Microbial Metabolism Notes
... Microbial Metabolism A. Metabolism 1. Anabolism A) synthesis reactions (building up) 2. Catabolism A) lysis reactions (breaking down) B. Enzymes 1. proteins that facilitate chemical reactions A) reactant (substrate) binds to a specific binding site (active site) on the enzyme resulting in a lowering ...
... Microbial Metabolism A. Metabolism 1. Anabolism A) synthesis reactions (building up) 2. Catabolism A) lysis reactions (breaking down) B. Enzymes 1. proteins that facilitate chemical reactions A) reactant (substrate) binds to a specific binding site (active site) on the enzyme resulting in a lowering ...
Building Monomers of Macromolecules
... Atoms join together to form molecules. These molecules are held together by bonds. In this lab you will use toothpicks to represent the bonds. Important note: use one toothpick to represent a single covalent bond, and two toothpicks to represent a double covalent bond. Remember, covalent bonds are b ...
... Atoms join together to form molecules. These molecules are held together by bonds. In this lab you will use toothpicks to represent the bonds. Important note: use one toothpick to represent a single covalent bond, and two toothpicks to represent a double covalent bond. Remember, covalent bonds are b ...
Study Guide-Carbon, monomers, polymers, amino acids, proteins
... -Why do cells need to synthesize biomolecules? -Understand the different type of molecular structures you can create with carbon-ring, branched, chains, double bonded, triple bonded? -Where do organisms obtain their carbon to build biomolecules and what process builds organic or biomolecules? -What ...
... -Why do cells need to synthesize biomolecules? -Understand the different type of molecular structures you can create with carbon-ring, branched, chains, double bonded, triple bonded? -Where do organisms obtain their carbon to build biomolecules and what process builds organic or biomolecules? -What ...
Name
... Answer in Note book 7.1 Overview of cell respiration 1) Contrast autotrophs with heterotrophs 2) Define cellular respiration 3) Define digestion 4) Define dehydrogenations 5) What are redox reactions? Why are they important in biological systems? 6) What is NADH? How does NAD+ become NADH? 7) Define ...
... Answer in Note book 7.1 Overview of cell respiration 1) Contrast autotrophs with heterotrophs 2) Define cellular respiration 3) Define digestion 4) Define dehydrogenations 5) What are redox reactions? Why are they important in biological systems? 6) What is NADH? How does NAD+ become NADH? 7) Define ...
Metabolism
... • H+ concentration gradient utilized to produce ATP (chemiosmosis) – 32-34 ATP produced per glucose molecule Glucose Anabolism • Glycogenesis - glucose glycogen – Takes place in liver & stimulated by insulin – Body can store about 500 grams of glycogen (25% liver, 75% muscle) – Glycogenolysis - ca ...
... • H+ concentration gradient utilized to produce ATP (chemiosmosis) – 32-34 ATP produced per glucose molecule Glucose Anabolism • Glycogenesis - glucose glycogen – Takes place in liver & stimulated by insulin – Body can store about 500 grams of glycogen (25% liver, 75% muscle) – Glycogenolysis - ca ...
The stuff of life
... hot H-burning to make intermediate elements. E.g. N is made by adding two protons to a C (12C + p -> 13C -> 13N + e- ; 13N + p -> 14N). ...
... hot H-burning to make intermediate elements. E.g. N is made by adding two protons to a C (12C + p -> 13C -> 13N + e- ; 13N + p -> 14N). ...
2_1 Slides
... is common component, but it is not present in all proteins) • Proteins are large organic compounds made of amino acids arranged into one or more linear chains that then fold into morecomplex 3-D structures… ...
... is common component, but it is not present in all proteins) • Proteins are large organic compounds made of amino acids arranged into one or more linear chains that then fold into morecomplex 3-D structures… ...
Ch 6 Metabolism: Fueling Cell Growth
... Compare and contrast aerobic and anaerobic respiration. Describe the chemical reactions and some products of fermentation. Categorize the various nutritional patterns among organisms according to energy and carbon source. ...
