Unit 05 - Lessons 1-4
... A. Nucleotides are made of a sugar, phosphate group, and a nitrogen base. A phosphate group ...
... A. Nucleotides are made of a sugar, phosphate group, and a nitrogen base. A phosphate group ...
Document
... 4. If an amino acid is glucogenic, it will not be degraded to ________. A) pyruvate B) glutamate C) fumarate D) acetoacetate 5. T/F It is possible for an amino acid to be both ketogenic and glucogenic. ...
... 4. If an amino acid is glucogenic, it will not be degraded to ________. A) pyruvate B) glutamate C) fumarate D) acetoacetate 5. T/F It is possible for an amino acid to be both ketogenic and glucogenic. ...
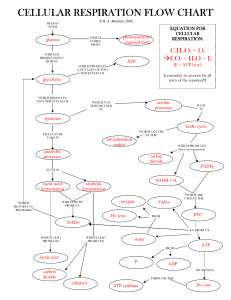
Carbohydrate Catabolism in the Presence of Oxygen Releases a
... Products: CO2 and acetate; acetate is then bound to coenzyme A (CoA) to form ...
... Products: CO2 and acetate; acetate is then bound to coenzyme A (CoA) to form ...
Ch. 7 Study Guide
... □ I can explain how glucose is oxidized during glycolysis and the Krebs Cycle to produce reducing power in NADH and FADH ...
... □ I can explain how glucose is oxidized during glycolysis and the Krebs Cycle to produce reducing power in NADH and FADH ...
Citric Acid Cycle in Anabolism
... 2) Uses NADP+ as a cofactor instead of NAD+. 3) Pyruvate carboxyl reduced to hydroxyl ...
... 2) Uses NADP+ as a cofactor instead of NAD+. 3) Pyruvate carboxyl reduced to hydroxyl ...
Extension worksheet – Option C - Cambridge Resources for the IB
... the active site to change so that the substrate cannot bind to it. Such inhibitors, if they bind reversibly, can act in end-product inhibition of metabolic reactions. End-product inhibition is an example of negative feedback. a ...
... the active site to change so that the substrate cannot bind to it. Such inhibitors, if they bind reversibly, can act in end-product inhibition of metabolic reactions. End-product inhibition is an example of negative feedback. a ...
biochemistry - Bioscience High School
... Fats and oils are triglycerides. Constructed of a 3 carbon alcohol called glycerol and three long chains of hydrocarbons called fatty acids. ...
... Fats and oils are triglycerides. Constructed of a 3 carbon alcohol called glycerol and three long chains of hydrocarbons called fatty acids. ...
Ch 4: Cellular Metabolism
... • The linkage carbon on the first sugar is always C1. So disaccharides can be named as sugar-(a,)1,#-sugar, where a or depends on the anomeric structure of the first sugar. For example, Maltose ...
... • The linkage carbon on the first sugar is always C1. So disaccharides can be named as sugar-(a,)1,#-sugar, where a or depends on the anomeric structure of the first sugar. For example, Maltose ...
Slide 1
... through many chemical processes Catabolism breaks food down into smaller molecular compounds and releases two forms of energy: heat and chemical Anabolism: a synthesis process Both processes take place inside cells continuously and concurrently ...
... through many chemical processes Catabolism breaks food down into smaller molecular compounds and releases two forms of energy: heat and chemical Anabolism: a synthesis process Both processes take place inside cells continuously and concurrently ...
BIOLOGY PRACTICE QUESTIONS GROUP II 1.To separate the
... (a) Oxygen is used in the dark reactions. (b) Carbon dioxide is released in the dark reactions. (c) Water is split in the light reactions. (d) Alcohol is produced by the light reactions. 40. During photosynthesis, molecules of oxygen are liberated from the "splitting" of water molecules due to the ( ...
... (a) Oxygen is used in the dark reactions. (b) Carbon dioxide is released in the dark reactions. (c) Water is split in the light reactions. (d) Alcohol is produced by the light reactions. 40. During photosynthesis, molecules of oxygen are liberated from the "splitting" of water molecules due to the ( ...
print last name first name
... (c)______________________. Halfway there, he catches his second wind and oxygen is resupplied to his muscles. Now pyruvate gets oxidized to (d)_________________________________ using the enzyme called pyruvate dehydrogenase complex. The substance from (d) enters the Krebs cycle and (e)_________ mole ...
... (c)______________________. Halfway there, he catches his second wind and oxygen is resupplied to his muscles. Now pyruvate gets oxidized to (d)_________________________________ using the enzyme called pyruvate dehydrogenase complex. The substance from (d) enters the Krebs cycle and (e)_________ mole ...
Proteins Multiple choice Proteins can be classified as Polyesters
... maltose. The pH of saliva is about 7, which is close to the optimum temperature of an enzyme. Amylase stops functioning when it enters the stomach which has a pH of 2. What happens to the enzyme on entering the stomach which would cause it to ...
... maltose. The pH of saliva is about 7, which is close to the optimum temperature of an enzyme. Amylase stops functioning when it enters the stomach which has a pH of 2. What happens to the enzyme on entering the stomach which would cause it to ...
