Additional Chemistry
... •Acids are substances that donate Hydrogen ions (H+) •Acids can be both strong and weak. H2SO4 → 2H+ + SO42•Bases are solids or liquids that neutralise acids •They are able to accept Hydrogen ions (H+) •They can be Metal Hydroxide or Metal Oxides HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l) CuO(s) + 2HCl ...
... •Acids are substances that donate Hydrogen ions (H+) •Acids can be both strong and weak. H2SO4 → 2H+ + SO42•Bases are solids or liquids that neutralise acids •They are able to accept Hydrogen ions (H+) •They can be Metal Hydroxide or Metal Oxides HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l) CuO(s) + 2HCl ...
Ch 5.1 The Nature of Chemical Reactions
... Objectives For this Chapter • Understand parts to a chemical equation (reactants, products, yeild sign, double arrow) • Conservation of matter is expressed through balancing chemical equations • Describe difference between endothermic and exothermic reactions ...
... Objectives For this Chapter • Understand parts to a chemical equation (reactants, products, yeild sign, double arrow) • Conservation of matter is expressed through balancing chemical equations • Describe difference between endothermic and exothermic reactions ...
Atoms in Combination: The Chemical Bond
... The periodic table of the elements. The weights of the elements increase from left to right. Each vertical column groups elements with similar chemical properties. ...
... The periodic table of the elements. The weights of the elements increase from left to right. Each vertical column groups elements with similar chemical properties. ...
Balancing Single Replacement Reactions - Kossmann
... Name: __________________________ Class: ____________________ Date: _____________ ...
... Name: __________________________ Class: ____________________ Date: _____________ ...
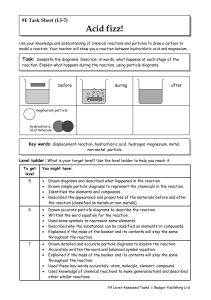
Year 9 Homework Task 9E-5 Reactions 5-7
... Drawn accurate particle diagrams to describe the reaction. Written the word equation for the reaction. Used some symbols to represent some elements. Described why the substances can be classified as elements or compounds. Explained if the mass of the beaker and its contents will stay the same throug ...
... Drawn accurate particle diagrams to describe the reaction. Written the word equation for the reaction. Used some symbols to represent some elements. Described why the substances can be classified as elements or compounds. Explained if the mass of the beaker and its contents will stay the same throug ...
Modelling Mass Transfer in Nitrification Processes Using
... oxidation products can be intercepted “kinetically”. • Metal oxides, especially transition metal oxides, form the basis of selective oxidation catalysts. • The catalyst performance in terms of activity and selectivity is strongly related to the lattice structure. • Most selective oxidation reactions ...
... oxidation products can be intercepted “kinetically”. • Metal oxides, especially transition metal oxides, form the basis of selective oxidation catalysts. • The catalyst performance in terms of activity and selectivity is strongly related to the lattice structure. • Most selective oxidation reactions ...
Ch. 2 The Chemistry of Life
... - Proteins – molecules that contain _____________, carbon, ____________, & ___________ - ____________ are made up of ________ of __________ __________ - Amino acids - ________________ with an __________ group on one end & a ___________ group on the other end, there are more than ____ in ___________ ...
... - Proteins – molecules that contain _____________, carbon, ____________, & ___________ - ____________ are made up of ________ of __________ __________ - Amino acids - ________________ with an __________ group on one end & a ___________ group on the other end, there are more than ____ in ___________ ...
PS7aChemistryReviewRevised
... they contain…not a new set of characteristics…they are relatively easy to separate. Compounds show new characteristics, not those of the substances they contain…they are generally harder to separate. ...
... they contain…not a new set of characteristics…they are relatively easy to separate. Compounds show new characteristics, not those of the substances they contain…they are generally harder to separate. ...
200 ways to pass the regents
... 81. Electronegativity is a measure of an element’s attraction for electrons. 82. Electronegativity increases as you go up and to the right on the Periodic Table. 83. The elements in Group 1 are the alkali metals. 84. The elements in Group 2 are the alkaline earth metals. 85. The elements in Group 17 ...
... 81. Electronegativity is a measure of an element’s attraction for electrons. 82. Electronegativity increases as you go up and to the right on the Periodic Table. 83. The elements in Group 1 are the alkali metals. 84. The elements in Group 2 are the alkaline earth metals. 85. The elements in Group 17 ...
REDOX EQUILIBRIA SL - chemistryatdulwich
... agent. Potassium atoms will displace/reduce ions of less reactive metals. Also, in the series above Au+ (gold ion) is the strongest oxidising agent as Au (gold atom) is the weakest reducing agent. Non-metals As non-metals usually gain electrons during reactions they are usually oxidising agents. Jus ...
... agent. Potassium atoms will displace/reduce ions of less reactive metals. Also, in the series above Au+ (gold ion) is the strongest oxidising agent as Au (gold atom) is the weakest reducing agent. Non-metals As non-metals usually gain electrons during reactions they are usually oxidising agents. Jus ...
