UNIT 2 BIOLOGICAL CHEMISTRY ORGANIC MOLECULES
... Co-factors: Inorganic ions that bind to the enzyme to help catalyze the reaction Co-enzymes: Organic molecules (not proteins) -generally are bound at the active site and the enzymatic rxn will not work w/o it. Coenzymes are found in small con. Because they are reusable. (vitamins, nucleotides, NAD) ...
... Co-factors: Inorganic ions that bind to the enzyme to help catalyze the reaction Co-enzymes: Organic molecules (not proteins) -generally are bound at the active site and the enzymatic rxn will not work w/o it. Coenzymes are found in small con. Because they are reusable. (vitamins, nucleotides, NAD) ...
RNA and Protein Synthesis
... 20. Describe the structure of proteins. A series of amino acids bonded together with peptide bonds. Enzymes 21. What organic compound are enzymes composed of?__proteins__How are they typically named (think word parts)? They typically end in “ase” 22. What happens to enzymes during/after a chemical r ...
... 20. Describe the structure of proteins. A series of amino acids bonded together with peptide bonds. Enzymes 21. What organic compound are enzymes composed of?__proteins__How are they typically named (think word parts)? They typically end in “ase” 22. What happens to enzymes during/after a chemical r ...
Unit1The Concept of Life - Sewanhaka Central High School District
... A generalized definition of life is the following: “Life is a continuous series of inter-related chemical reactions that require a source of energy from the environment”. ...
... A generalized definition of life is the following: “Life is a continuous series of inter-related chemical reactions that require a source of energy from the environment”. ...
Exam 2 for Review - philipdarrenjones.com
... maximum number of ATP molecules that could be made through substrate-level phosphorylation (one time through)? A) 1 B) 2 C) 11 D) 12 E) 24 ...
... maximum number of ATP molecules that could be made through substrate-level phosphorylation (one time through)? A) 1 B) 2 C) 11 D) 12 E) 24 ...
Gen Bio Midterm Review SG KEY 2015
... D. Label the following cells as PLANT or ANIMAL. Then label their correct organelles. ...
... D. Label the following cells as PLANT or ANIMAL. Then label their correct organelles. ...
Macromolecules Quiz
... b: a polysaccharide that gives plants their strength; is also dietary fiber c: What type of carbohydrate is lactose? d: these are the monomers of carbohydrates e: The color of a positive benedicts test (test for monosacharides) is ___________. ...
... b: a polysaccharide that gives plants their strength; is also dietary fiber c: What type of carbohydrate is lactose? d: these are the monomers of carbohydrates e: The color of a positive benedicts test (test for monosacharides) is ___________. ...
Metabolic engineering Synthetic Biology
... Practice of optimizing genetic and regulatory processes within cells to increase the cells' production of a substance. Metabolic engineers commonly work to reduce cellular energy use (i.e, the energetic cost of cell reproduction or proliferation) and to reduce waste production. Direct deletion ...
... Practice of optimizing genetic and regulatory processes within cells to increase the cells' production of a substance. Metabolic engineers commonly work to reduce cellular energy use (i.e, the energetic cost of cell reproduction or proliferation) and to reduce waste production. Direct deletion ...
1st exam
... 1) Normally about 80% of the carbohydrate in the diet is digested and absorbed. 2)About 90 % of the digestion and absorption of food takes place in the small intestine. 3)Glucose & fructose are absorbed by secondary active transport 4) Glycolysis is the major pathway for utilizing of glucose and is ...
... 1) Normally about 80% of the carbohydrate in the diet is digested and absorbed. 2)About 90 % of the digestion and absorption of food takes place in the small intestine. 3)Glucose & fructose are absorbed by secondary active transport 4) Glycolysis is the major pathway for utilizing of glucose and is ...
Energy
... Exergonic and Endergonic reactions: ∆G = ∆H – T∆S Spontaneous reactions release free energy, which is available to do work. Exergonic reactions have A negative G value. Endergonic reactions are a nonspontaneous reaction or process that absorbs free energy and has a positive ∆G. ...
... Exergonic and Endergonic reactions: ∆G = ∆H – T∆S Spontaneous reactions release free energy, which is available to do work. Exergonic reactions have A negative G value. Endergonic reactions are a nonspontaneous reaction or process that absorbs free energy and has a positive ∆G. ...
AP Biology: Chapter 9
... 7. What is the role of NAD+ & FAD+2 in respiration? 8. Explain why respiration is considered exergonic. 9. Glycolysis starts with _____________________ and produces 10. The Kreb’s cycle takes place in the: 11. Pyruvate is converted to ___________________________________ before the Krebs cycle. 12. T ...
... 7. What is the role of NAD+ & FAD+2 in respiration? 8. Explain why respiration is considered exergonic. 9. Glycolysis starts with _____________________ and produces 10. The Kreb’s cycle takes place in the: 11. Pyruvate is converted to ___________________________________ before the Krebs cycle. 12. T ...
Bio150 Practice Exam 2 Name
... The light-dependent reactions produce water as a by-product; the light-independent reactions produce carbon dioxide as a waste product. e. The products of the light-dependent reactions are ADP, NADP+, and O2; the products of the light-independent reactions are ATP, NADPH, and sugar. Bonus Questions: ...
