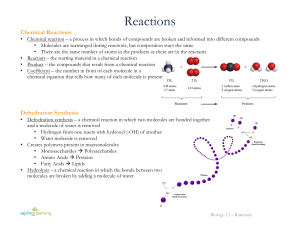
Reactions
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
Oxidation numbers
... battery, are few examples of a very common and important type of chemical reaction. These chemical changes are all classified as "electron-transfer" or oxidation-reduction reactions. The term, oxidation , was derived from the observation that almost all elements reacted with oxygen to form compounds ...
... battery, are few examples of a very common and important type of chemical reaction. These chemical changes are all classified as "electron-transfer" or oxidation-reduction reactions. The term, oxidation , was derived from the observation that almost all elements reacted with oxygen to form compounds ...
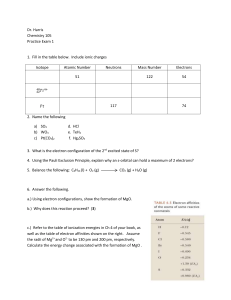
Dr. Harris Chemistry 105 Practice Exam 1 Isotope Atomic Number
... 300 nm, and assuming that the emission rate is constant, how many photons are emitted per minute? ...
... 300 nm, and assuming that the emission rate is constant, how many photons are emitted per minute? ...
Chapter 12 Oxidation-Reduction Reactions
... The theoretical definition of oxidation is the loss of electrons. The operational definition of reduction is the extracting of metals from metal ore. The theoretical definition of reduction is the gain of electrons. If one atom is oxidized in a reaction, another atom must be reduced. An oxidizing ag ...
... The theoretical definition of oxidation is the loss of electrons. The operational definition of reduction is the extracting of metals from metal ore. The theoretical definition of reduction is the gain of electrons. If one atom is oxidized in a reaction, another atom must be reduced. An oxidizing ag ...
Chem 1411 Chapter 4
... An oxidation reaction is the one that involves loss of electrons and a reduction reaction involves the gain of electrons. A reaction in which oxidation and reduction occurs simultaneously is called a redox reaction. The species that undergoes oxidation is the reductant (reducing agent); and the spec ...
... An oxidation reaction is the one that involves loss of electrons and a reduction reaction involves the gain of electrons. A reaction in which oxidation and reduction occurs simultaneously is called a redox reaction. The species that undergoes oxidation is the reductant (reducing agent); and the spec ...
Chemical Reactions PPT
... Since oxidation is the loss of electrons and reduction is the gain of electrons, they must occur simultaneously. ** Any chemical process in which elements undergo changes in oxidation number is an oxidation – reduction reaction , or redox reaction for short.** ...
... Since oxidation is the loss of electrons and reduction is the gain of electrons, they must occur simultaneously. ** Any chemical process in which elements undergo changes in oxidation number is an oxidation – reduction reaction , or redox reaction for short.** ...
Chapter 5—Chemical Reactions
... In this equation, A and B represent elements and AX and BX are compounds. • An example of a single replacement reaction is: ...
... In this equation, A and B represent elements and AX and BX are compounds. • An example of a single replacement reaction is: ...
3.10 Neutralization
... ZnS(s) + 2HCl(aq) → ZnCl2(aq) + H2S(g) ZnS(s) + 2H+ + 2Cl- → Zn2+ + 2Cl- + H2S(g) ⇒ZnS(s) + 2H+ → Zn2+ + H2S(g) – H+ is present in the form of H3O+ ...
... ZnS(s) + 2HCl(aq) → ZnCl2(aq) + H2S(g) ZnS(s) + 2H+ + 2Cl- → Zn2+ + 2Cl- + H2S(g) ⇒ZnS(s) + 2H+ → Zn2+ + H2S(g) – H+ is present in the form of H3O+ ...
1 Types of Chemical Reactions
... Sudden dramatic changes in temperature occur. A new solid suddenly appears. ...
... Sudden dramatic changes in temperature occur. A new solid suddenly appears. ...
File - Ingolstadt Academy
... Dilution Colligative properties (FP depression, BP elevation) Acids and Bases: Properties of Acids and Bases Definitions of Acids and Bases (Arrhenius, Bronsted-Lowery, Lewis) Ka, Kb expressions Strengths of acids and bases (Strong vs. Weak) Calculating pH, pOH, Kw Buffers Redox Reac ...
... Dilution Colligative properties (FP depression, BP elevation) Acids and Bases: Properties of Acids and Bases Definitions of Acids and Bases (Arrhenius, Bronsted-Lowery, Lewis) Ka, Kb expressions Strengths of acids and bases (Strong vs. Weak) Calculating pH, pOH, Kw Buffers Redox Reac ...
Chemistry of Life - juan-roldan
... ◦ Measure of how acidic or basic a substance is ◦ The negative logarithm of the hydrogen ion concentration ◦ Expressed in moles per liter ...
... ◦ Measure of how acidic or basic a substance is ◦ The negative logarithm of the hydrogen ion concentration ◦ Expressed in moles per liter ...
Lecture 5 – Chemical Reactions
... Rule 6: The algebraic sum of the O.N.’s of all atoms in a complete compound formula equals zero. g. Rule 7: The algebraic sum of the O.N.’s of all atoms in a poly atomic ion is equal to the charge on the ion. If in a reaction the oxidation number for an element increases, it is oxidized; conversely, ...
