General Chemistry - Review for final exam: (Make sure you bring
... e. 2nd ionization energy 41. What are cations and anions? Which type of elements form each? 42. Write the ions formed by the following: State how many electrons were gained or lost. a. F b. Ca c. H d. S e. Al 43. What is the octet rule? 44. What are some basic properties of ionic compounds? 45. What ...
... e. 2nd ionization energy 41. What are cations and anions? Which type of elements form each? 42. Write the ions formed by the following: State how many electrons were gained or lost. a. F b. Ca c. H d. S e. Al 43. What is the octet rule? 44. What are some basic properties of ionic compounds? 45. What ...
PPT
... • Many reactions take place between compounds or elements that are dissolved in water. Ionic compounds and some polar covalent compounds break apart (dissociate) when they dissolve in water and form ions. ...
... • Many reactions take place between compounds or elements that are dissolved in water. Ionic compounds and some polar covalent compounds break apart (dissociate) when they dissolve in water and form ions. ...
Name:______ Chemistry 114 First Hour Exam
... 5.Draw the structures for the following organic compounds. Some the given names may be incorrect. If a name is not correct, give the correct name for the compound 2-methyl-6-ethylhexane ...
... 5.Draw the structures for the following organic compounds. Some the given names may be incorrect. If a name is not correct, give the correct name for the compound 2-methyl-6-ethylhexane ...
File
... 3. __________________: When two or more elements combine chemically you get a mixture. 4. __________________: The properties of elements do not change when mixtures are formed. 5. __________________: Particles in a solid are not packed at all. 6. __________________: Solids keep their shape. 7. _____ ...
... 3. __________________: When two or more elements combine chemically you get a mixture. 4. __________________: The properties of elements do not change when mixtures are formed. 5. __________________: Particles in a solid are not packed at all. 6. __________________: Solids keep their shape. 7. _____ ...
S3 Chemistry - eduBuzz.org
... Learning Outcomes for Atomic Theory and Periodic Table S3 Chemistry Identify the 3 sub atomic particles in an atom State the location, charge and mass of each sub atomic particle Calculate the number of protons, neutrons and electrons in an atom Identify whether a species has an equal or un ...
... Learning Outcomes for Atomic Theory and Periodic Table S3 Chemistry Identify the 3 sub atomic particles in an atom State the location, charge and mass of each sub atomic particle Calculate the number of protons, neutrons and electrons in an atom Identify whether a species has an equal or un ...
Learning Outcomes for Chemical Reactions and
... Learning Outcomes for Atomic Theory and Periodic Table S3 Chemistry • Identify the 3 sub atomic particles in an atom • State the location, charge and mass of each sub atomic particle • Calculate the number of protons, neutrons and electrons in an atom • Identify whether a species has an equal or un ...
... Learning Outcomes for Atomic Theory and Periodic Table S3 Chemistry • Identify the 3 sub atomic particles in an atom • State the location, charge and mass of each sub atomic particle • Calculate the number of protons, neutrons and electrons in an atom • Identify whether a species has an equal or un ...
Chapter 13 Notes
... Oxidation is a type of reaction in which an element loses one or more electrons. Reduction is a type of reaction in which an element gains one or more electrons. A single replacement reaction that involves oxidation and reduction occurs in the steel-making process. ...
... Oxidation is a type of reaction in which an element loses one or more electrons. Reduction is a type of reaction in which an element gains one or more electrons. A single replacement reaction that involves oxidation and reduction occurs in the steel-making process. ...
3rd Quarter Test
... 19) Equilibrium is reached in all reversible chemical reactions when the a) forward reaction stops b) reverse reaction stops c) concentration of the reactants and the products becomes equal d) rates of the opposing reaction becomes equal 20) For a chemical system at equilibrium, a rise in temperatur ...
... 19) Equilibrium is reached in all reversible chemical reactions when the a) forward reaction stops b) reverse reaction stops c) concentration of the reactants and the products becomes equal d) rates of the opposing reaction becomes equal 20) For a chemical system at equilibrium, a rise in temperatur ...
File
... SCH4U b) Determine the oxidation number of the sulfur in the sulfate ion, SO4-2. Remember that the sum of the oxidation numbers must equal the charge of the ion. Step 1: Write All Known Oxidation Numbers Use the symbol N to represent the oxidation number of sulfur in the sulfate. ...
... SCH4U b) Determine the oxidation number of the sulfur in the sulfate ion, SO4-2. Remember that the sum of the oxidation numbers must equal the charge of the ion. Step 1: Write All Known Oxidation Numbers Use the symbol N to represent the oxidation number of sulfur in the sulfate. ...
NAME REVIEW 1: JUST THE BASICS ___1) In which material are
... the cathode 4) Reduction occurs at both the anode and the cathode ___8) Which statement correctly describes an endothermic chemical reaction? 1) The products have higher potential energy than the reactants and the ∆H is negative ...
... the cathode 4) Reduction occurs at both the anode and the cathode ___8) Which statement correctly describes an endothermic chemical reaction? 1) The products have higher potential energy than the reactants and the ∆H is negative ...
Sample Paper - Army Public School Jammu Cantt
... for either Cr or Mn metals What can be inferred from the magnetic moment of the complex K4[Mn(CN)6] Magnetic moment: 2.2 BM? Describe the type of hybridisation for the complex ion [Fe(H2O)6]2+. Write the IUPAC name of the ionisation isomer of the coordination compound [Co(NH3)5Br]SO4. Give one chemi ...
