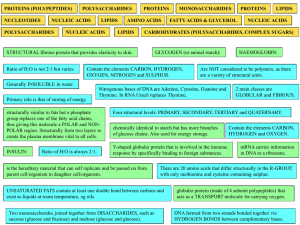
Organic Compounds
... Amino acids are the building blocks of proteins. There are 20 essential amino acids. All amino acids have the same Amino group and carboxyl groups, but each amino acid has its own unique R- group. Only 20 amino acids can combine in different arrangements to form all of the many different kinds of pr ...
... Amino acids are the building blocks of proteins. There are 20 essential amino acids. All amino acids have the same Amino group and carboxyl groups, but each amino acid has its own unique R- group. Only 20 amino acids can combine in different arrangements to form all of the many different kinds of pr ...
Organic Compounds PowerPoint PDF
... Amino acids are the building blocks of proteins. There are 20 essential amino acids. All amino acids have the same Amino group and carboxyl groups, but each amino acid has its own unique R- group. Only 20 amino acids can combine in different arrangements to form all of the many different kinds of pr ...
... Amino acids are the building blocks of proteins. There are 20 essential amino acids. All amino acids have the same Amino group and carboxyl groups, but each amino acid has its own unique R- group. Only 20 amino acids can combine in different arrangements to form all of the many different kinds of pr ...
Biochemistry
... Describe the structure and function of organic molecules Demonstrate how small molecules are joined together to make larger molecules ...
... Describe the structure and function of organic molecules Demonstrate how small molecules are joined together to make larger molecules ...
Make It – Break It
... From the indicated starting compound(s) use metabolic pathways to make one molecule of the indicated compound. For this assignment portion, you can assume that you have all ATP or NAD(P)H and non-organic substrates (e.g. ammonia) needed for biosynthetic reactions. Diagram the pathways involved, clea ...
... From the indicated starting compound(s) use metabolic pathways to make one molecule of the indicated compound. For this assignment portion, you can assume that you have all ATP or NAD(P)H and non-organic substrates (e.g. ammonia) needed for biosynthetic reactions. Diagram the pathways involved, clea ...
No Slide Title
... All cells must make their own ATP from nutrients they have either synthesized (autotrophs) or consumed ...
... All cells must make their own ATP from nutrients they have either synthesized (autotrophs) or consumed ...
43) What are the membrane structures that function in active
... E) transport. 45) Which oI the following statements is correcfabout diJfusion? A) It is very rapid over long distances. B) It requires an exPenditure of energy by the cell. of lower C) It is a passive processin which molecules move from a region of higher concentration to a region concentration. D) ...
... E) transport. 45) Which oI the following statements is correcfabout diJfusion? A) It is very rapid over long distances. B) It requires an exPenditure of energy by the cell. of lower C) It is a passive processin which molecules move from a region of higher concentration to a region concentration. D) ...
ppt2 - NMSU Astronomy
... key” basis, it would be expected that this phenomenon would be present in life elsewhere. ...
... key” basis, it would be expected that this phenomenon would be present in life elsewhere. ...
Organic Compounds
... • There are two types of proteins – fibrous and globular. • Fibrous protein (found in skin, tendons, bones, and muscles) does not dissolve in water (hydrophobic). • Globular protein (found in enzymes, some hormones, and hemoglobin) can dissolve in water (hydrophilic). ...
... • There are two types of proteins – fibrous and globular. • Fibrous protein (found in skin, tendons, bones, and muscles) does not dissolve in water (hydrophobic). • Globular protein (found in enzymes, some hormones, and hemoglobin) can dissolve in water (hydrophilic). ...
Biology
... • Found in all proteins and nucleic acids. • Major nonliving source is N2 in the atmosphere. • Makes its way into the food chain via nitrogen fixing bacteria, which convert it into a usable form of N2 that can be used by producers and passed on to consumers in the food chain. • Returned back to the ...
... • Found in all proteins and nucleic acids. • Major nonliving source is N2 in the atmosphere. • Makes its way into the food chain via nitrogen fixing bacteria, which convert it into a usable form of N2 that can be used by producers and passed on to consumers in the food chain. • Returned back to the ...