... Compare and contrast aerobic and anaerobic respiration. Describe the chemical reactions and some products of fermentation. Categorize the various nutritional patterns among organisms according to energy and carbon source. ...
Ch 6 Metabolism: Fueling Cell Growth
... Compare and contrast aerobic and anaerobic respiration. Describe the chemical reactions and some products of fermentation. Categorize the various nutritional patterns among organisms according to energy and carbon source. ...
... Compare and contrast aerobic and anaerobic respiration. Describe the chemical reactions and some products of fermentation. Categorize the various nutritional patterns among organisms according to energy and carbon source. ...
Test Review Unit 1
... 8) What is metabolism (metabolic activity)? 9) What is homeostasis? Explain how the human body maintains homeostasis (one example). 10) What is asexual reproduction? What kind of offspring does it produce (compared to the parent)? 11) What is sexual reproduction? What kind of offspring does it prod ...
... 8) What is metabolism (metabolic activity)? 9) What is homeostasis? Explain how the human body maintains homeostasis (one example). 10) What is asexual reproduction? What kind of offspring does it produce (compared to the parent)? 11) What is sexual reproduction? What kind of offspring does it prod ...
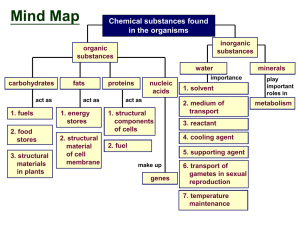
Biological Chemistry - Nutley Public Schools
... What are the 4 classes of organic compounds? What 3 elements are carbohydrates made up of? What carbohydrate is considered a universal source ...
... What are the 4 classes of organic compounds? What 3 elements are carbohydrates made up of? What carbohydrate is considered a universal source ...
BIOLOGY * CHAPTER 2
... What are the 4 classes of organic compounds? What 3 elements are carbohydrates made up of? What carbohydrate is considered a universal source ...
... What are the 4 classes of organic compounds? What 3 elements are carbohydrates made up of? What carbohydrate is considered a universal source ...
CH 2 -CH 2 -CH 2 -CH 2 -CH 2
... • Catalyst : inorganic or organic substance which speeds up the rate of a chemical reaction without entering the reaction itself • enzymes: organic catalysts made of protein • most enzyme names end in -ase • enzymes lower the energy needed to start a chemical reaction. (activation energy) • begin to ...
... • Catalyst : inorganic or organic substance which speeds up the rate of a chemical reaction without entering the reaction itself • enzymes: organic catalysts made of protein • most enzyme names end in -ase • enzymes lower the energy needed to start a chemical reaction. (activation energy) • begin to ...
NAME: : :______ Honors Biology Reading Guide – Chapter 6
... 69. One mole of glucose stores _____kcal of energy which is then utilized by living things when carbohydrates are oxidized in ______________________________________________. Mitochondria and Cellular Respiration 70. Write the overall equation for cellular respiration. ...
... 69. One mole of glucose stores _____kcal of energy which is then utilized by living things when carbohydrates are oxidized in ______________________________________________. Mitochondria and Cellular Respiration 70. Write the overall equation for cellular respiration. ...
BIO 219 Spring 2013 Outline for “Cell Metabolism” Energy (ATP
... Five integral membrane proteins NADH & FADH2 Oxygen (Why do we need it?) Net yield Fermentation Glycolysis Lactic acid production What about fats and proteins as an energy source? Objectives: By the end of lecture today you should be able to . . . (1) Describe the similarities and differences betwee ...
... Five integral membrane proteins NADH & FADH2 Oxygen (Why do we need it?) Net yield Fermentation Glycolysis Lactic acid production What about fats and proteins as an energy source? Objectives: By the end of lecture today you should be able to . . . (1) Describe the similarities and differences betwee ...
Solutions to problems
... own kind, growth and development in patterns determined by heredity, energy utilization to fuel activities, response to the environment or changes in surroundings, and evolutionary adaptation to environments. 5. Carbohydrates are sugars and starches that provide energy and structure to cells. Lipids ...