Document
... Maintain maximum cell metabolism Carbohydrates Energy source Glucose and galactose Vitamins Precursors for numerous co-factors B group vitamins necessary for cell growth and proliferation Common vitamins found in basic media are riboflavin, thiamine and biotin Trace Elements Zinc, copp ...
... Maintain maximum cell metabolism Carbohydrates Energy source Glucose and galactose Vitamins Precursors for numerous co-factors B group vitamins necessary for cell growth and proliferation Common vitamins found in basic media are riboflavin, thiamine and biotin Trace Elements Zinc, copp ...
Biochem 462 - public.asu.edu
... I want you to determine the number of oxygen molecules (O2, not ½ O2) required for the complete oxidation of one 16 carbon fatty acid. Please do this in three steps (you need to explain your reasoning for full credit). If you cannot do one step, make an assumption and do the next one. a) Determine t ...
... I want you to determine the number of oxygen molecules (O2, not ½ O2) required for the complete oxidation of one 16 carbon fatty acid. Please do this in three steps (you need to explain your reasoning for full credit). If you cannot do one step, make an assumption and do the next one. a) Determine t ...
Oxidative phosphorylation (mitochondria)
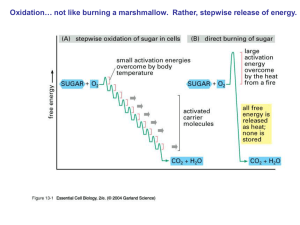
... Oxidation… not like burning a marshmallow. Rather, stepwise release of energy. ...
... Oxidation… not like burning a marshmallow. Rather, stepwise release of energy. ...
Fall 08 – BIOL 1000 – 1st lecture test – 12:30 Use the information
... C. all contain hydrogen D. all of the above 11. Lactose (a disaccharide) is formed when glucose and galactose (monosaccharides) are bonded together and water is given off. This process is an example of: A. hydrolysis B. polar bonding C. hydrogen bonding D. dehydration synthesis 12. In the scientific ...
... C. all contain hydrogen D. all of the above 11. Lactose (a disaccharide) is formed when glucose and galactose (monosaccharides) are bonded together and water is given off. This process is an example of: A. hydrolysis B. polar bonding C. hydrogen bonding D. dehydration synthesis 12. In the scientific ...
Chapter 5 Molecules of Life
... 3. Waxes – long-chain fatty acids linked to long chain alcohols – Ex: Wax coating on cherries, wax in human ears ...
... 3. Waxes – long-chain fatty acids linked to long chain alcohols – Ex: Wax coating on cherries, wax in human ears ...
Chapter 9 / Energy-Releasing Pathways and Biosynthesis I
... Differ in their final electron acceptor Cellular respiration produces more ATP Pyruvate is a key juncture in catabolism Glycolysis occurs in nearly all organisms ...
... Differ in their final electron acceptor Cellular respiration produces more ATP Pyruvate is a key juncture in catabolism Glycolysis occurs in nearly all organisms ...
Name
... r. Exergonic reaction - Energy is released s. Endergonic reaction - Energy is absobrbed 4. Which element is present in all ORGANIC molecules? Carbon 5. Polymers are formed by the joining together of monomers, in a reaction called dehydration where water is taken out to join two molecules. ...
... r. Exergonic reaction - Energy is released s. Endergonic reaction - Energy is absobrbed 4. Which element is present in all ORGANIC molecules? Carbon 5. Polymers are formed by the joining together of monomers, in a reaction called dehydration where water is taken out to join two molecules. ...
Metabolism

Metabolism (from Greek: μεταβολή metabolē, ""change"") is the set of life-sustaining chemical transformations within the cells of living organisms. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to all chemical reactions that occur in living organisms, including digestion and the transport of substances into and between different cells, in which case the set of reactions within the cells is called intermediary metabolism or intermediate metabolism.Metabolism is usually divided into two categories: catabolism, the breaking down of organic matter by way of cellular respiration, and anabolism, the building up of components of cells such as proteins and nucleic acids. Usually, breaking down releases energy and building up consumes energy.The chemical reactions of metabolism are organized into metabolic pathways, in which one chemical is transformed through a series of steps into another chemical, by a sequence of enzymes. Enzymes are crucial to metabolism because they allow organisms to drive desirable reactions that require energy that will not occur by themselves, by coupling them to spontaneous reactions that release energy. Enzymes act as catalysts that allow the reactions to proceed more rapidly. Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells.The metabolic system of a particular organism determines which substances it will find nutritious and which poisonous. For example, some prokaryotes use hydrogen sulfide as a nutrient, yet this gas is poisonous to animals. The speed of metabolism, the metabolic rate, influences how much food an organism will require, and also affects how it is able to obtain that food.A striking feature of metabolism is the similarity of the basic metabolic pathways and components between even vastly different species. For example, the set of carboxylic acids that are best known as the intermediates in the citric acid cycle are present in all known organisms, being found in species as diverse as the unicellular bacterium Escherichia coli and huge multicellular organisms like elephants. These striking similarities in metabolic pathways are likely due to their early appearance in evolutionary history, and their retention because of their efficacy.