Study of oxygen fugacity influence on redox state of iron in
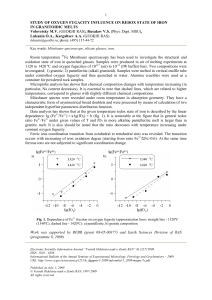
... 1120 to 1420 ºC and oxygen fugacities of 10-0.7 (air) to 10-13 (IW buffer) bars. Two compositions were investigated: 1) granitic; 2) pantelleritic (alkali granitoid). Samples were melted in vertical muffle tube under controlled oxygen fugacity and then quenched in water. Alumina crucibles were used ...
... 1120 to 1420 ºC and oxygen fugacities of 10-0.7 (air) to 10-13 (IW buffer) bars. Two compositions were investigated: 1) granitic; 2) pantelleritic (alkali granitoid). Samples were melted in vertical muffle tube under controlled oxygen fugacity and then quenched in water. Alumina crucibles were used ...
Introductory Chemistry Test Review
... 9. For the following chemical compounds, predict whether each will be soluble or insoluble in aqueous solution. a. Al(OH)3 b. Hg2Cl2 c. (NH4)2CO3 10. For the following aqueous chemical reactions, predict the possible products and identify any products that will be insoluble. a. CaCl2 + K2S b. MgCl2 ...
... 9. For the following chemical compounds, predict whether each will be soluble or insoluble in aqueous solution. a. Al(OH)3 b. Hg2Cl2 c. (NH4)2CO3 10. For the following aqueous chemical reactions, predict the possible products and identify any products that will be insoluble. a. CaCl2 + K2S b. MgCl2 ...
Chapter 4 Reactions in Aqueous Solutions
... Al(s) Al3+(aq) + 3e Ni2+(aq) + 2e Ni(s) – Multiply by a common factor to equalize electrons (the number of electrons lost must equal number of electrons gained) 2 [Al(s) Al3+(aq) + 3e ] 3 [Ni2+(aq) + 2e Ni(s) ] ...
... Al(s) Al3+(aq) + 3e Ni2+(aq) + 2e Ni(s) – Multiply by a common factor to equalize electrons (the number of electrons lost must equal number of electrons gained) 2 [Al(s) Al3+(aq) + 3e ] 3 [Ni2+(aq) + 2e Ni(s) ] ...
What do you know about light?
... • On its own the atomic number does not tell us the number of neutrons in an element. In order to determine the number of neutrons we need the mass number. ...
... • On its own the atomic number does not tell us the number of neutrons in an element. In order to determine the number of neutrons we need the mass number. ...
TERM 2 Unit 3 YR 9 SCI It is elementary
... 9 SCIENCE – Term 2 Unit 3: IT’S ELEMENTARY (5 WEEKS) ...
... 9 SCIENCE – Term 2 Unit 3: IT’S ELEMENTARY (5 WEEKS) ...
C1 Topic 1 Fundamental Ideas and The Earth REVISION Elements
... Higher Only 18. Why did Miller and Urey use methane, ammonia and hydrogen for the earth’s early experiment? 19. Why does Miller and Urey’s experiment only provide weak evidence? 20. Miller and Urey’s experiment suggested how ………….. …………….. were formed 21. What theory did the Miller and Urey experime ...
... Higher Only 18. Why did Miller and Urey use methane, ammonia and hydrogen for the earth’s early experiment? 19. Why does Miller and Urey’s experiment only provide weak evidence? 20. Miller and Urey’s experiment suggested how ………….. …………….. were formed 21. What theory did the Miller and Urey experime ...
General Chemistry First Semester Review General
... Phase notations (solid, liquids, gases, aqueous) - aqueous (aq) is written if a solution is used - pure liquids (not a mixture of something) use (l) - solid: This could refer to a multitude of different substances: metals, flakes, crystals, and precipitates; use (s) - gas: Use (g), these are usually ...