... Rule 6: The algebraic sum of the O.N.’s of all atoms in a complete compound formula equals zero. g. Rule 7: The algebraic sum of the O.N.’s of all atoms in a poly atomic ion is equal to the charge on the ion. If in a reaction the oxidation number for an element increases, it is oxidized; conversely, ...
Redox Reactions C12-1-10
... electrons are partially transferred from hydrogen to oxygen. Oxygen is a more electronegative element than hydrogen. The electron pair in the covalent bond is shifted toward oxygen resulting in a partial negative charge on oxygen and partial positive charge on hydrogen. Both reactions above are exam ...
... electrons are partially transferred from hydrogen to oxygen. Oxygen is a more electronegative element than hydrogen. The electron pair in the covalent bond is shifted toward oxygen resulting in a partial negative charge on oxygen and partial positive charge on hydrogen. Both reactions above are exam ...
Test-tube Reactions - University of Manitoba
... electrons are partially transferred from hydrogen to oxygen. Oxygen is a more electronegative element than hydrogen. The electron pair in the covalent bond is shifted toward oxygen resulting in a partial negative charge on oxygen and partial positive charge on hydrogen. Both reactions above are exam ...
... electrons are partially transferred from hydrogen to oxygen. Oxygen is a more electronegative element than hydrogen. The electron pair in the covalent bond is shifted toward oxygen resulting in a partial negative charge on oxygen and partial positive charge on hydrogen. Both reactions above are exam ...
Chemical Reactions: Helpful Hints
... Reaction 10 involves a solid metal (Zn in the 0 oxidation state) and an aqueous metal ion (Ag+ in the 1+ oxidation state). Did you observe a band of shiny metal being formed at the interface of the solid and the solution (Hmm, what could that be? What was in solution that would give such luster?) R ...
... Reaction 10 involves a solid metal (Zn in the 0 oxidation state) and an aqueous metal ion (Ag+ in the 1+ oxidation state). Did you observe a band of shiny metal being formed at the interface of the solid and the solution (Hmm, what could that be? What was in solution that would give such luster?) R ...
Chemical Reactions
... What is a chemical reaction? • A chemical reaction is the process by which the atoms of one or more substances are rearranged to form ...
... What is a chemical reaction? • A chemical reaction is the process by which the atoms of one or more substances are rearranged to form ...
Chapter 3 – part I Sections 1-3
... Precipitation is a type of DR Rxn and a solid forms AX (aq) + ...
... Precipitation is a type of DR Rxn and a solid forms AX (aq) + ...
CH 115 Exam 2 - UAB General Chemistry Supplemental Instruction
... A. This is a gas-forming reaction B. The CO2 comes from the rapid decomposition of H2CO3 C. The reaction is already balanced ...
... A. This is a gas-forming reaction B. The CO2 comes from the rapid decomposition of H2CO3 C. The reaction is already balanced ...
Gizmos: Types of Reactions
... Introduction: Chemical equations show how compounds and elements react with one another. An element is a substance consisting of one kind of atom, such as aluminum (Al) or oxygen gas (O2). A compound is a substance made of more than one kind of atom, such as water (H2O) or table salt (NaCl). Questio ...
... Introduction: Chemical equations show how compounds and elements react with one another. An element is a substance consisting of one kind of atom, such as aluminum (Al) or oxygen gas (O2). A compound is a substance made of more than one kind of atom, such as water (H2O) or table salt (NaCl). Questio ...
California Chemistry Standards Test
... Which has a sour taste a. base b. metal c. acid d. salt Which of the following elements would combine w/ chlorine to form an ionic bond a. Ar b. S c. Si d. Mg The formula for the hydronium ion is a. H+ b. H3O+ c. OH- d. HCa5(PO4)3 is held together by a. freely moving electrons b. hydrogen bonds bet ...
... Which has a sour taste a. base b. metal c. acid d. salt Which of the following elements would combine w/ chlorine to form an ionic bond a. Ar b. S c. Si d. Mg The formula for the hydronium ion is a. H+ b. H3O+ c. OH- d. HCa5(PO4)3 is held together by a. freely moving electrons b. hydrogen bonds bet ...
1b-Redox FIB notes and practice
... In this reaction, the oxidation number of silver decreases from +1 to 0. This is _____________: silver is ______________ from Ag +1 to Ag 0. On the other hand, copper is ______________ in this reaction from Cu 0 to Cu +2. These results agree with those obtained by analyzing the reaction by using ele ...
... In this reaction, the oxidation number of silver decreases from +1 to 0. This is _____________: silver is ______________ from Ag +1 to Ag 0. On the other hand, copper is ______________ in this reaction from Cu 0 to Cu +2. These results agree with those obtained by analyzing the reaction by using ele ...
chemical reaction
... the oxidation state(O.S) of an atom in the pure (uncombined) element is 0. The total (sum) of the oxidation state of all the atoms in a molecule or formula unit is 0. For an ion total of the oxidation state is equal to the charge on the ion. In their compounds the alkali metals (1a groups Li, Na, K, ...
... the oxidation state(O.S) of an atom in the pure (uncombined) element is 0. The total (sum) of the oxidation state of all the atoms in a molecule or formula unit is 0. For an ion total of the oxidation state is equal to the charge on the ion. In their compounds the alkali metals (1a groups Li, Na, K, ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.