavior is the same as nonradioactive isotopes. II. They emit various types of radiation. III. The nuclear reaction is unaffected by the chemical state of the isotope. ...
Radioactivity
... Beta Particles • Symbol is Greek letter, beta (β) • High speed negatively charged particles that come from the nucleus – hmmm… • Neutron (neutral) actually decays to form a p+ and an e-, e- is ejected from nucleus • Travel farther than α, but can be stopped by 3 mm of Al, or 10 mm of wood • Can cau ...
... Beta Particles • Symbol is Greek letter, beta (β) • High speed negatively charged particles that come from the nucleus – hmmm… • Neutron (neutral) actually decays to form a p+ and an e-, e- is ejected from nucleus • Travel farther than α, but can be stopped by 3 mm of Al, or 10 mm of wood • Can cau ...
Gamma ray
Gamma radiation, also known as gamma rays, and denoted by the Greek letter γ, refers to electromagnetic radiation of an extremely high frequency and therefore consists of high-energy photons. Gamma rays are ionizing radiation, and are thus biologically hazardous. They are classically produced by the decay of atomic nuclei as they transition from a high energy state to a lower state known as gamma decay, but may also be produced by other processes. Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900, while studying radiation emitted from radium. Villard's radiation was named ""gamma rays"" by Ernest Rutherford in 1903.Natural sources of gamma rays on Earth include gamma decay from naturally occurring radioisotopes, and secondary radiation from atmospheric interactions with cosmic ray particles. Rare terrestrial natural sources produce gamma rays that are not of a nuclear origin, such as lightning strikes and terrestrial gamma-ray flashes. Additionally, gamma rays are produced by a number of astronomical processes in which very high-energy electrons are produced, that in turn cause secondary gamma rays via bremsstrahlung, inverse Compton scattering, and synchrotron radiation. However, a large fraction of such astronomical gamma rays are screened by Earth's atmosphere and can only be detected by spacecraft.Gamma rays typically have frequencies above 10 exahertz (or >1019 Hz), and therefore have energies above 100 keV and wavelengths less than 10 picometers (10−12 meter), which is less than the diameter of an atom. However, this is not a hard and fast definition, but rather only a rule-of-thumb description for natural processes. Electromagnetic radiation from radioactive decay of atomic nuclei is referred to as ""gamma rays"" no matter its energy, so that there is no lower limit to gamma energy derived from radioactive decay. This radiation commonly has energy of a few hundred keV, and almost always less than 10 MeV. In astronomy, gamma rays are defined by their energy, and no production process needs to be specified. The energies of gamma rays from astronomical sources range to over 10 TeV, an energy far too large to result from radioactive decay. A notable example is extremely powerful bursts of high-energy radiation referred to as long duration gamma-ray bursts, of energies higher than can be produced by radioactive decay. These bursts of gamma rays, thought to be due to the collapse of stars called hypernovae, are the most powerful events so far discovered in the cosmos.