... own kind, growth and development in patterns determined by heredity, energy utilization to fuel activities, response to the environment or changes in surroundings, and evolutionary adaptation to environments. 5. Carbohydrates are sugars and starches that provide energy and structure to cells. Lipids ...
Slide 1
... Translates the genetic message in RNA into the production of protein. (It is the site of protein synthesis.) ...
... Translates the genetic message in RNA into the production of protein. (It is the site of protein synthesis.) ...
BIOL 1301 Module 3 - Metabolism – Learning Outcomes Chapters: 6
... List three stages of cellular respiration, location of each stage in the eukaryotic cell, and describe the sequence of compounds through which electrons flow. List two stages of photosynthesis, location of each stage in the chloroplast, and summarize the cooperation between the stages. Describe gene ...
... List three stages of cellular respiration, location of each stage in the eukaryotic cell, and describe the sequence of compounds through which electrons flow. List two stages of photosynthesis, location of each stage in the chloroplast, and summarize the cooperation between the stages. Describe gene ...
activity 2-2. organic chemistry
... of organic compound in cells. They are madeare upthe of most manyabundant amino acid bonded together. Proteins, which may be very large and complex, play a wide variety of roles in the cell. Some are structural, others are hormones, neurohumors, enzymes, or pigments. Amino acids are made up of carbo ...
... of organic compound in cells. They are madeare upthe of most manyabundant amino acid bonded together. Proteins, which may be very large and complex, play a wide variety of roles in the cell. Some are structural, others are hormones, neurohumors, enzymes, or pigments. Amino acids are made up of carbo ...
Cellular Respiration
... – In inner mitochondrial membrane – Electrons are delivered by NADH – Electrons move down chain of proteins – H+ build up in mitochondrial intermembrane space due to movement of electrons ATP synthase is powered by H+ movement across membrane 26 ATP are produced ½ O2 + 2 H+ H2O {oxygen is fina ...
... – In inner mitochondrial membrane – Electrons are delivered by NADH – Electrons move down chain of proteins – H+ build up in mitochondrial intermembrane space due to movement of electrons ATP synthase is powered by H+ movement across membrane 26 ATP are produced ½ O2 + 2 H+ H2O {oxygen is fina ...
Metabolism

Metabolism (from Greek: μεταβολή metabolē, ""change"") is the set of life-sustaining chemical transformations within the cells of living organisms. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to all chemical reactions that occur in living organisms, including digestion and the transport of substances into and between different cells, in which case the set of reactions within the cells is called intermediary metabolism or intermediate metabolism.Metabolism is usually divided into two categories: catabolism, the breaking down of organic matter by way of cellular respiration, and anabolism, the building up of components of cells such as proteins and nucleic acids. Usually, breaking down releases energy and building up consumes energy.The chemical reactions of metabolism are organized into metabolic pathways, in which one chemical is transformed through a series of steps into another chemical, by a sequence of enzymes. Enzymes are crucial to metabolism because they allow organisms to drive desirable reactions that require energy that will not occur by themselves, by coupling them to spontaneous reactions that release energy. Enzymes act as catalysts that allow the reactions to proceed more rapidly. Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells.The metabolic system of a particular organism determines which substances it will find nutritious and which poisonous. For example, some prokaryotes use hydrogen sulfide as a nutrient, yet this gas is poisonous to animals. The speed of metabolism, the metabolic rate, influences how much food an organism will require, and also affects how it is able to obtain that food.A striking feature of metabolism is the similarity of the basic metabolic pathways and components between even vastly different species. For example, the set of carboxylic acids that are best known as the intermediates in the citric acid cycle are present in all known organisms, being found in species as diverse as the unicellular bacterium Escherichia coli and huge multicellular organisms like elephants. These striking similarities in metabolic pathways are likely due to their early appearance in evolutionary history, and their retention because of their efficacy.