... Phase notations (solid, liquids, gases, aqueous) - aqueous (aq) is written if a solution is used - pure liquids (not a mixture of something) use (l) - solid: This could refer to a multitude of different substances: metals, flakes, crystals, and precipitates; use (s) - gas: Use (g), these are usually ...
Types of Chemical Reactions
... of a combination of carbon and hydrogen) to form water and carbon dioxide. These reactions are exothermic, meaning they produce heat. This reaction can be expressed as: ...
... of a combination of carbon and hydrogen) to form water and carbon dioxide. These reactions are exothermic, meaning they produce heat. This reaction can be expressed as: ...
Review-Semester Final (Part I)
... 18. Which holds its electrons more tightly- metals or nonmetals? How does this affect the properties of each? ...
... 18. Which holds its electrons more tightly- metals or nonmetals? How does this affect the properties of each? ...
Matter and Measurement
... 1) The oxidation state on any atom in its elemental form is zero (H2, O2, Na(s)) 2) The sum of the oxidation states of all atoms in a neutral compound is zero (CH4, NH3) 3) The sum of the oxidation states of all atoms in an ion is equal to the charge on the ion (NO3-, SO42-) 4) The oxidation state o ...
... 1) The oxidation state on any atom in its elemental form is zero (H2, O2, Na(s)) 2) The sum of the oxidation states of all atoms in a neutral compound is zero (CH4, NH3) 3) The sum of the oxidation states of all atoms in an ion is equal to the charge on the ion (NO3-, SO42-) 4) The oxidation state o ...
+ CuO Cu + O
... a-gives oxygen b- removes oxygen c-gives hydrogen d- (b and c) 11- When sodium atom loses an electron from its outermost energy level, it becomes ………………… a- oxidized b-reducing agent c- reduced d- (a and b) Put (√) or (x) and correct the mistake: 1- Sulphur dioxide is produced on heating blue copper ...
... a-gives oxygen b- removes oxygen c-gives hydrogen d- (b and c) 11- When sodium atom loses an electron from its outermost energy level, it becomes ………………… a- oxidized b-reducing agent c- reduced d- (a and b) Put (√) or (x) and correct the mistake: 1- Sulphur dioxide is produced on heating blue copper ...
Unit 5 Chemical Properties and Changes Video Notes A ______ is a
... ________________________ A change that alters the identity of a substance resulting in a new substance or substances with different properties ________________________ Those characteristics that can be observed when a chemical reaction changes the identity of the substance, such as potential to rus ...
... ________________________ A change that alters the identity of a substance resulting in a new substance or substances with different properties ________________________ Those characteristics that can be observed when a chemical reaction changes the identity of the substance, such as potential to rus ...
Reaction Predictions
... An electrolysis reaction is a reaction in which a nonspontaneous redox reaction is brought about by the passage of current under sufficient external electrical potential. The devices in which electrolysis reactions occur are called electrolytic cells. In theory, E° values (Standard Reduction Potenti ...
... An electrolysis reaction is a reaction in which a nonspontaneous redox reaction is brought about by the passage of current under sufficient external electrical potential. The devices in which electrolysis reactions occur are called electrolytic cells. In theory, E° values (Standard Reduction Potenti ...
Problem Set 2
... a) The oxidation step: ----------------------------------------------------b) The reduction step: ------------------------------------------------------c) The oxidizing agent: ------------------------------------------------------d) The reducing agent: ----------------------------------------------- ...
... a) The oxidation step: ----------------------------------------------------b) The reduction step: ------------------------------------------------------c) The oxidizing agent: ------------------------------------------------------d) The reducing agent: ----------------------------------------------- ...
Aeration
... Fe+3 (oxidized) -------------------- Fe+2 (reduced) Mn+4 (oxidized) --------------------- Mn+2 (reduced) NO3- (oxidized) ---------------------- N2 (reduced) Well Oxidized: sufficient Oxygen ...