... for either Cr or Mn metals What can be inferred from the magnetic moment of the complex K4[Mn(CN)6] Magnetic moment: 2.2 BM? Describe the type of hybridisation for the complex ion [Fe(H2O)6]2+. Write the IUPAC name of the ionisation isomer of the coordination compound [Co(NH3)5Br]SO4. Give one chemi ...
2A Final Exam Review Worksheet
... o Isotope = occurs when the number of neutrons vary for a particular element. The number of protons do not vary. o Given any two of these variables, be able to solve for the third: weighted average atomic mass, isotopic mass, fractional abundance Counting protons, neutrons, and electrons in elements ...
... o Isotope = occurs when the number of neutrons vary for a particular element. The number of protons do not vary. o Given any two of these variables, be able to solve for the third: weighted average atomic mass, isotopic mass, fractional abundance Counting protons, neutrons, and electrons in elements ...
Chemical Basis of Life
... Closer to the nucleus = lower energy and filled first Octet rule: hold up to 8 electrons (not 1st = 2) ...
... Closer to the nucleus = lower energy and filled first Octet rule: hold up to 8 electrons (not 1st = 2) ...
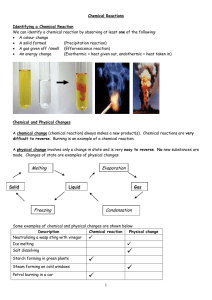
Chemical Reactions
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
Chemical Reactions
... Synthesis Reaction • Synthesis – 2 substances (reactants) combine to form a new substance (product). – Substances are either atoms (elements) or compounds in this case. A + ...
... Synthesis Reaction • Synthesis – 2 substances (reactants) combine to form a new substance (product). – Substances are either atoms (elements) or compounds in this case. A + ...
Chem 30A Final Exam
... 14. Write correct chemical formulas for the following compounds. In each case, indicate whether the compound involves ionic bonding, covalent bonding, or both. formula? ionic? covalent? both? potassium carbonate ...
... 14. Write correct chemical formulas for the following compounds. In each case, indicate whether the compound involves ionic bonding, covalent bonding, or both. formula? ionic? covalent? both? potassium carbonate ...
Table showing examples of Complex ions with their bond
... The d-block elements generally exhibit the characteristic properties of metals because the outer electrons (d) are loosely bound and readily enter into metallic bond formation. The metallic radius decreased in passing from Sc to Ni. Addition of electrons might be expected to result in an increase in ...
... The d-block elements generally exhibit the characteristic properties of metals because the outer electrons (d) are loosely bound and readily enter into metallic bond formation. The metallic radius decreased in passing from Sc to Ni. Addition of electrons might be expected to result in an increase in ...
Chapter 4
... Predict the products of some simple types of chemical reactions: combination, decomposition, displacement, and metathesis reactions. ...
... Predict the products of some simple types of chemical reactions: combination, decomposition, displacement, and metathesis reactions. ...
Polyatomic Ions (Memorize for Wednesday, January 31
... CHEMICAL NOMENCLATURE Oxidation Numbers (similar to valence electrons) Group 1 = +1 Group 2 = +2 Groups 13-18 = group # - 10 for most elements d-block elements must be looked up (groups 3-12 use roman numerals) oxidation numbers for ALL compounds MUST equal zero Metal-Non-metal (binary ionic compoun ...
... CHEMICAL NOMENCLATURE Oxidation Numbers (similar to valence electrons) Group 1 = +1 Group 2 = +2 Groups 13-18 = group # - 10 for most elements d-block elements must be looked up (groups 3-12 use roman numerals) oxidation numbers for ALL compounds MUST equal zero Metal-Non-metal (binary ionic compoun ...
Valence electrons and Lewis Dot Structures
... or more atoms of the same element, or a group of atoms of different elements that have combined to form a compound. ...
... or more atoms of the same element, or a group of atoms of different elements that have combined to form a compound. ...
Slide 1
... reaction; it shows the complete formulas of all reactants and products. • However, although this equation shows the reactants and products of the reaction, it does not give a very clear picture of what actually occurs in solution. • The complete ionic equation, better represents the actual forms of ...
... reaction; it shows the complete formulas of all reactants and products. • However, although this equation shows the reactants and products of the reaction, it does not give a very clear picture of what actually occurs in solution. • The complete ionic equation, better represents the actual forms of ...
Chapter 4 - GEOCITIES.ws
... Write the half reactions for the following. Na + Cl2 Na+ + ClSO3- + H+ + MnO4- SO4- + H2O + Mn+2 ...
... Write the half reactions for the following. Na + Cl2 Na+ + ClSO3- + H+ + MnO4- SO4- + H2O + Mn+2 ...
12-3: Lewis Structures
... o Octet Rule—most elements will be surrounded by 8 dots, representing noble gas configuration Hydrogen is full with 2 electrons (2 dots on one side)—so it is like helium Draw the Lewis structures for: H Ca N F ...
... o Octet Rule—most elements will be surrounded by 8 dots, representing noble gas configuration Hydrogen is full with 2 electrons (2 dots on one side)—so it is like helium Draw the Lewis structures for: H Ca N F ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.