Exam I Review - Iowa State University
... d. direct synthesis of ATP by the tricarboxylic acid cycle. 161. Which of the following statements about NAD+ is FALSE? a. NAD+ is reduced to NADH during both glycolysis and the citric acid cycle. b. NAD+ has more chemical potential energy than NADH. c. NAD+ can receive electrons for use in electron ...
... d. direct synthesis of ATP by the tricarboxylic acid cycle. 161. Which of the following statements about NAD+ is FALSE? a. NAD+ is reduced to NADH during both glycolysis and the citric acid cycle. b. NAD+ has more chemical potential energy than NADH. c. NAD+ can receive electrons for use in electron ...
Macromolecules 2016
... Water is released and energy is stored in the newly formed chemical bonds. • 4. Hydrolysis: A chemical process where a large molecule is broken down into smaller molecules. Water is required and energy is released. Digestion is a series of hydrolytic ...
... Water is released and energy is stored in the newly formed chemical bonds. • 4. Hydrolysis: A chemical process where a large molecule is broken down into smaller molecules. Water is required and energy is released. Digestion is a series of hydrolytic ...
Macromolecules biologyjunction
... outer electrons and can form four bonds. Carbon can form single bonds with another atom and also bond to other carbon molecules forming double, triple, or quadruple bonds. Organic compounds also contain hydrogen. Since hydrogen has only one electron, it can form only single bonds. Each small organic ...
... outer electrons and can form four bonds. Carbon can form single bonds with another atom and also bond to other carbon molecules forming double, triple, or quadruple bonds. Organic compounds also contain hydrogen. Since hydrogen has only one electron, it can form only single bonds. Each small organic ...
Biochemistry Notes
... 1. Tetravalent - can form four bonds with other elements 2. May form double bonds and triple bonds. 3. Bond with itself forming Chains of various lengths 4. The chains may branch. 5. May form rings ...
... 1. Tetravalent - can form four bonds with other elements 2. May form double bonds and triple bonds. 3. Bond with itself forming Chains of various lengths 4. The chains may branch. 5. May form rings ...
1) Where does glycolysis occur in the cell
... 25) During the synthesis of proteins, ribosomes _____________. a) open DNA so it can be transcribed b) transcribe a sequence of mRNA c) translate a sequence of mRNA into a protein d) transport a particular amino acid during translation e) help proteins to fold after they are made. ...
... 25) During the synthesis of proteins, ribosomes _____________. a) open DNA so it can be transcribed b) transcribe a sequence of mRNA c) translate a sequence of mRNA into a protein d) transport a particular amino acid during translation e) help proteins to fold after they are made. ...
Review Sheet for Lecture Exam 2 Chapter Five Structure and
... Glycosidic linkage. Dehydration reactions, Hydrolysis reactions 2. Structure and function of Lipids (Fats, Phospholipids, Steroids). Ester linkage, saturated and unsaturated fats 3. Structure and function of proteins. Levels of protein structure (primary, secondary, tertiary and quaternary) Peptide ...
... Glycosidic linkage. Dehydration reactions, Hydrolysis reactions 2. Structure and function of Lipids (Fats, Phospholipids, Steroids). Ester linkage, saturated and unsaturated fats 3. Structure and function of proteins. Levels of protein structure (primary, secondary, tertiary and quaternary) Peptide ...
Chapter 2: Major Metabolic Pathway
... •Organisms are divided into autotrophs and heterotrophs according to their energy pathways. •Autotrophs are those organisms that are able to make energy-containing organic molecules from inorganic raw material by using basic energy sources such as sunlight. Plants are the prime example of autotrophs ...
... •Organisms are divided into autotrophs and heterotrophs according to their energy pathways. •Autotrophs are those organisms that are able to make energy-containing organic molecules from inorganic raw material by using basic energy sources such as sunlight. Plants are the prime example of autotrophs ...