... Fe+3 (oxidized) -------------------- Fe+2 (reduced) Mn+4 (oxidized) --------------------- Mn+2 (reduced) NO3- (oxidized) ---------------------- N2 (reduced) Well Oxidized: sufficient Oxygen ...
Dr Davids Essential Chemistry Definitions Bk1
... It has a numerical value of 6.02 x 1023 mol-1 Oxidation number: The difference between the number of electrons associated with an element in a compound and the element itself. Just for the purpose of assigning oxidation numbers all compounds are considered to contain ions, eg, CO2, C 4+ 2O 2- (but i ...
... It has a numerical value of 6.02 x 1023 mol-1 Oxidation number: The difference between the number of electrons associated with an element in a compound and the element itself. Just for the purpose of assigning oxidation numbers all compounds are considered to contain ions, eg, CO2, C 4+ 2O 2- (but i ...
Chemical Reactions and Equations
... What is a ‘Chemical Equation’? A Chemical Equation represents a chemical reaction. A complete chemical equation represents the reactants, products, and their physical states symbolically. A chemical equation also indicates a number of atoms of each element involved in a reaction. A chemical equation ...
... What is a ‘Chemical Equation’? A Chemical Equation represents a chemical reaction. A complete chemical equation represents the reactants, products, and their physical states symbolically. A chemical equation also indicates a number of atoms of each element involved in a reaction. A chemical equation ...
Chemical Reactions and Equations
... What is a ‘Chemical Equation’? A Chemical Equation represents a chemical reaction. A complete chemical equation represents the reactants, products, and their physical states symbolically. A chemical equation also indicates a number of atoms of each element involved in a reaction. A chemical equation ...
... What is a ‘Chemical Equation’? A Chemical Equation represents a chemical reaction. A complete chemical equation represents the reactants, products, and their physical states symbolically. A chemical equation also indicates a number of atoms of each element involved in a reaction. A chemical equation ...
105
... number decreases. When balancing equations by the half-reaction method in section 10.3, you sometimes used oxidation numbers to determine the reactant(s) and product(s) in each half-reaction. In fact, you can use oxidation numbers to balance a chemical equation by a new method. The oxidation number ...
... number decreases. When balancing equations by the half-reaction method in section 10.3, you sometimes used oxidation numbers to determine the reactant(s) and product(s) in each half-reaction. In fact, you can use oxidation numbers to balance a chemical equation by a new method. The oxidation number ...
- Palisades School District
... compounds M(OH)2 and MCO3 , where M represents an unidentified metal. (a) Identify the charge of the M ion in the ionic compounds above. (b) At 25°C, a saturated solution of M(OH)2 has a pH of 9.15. Calculate the molar concentration of OH-(aq) in the saturated solution. 2. Zinc metal is added to a h ...
... compounds M(OH)2 and MCO3 , where M represents an unidentified metal. (a) Identify the charge of the M ion in the ionic compounds above. (b) At 25°C, a saturated solution of M(OH)2 has a pH of 9.15. Calculate the molar concentration of OH-(aq) in the saturated solution. 2. Zinc metal is added to a h ...
Introduction_to_Geochemistry_Pre-Lecture_Quiz
... detach the loosest electron from atoms of that element. (e) All alkali metals have similar chemical properties. (f) Alkali earths have one electron in the outer shell. (g) Electronegativity is the amount of negative charge on an atom. (h) Ca has a valency of 2. (i) True ionic bonds are unknown and a ...
... detach the loosest electron from atoms of that element. (e) All alkali metals have similar chemical properties. (f) Alkali earths have one electron in the outer shell. (g) Electronegativity is the amount of negative charge on an atom. (h) Ca has a valency of 2. (i) True ionic bonds are unknown and a ...
10 TEST 2 (of 3)
... (b) Hess’s Law: The change in ______________ for a stepwise process is the sum of the changes of the steps. ...
... (b) Hess’s Law: The change in ______________ for a stepwise process is the sum of the changes of the steps. ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.