REDOX EQUILIBRIA SL - chemistryatdulwich
... agent. Potassium atoms will displace/reduce ions of less reactive metals. Also, in the series above Au+ (gold ion) is the strongest oxidising agent as Au (gold atom) is the weakest reducing agent. Non-metals As non-metals usually gain electrons during reactions they are usually oxidising agents. Jus ...
... agent. Potassium atoms will displace/reduce ions of less reactive metals. Also, in the series above Au+ (gold ion) is the strongest oxidising agent as Au (gold atom) is the weakest reducing agent. Non-metals As non-metals usually gain electrons during reactions they are usually oxidising agents. Jus ...
Chapter 4 - profpaz.com
... Chemists have devised a scheme to track electrons before and after a reaction in order to simplify this process. In this scheme, a number (oxidation state or number) is assigned to each element assuming that the shared electrons between two atoms belong to the one with the most attraction for these ...
... Chemists have devised a scheme to track electrons before and after a reaction in order to simplify this process. In this scheme, a number (oxidation state or number) is assigned to each element assuming that the shared electrons between two atoms belong to the one with the most attraction for these ...
REDOX EQUILIBRIA SL - chemistryatdulwich
... agent. Potassium atoms will displace/reduce ions of less reactive metals. Also, in the series above Au+ (gold ion) is the strongest oxidising agent as Au (gold atom) is the weakest reducing agent. Non-metals As non-metals usually gain electrons during reactions they are usually oxidising agents. Jus ...
... agent. Potassium atoms will displace/reduce ions of less reactive metals. Also, in the series above Au+ (gold ion) is the strongest oxidising agent as Au (gold atom) is the weakest reducing agent. Non-metals As non-metals usually gain electrons during reactions they are usually oxidising agents. Jus ...
Classifying Chemical Reactions by What Atoms Do
... 2 Na(s) + Cl2(g) → 2 NaCl(s) The reactions involve a metal reacting with a nonmetal. In addition, both reactions involve the conversion of free elements into ions. ...
... 2 Na(s) + Cl2(g) → 2 NaCl(s) The reactions involve a metal reacting with a nonmetal. In addition, both reactions involve the conversion of free elements into ions. ...
Chemistry B1A - Bakersfield College
... Describe the development of the periodic table, how it was originally arranged, how it is currently arranged, what standard is used to determine the atomic weight and what information can be determine from the table. ...
... Describe the development of the periodic table, how it was originally arranged, how it is currently arranged, what standard is used to determine the atomic weight and what information can be determine from the table. ...
2. Chemistry of Living Things Outline
... catalyze. In organisms, _____________ allow the chemical reactions of ______________ to take place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but must be present fo ...
... catalyze. In organisms, _____________ allow the chemical reactions of ______________ to take place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but must be present fo ...
Chemistry of Living Things Outline
... reaction they catalyze. In organisms, _____________ allow the chemical reactions of ______________ to take place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but ...
... reaction they catalyze. In organisms, _____________ allow the chemical reactions of ______________ to take place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but ...
Pauling Scale of Electronegativities for the Various Elements
... The driving force for metathesis reactions is the removal of ions from solution. Therefore, to predict the direction of a metathesis reaction one simply determines the degree of removal of ions from solution by the reactants and products. Prediction products is trivial: cations and anions merely exc ...
... The driving force for metathesis reactions is the removal of ions from solution. Therefore, to predict the direction of a metathesis reaction one simply determines the degree of removal of ions from solution by the reactants and products. Prediction products is trivial: cations and anions merely exc ...
13.0 Redox Reactions PowerPoint
... ▫ Remember the reactions can be read both directions so really you have both oxidation and reduction half reactions Ag+(aq) + 1 e- Ag(s) ...
... ▫ Remember the reactions can be read both directions so really you have both oxidation and reduction half reactions Ag+(aq) + 1 e- Ag(s) ...
9F Reactivity - Parrs Wood High School
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
Chemical Reactions
... Reactants and Products • In a chemical reaction, the substances that undergo change are called reactants • The new substances formed as a result of that change are called products • The arrow in the equation means that the reaction of the reactants yields to the product ...
... Reactants and Products • In a chemical reaction, the substances that undergo change are called reactants • The new substances formed as a result of that change are called products • The arrow in the equation means that the reaction of the reactants yields to the product ...
Lecture 1: RDCH 710 Introduction
... Difficulty in understanding data Chemical forms of species • Determine ratios of each redox species from XANES Use Nernst equation to determine potentials ...
... Difficulty in understanding data Chemical forms of species • Determine ratios of each redox species from XANES Use Nernst equation to determine potentials ...
Document
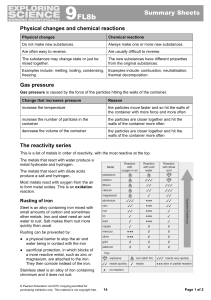
... In a chemical reaction a new substance is always formed. Most chemical changes are not easily reversed; they are irreversible. In a physical change no new substance is formed. Melting and evaporation are examples of physical changes. Physical changes are usually reversible. You can tell that a react ...
... In a chemical reaction a new substance is always formed. Most chemical changes are not easily reversed; they are irreversible. In a physical change no new substance is formed. Melting and evaporation are examples of physical changes. Physical changes are usually reversible. You can tell that a react ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.