... The light-dependent reactions produce water as a by-product; the light-independent reactions produce carbon dioxide as a waste product. e. The products of the light-dependent reactions are ADP, NADP+, and O2; the products of the light-independent reactions are ATP, NADPH, and sugar. Bonus Questions: ...
Subject name
... 10. Biological catalysis – enzyme structure, classes, substrate specificity, optima, structural and kinetic models of enzymatic action; regulatory mechanisms. Fundamentals of cellular metabolism regulation and bioenergetics - main anabolic and catabolic pathways and cycles and their localization wit ...
... 10. Biological catalysis – enzyme structure, classes, substrate specificity, optima, structural and kinetic models of enzymatic action; regulatory mechanisms. Fundamentals of cellular metabolism regulation and bioenergetics - main anabolic and catabolic pathways and cycles and their localization wit ...
Bio101 Chapters 5, 6, 7 Practice Quiz Name
... A) Potential energy; kinetic energy B) Kinetic energy; potential energy 9. ATP contains A) one phosphate group B) two phosphate groups C) three phosphate groups D) four phosphate groups 10. Most of a cell's enzymes are A) lipids. B) proteins. C) amino acids. D) nucleic acids. E) carbohydrates. 11. I ...
... A) Potential energy; kinetic energy B) Kinetic energy; potential energy 9. ATP contains A) one phosphate group B) two phosphate groups C) three phosphate groups D) four phosphate groups 10. Most of a cell's enzymes are A) lipids. B) proteins. C) amino acids. D) nucleic acids. E) carbohydrates. 11. I ...
STUDY GUIDE FOR CELLULAR RESPIRATION Cellular
... c. Hydrogens diffuse across the membrane back to the inside via a carrier protein that ads a PHOSPHATE group to ADP d. ADP + PO4 = ATP e. At the end of the chain spent electrons, Hydrogen ions and O2 combine to form H20. RESPIRATION IN THE ABSENCE OF OXYGEN ...
... c. Hydrogens diffuse across the membrane back to the inside via a carrier protein that ads a PHOSPHATE group to ADP d. ADP + PO4 = ATP e. At the end of the chain spent electrons, Hydrogen ions and O2 combine to form H20. RESPIRATION IN THE ABSENCE OF OXYGEN ...
Molecules of life
... Carbon has 4 unpaired electrons, makes 4 covalent bonds Hydrocarbon chain carbon-hydrogen chains ...
... Carbon has 4 unpaired electrons, makes 4 covalent bonds Hydrocarbon chain carbon-hydrogen chains ...
Biochemistry Notes
... positive (+) charge, in nucleus b. neutrons – mass 1 amu, no charge (neutral), in nucleus c. electrons – no mass, negative charge (-), orbits the nucleus in the electron cloud, involved in bonding 3. element – a substance of one type of atom, can not be broken down in to simpler substances 4. 25 ele ...
... positive (+) charge, in nucleus b. neutrons – mass 1 amu, no charge (neutral), in nucleus c. electrons – no mass, negative charge (-), orbits the nucleus in the electron cloud, involved in bonding 3. element – a substance of one type of atom, can not be broken down in to simpler substances 4. 25 ele ...
1st semester exam study guide
... Microscopes are used for magnification Properties of life: heredity, Response/stimulus, adaptation, metabolism, reproduce, grow and develop Homeostasis ...
... Microscopes are used for magnification Properties of life: heredity, Response/stimulus, adaptation, metabolism, reproduce, grow and develop Homeostasis ...
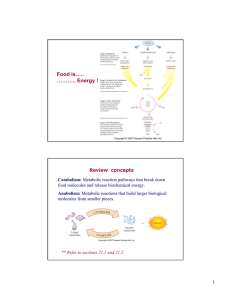
Metabolism

Metabolism (from Greek: μεταβολή metabolē, ""change"") is the set of life-sustaining chemical transformations within the cells of living organisms. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to all chemical reactions that occur in living organisms, including digestion and the transport of substances into and between different cells, in which case the set of reactions within the cells is called intermediary metabolism or intermediate metabolism.Metabolism is usually divided into two categories: catabolism, the breaking down of organic matter by way of cellular respiration, and anabolism, the building up of components of cells such as proteins and nucleic acids. Usually, breaking down releases energy and building up consumes energy.The chemical reactions of metabolism are organized into metabolic pathways, in which one chemical is transformed through a series of steps into another chemical, by a sequence of enzymes. Enzymes are crucial to metabolism because they allow organisms to drive desirable reactions that require energy that will not occur by themselves, by coupling them to spontaneous reactions that release energy. Enzymes act as catalysts that allow the reactions to proceed more rapidly. Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells.The metabolic system of a particular organism determines which substances it will find nutritious and which poisonous. For example, some prokaryotes use hydrogen sulfide as a nutrient, yet this gas is poisonous to animals. The speed of metabolism, the metabolic rate, influences how much food an organism will require, and also affects how it is able to obtain that food.A striking feature of metabolism is the similarity of the basic metabolic pathways and components between even vastly different species. For example, the set of carboxylic acids that are best known as the intermediates in the citric acid cycle are present in all known organisms, being found in species as diverse as the unicellular bacterium Escherichia coli and huge multicellular organisms like elephants. These striking similarities in metabolic pathways are likely due to their early appearance in evolutionary history, and their retention because of their efficacy.