Major Metabolic Pathway
... heterotrophs according to their energy pathways. •Autotrophs are those organisms that are able to make energy-containing organic molecules from inorganic raw material by using basic energy sources such as sunlight. Plants are the prime example of autotrophs, using photosynthesis. •All other organism ...
... heterotrophs according to their energy pathways. •Autotrophs are those organisms that are able to make energy-containing organic molecules from inorganic raw material by using basic energy sources such as sunlight. Plants are the prime example of autotrophs, using photosynthesis. •All other organism ...
FERMENTATION: an anaerobic biological reaction process in which
... • In bacteria, the trp repressor protein inhibits the transcription of a suite of genes coding for enyzmes required for the synthesis of the amino acid tryptophan • In the absence of tryptophan, the recognition helices are not in the proper orientation to contact the promoter DNA; no repressor binds ...
... • In bacteria, the trp repressor protein inhibits the transcription of a suite of genes coding for enyzmes required for the synthesis of the amino acid tryptophan • In the absence of tryptophan, the recognition helices are not in the proper orientation to contact the promoter DNA; no repressor binds ...
Cellular Respiration
... • The mitochondria are the engines of our cells where sugar is burned for fuel and the exhaust is CO2 and H2O. ...
... • The mitochondria are the engines of our cells where sugar is burned for fuel and the exhaust is CO2 and H2O. ...
Topic 2.4 Proteins Study Guide Amino acids are linked together by
... Amino acids are linked together by condensation to form polypeptides. There are 20 different amino acids in polypeptides synthesized on ribosomes. Amino acids can be linked together in any sequence giving a huge range of possible polypeptides. The amino acid sequence of polypeptides is coded for by ...
... Amino acids are linked together by condensation to form polypeptides. There are 20 different amino acids in polypeptides synthesized on ribosomes. Amino acids can be linked together in any sequence giving a huge range of possible polypeptides. The amino acid sequence of polypeptides is coded for by ...
biol 3 biomolecules table activity
... simplest is CHOLESTEROL that consists of a 4 ringed carbon structure and has a structural role in the plasma membrane. Other steroids act as HORMONES, eg testosterone and progesterone. SATURATED FATS contain no double bonds between carbons and exist as solids at room temperature. ...
... simplest is CHOLESTEROL that consists of a 4 ringed carbon structure and has a structural role in the plasma membrane. Other steroids act as HORMONES, eg testosterone and progesterone. SATURATED FATS contain no double bonds between carbons and exist as solids at room temperature. ...
Metabolism

Metabolism (from Greek: μεταβολή metabolē, ""change"") is the set of life-sustaining chemical transformations within the cells of living organisms. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to all chemical reactions that occur in living organisms, including digestion and the transport of substances into and between different cells, in which case the set of reactions within the cells is called intermediary metabolism or intermediate metabolism.Metabolism is usually divided into two categories: catabolism, the breaking down of organic matter by way of cellular respiration, and anabolism, the building up of components of cells such as proteins and nucleic acids. Usually, breaking down releases energy and building up consumes energy.The chemical reactions of metabolism are organized into metabolic pathways, in which one chemical is transformed through a series of steps into another chemical, by a sequence of enzymes. Enzymes are crucial to metabolism because they allow organisms to drive desirable reactions that require energy that will not occur by themselves, by coupling them to spontaneous reactions that release energy. Enzymes act as catalysts that allow the reactions to proceed more rapidly. Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells.The metabolic system of a particular organism determines which substances it will find nutritious and which poisonous. For example, some prokaryotes use hydrogen sulfide as a nutrient, yet this gas is poisonous to animals. The speed of metabolism, the metabolic rate, influences how much food an organism will require, and also affects how it is able to obtain that food.A striking feature of metabolism is the similarity of the basic metabolic pathways and components between even vastly different species. For example, the set of carboxylic acids that are best known as the intermediates in the citric acid cycle are present in all known organisms, being found in species as diverse as the unicellular bacterium Escherichia coli and huge multicellular organisms like elephants. These striking similarities in metabolic pathways are likely due to their early appearance in evolutionary history, and their retention because of their efficacy.