BC10TEXTCH05_Sec3
... Chemists in the early 19th century knew that organisms produce a huge number of compounds containing carbon. The chemists called these organic compounds because the compounds were made by organisms. Today, we use the term organic compound to refer to almost all carbon-containing compounds, whether t ...
... Chemists in the early 19th century knew that organisms produce a huge number of compounds containing carbon. The chemists called these organic compounds because the compounds were made by organisms. Today, we use the term organic compound to refer to almost all carbon-containing compounds, whether t ...
Biology project Lz
... carbon—even though many of the organic compounds known today have no connection to any substance found in living organisms. There is no single "official" definition of an organic compound. Some textbooks define an organic compound as one that contains one or more C-H bonds. Others include C-C bonds ...
... carbon—even though many of the organic compounds known today have no connection to any substance found in living organisms. There is no single "official" definition of an organic compound. Some textbooks define an organic compound as one that contains one or more C-H bonds. Others include C-C bonds ...
Towards the atomic level protein sequence analysis
... growth and developments, affected by plant–water relations. A majority of plant aquaporins have been found to share vital structural features with the human aquaporin and show water-transporting ability in various functional assays, and some have been shown experimentally to be of significant import ...
... growth and developments, affected by plant–water relations. A majority of plant aquaporins have been found to share vital structural features with the human aquaporin and show water-transporting ability in various functional assays, and some have been shown experimentally to be of significant import ...
organic chemistry - Peoria Public Schools
... Complexity of molecules: has a different effect on the melting point than on the boiling point. See table below: Straight chained alkanes have higher boiling points than branched alkanes of similar molecular mass as there is more contact/larger surface area between the straight-chained molecules and ...
... Complexity of molecules: has a different effect on the melting point than on the boiling point. See table below: Straight chained alkanes have higher boiling points than branched alkanes of similar molecular mass as there is more contact/larger surface area between the straight-chained molecules and ...
2–3 Carbon Compounds
... Lipids are generally not soluble in water. Lipids are made mostly from carbon and hydrogen atoms. ...
... Lipids are generally not soluble in water. Lipids are made mostly from carbon and hydrogen atoms. ...
e is nline ion any er.`
... present, and human health interest because the arsenicals occur in many common foods. The highest arsenic concentrations are found in seafoods, and many studies have reported on the type of arsenic compounds present in such foods.1 The vast majority of this work has dealt with water-soluble compound ...
... present, and human health interest because the arsenicals occur in many common foods. The highest arsenic concentrations are found in seafoods, and many studies have reported on the type of arsenic compounds present in such foods.1 The vast majority of this work has dealt with water-soluble compound ...
File
... Many lipids are formed from glycerol and a. fatty acids. b. monosaccharides. c. amino acids. ...
... Many lipids are formed from glycerol and a. fatty acids. b. monosaccharides. c. amino acids. ...
CHAPTER 4 DISTRIBUTION OF CARBON, SULPHUR, NITROGEN
... protein is highly stable at that number of atoms per residue. Specifically, individual amino acids and whole proteins can vary greatly in their content of carbon. Since, carbon is the only element that contributes towards the hydrophobic interaction, a dominant force, which helps in maintaining the ...
... protein is highly stable at that number of atoms per residue. Specifically, individual amino acids and whole proteins can vary greatly in their content of carbon. Since, carbon is the only element that contributes towards the hydrophobic interaction, a dominant force, which helps in maintaining the ...
Slide ()
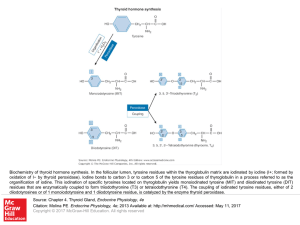
... Biochemistry of thyroid hormone synthesis. In the follicular lumen, tyrosine residues within the thyroglobulin matrix are iodinated by iodine (I+; formed by oxidation of I− by thyroid peroxidase). Iodine bonds to carbon 3 or to carbon 5 of the tyrosine residues of thyroglobulin in a process referred ...
... Biochemistry of thyroid hormone synthesis. In the follicular lumen, tyrosine residues within the thyroglobulin matrix are iodinated by iodine (I+; formed by oxidation of I− by thyroid peroxidase). Iodine bonds to carbon 3 or to carbon 5 of the tyrosine residues of thyroglobulin in a process referred ...
cycle - realfuture.org
... discussion on microbial diversity. In addition to requiring energy, all known forms of life are carbon-based and require carbon as their primary macronutrient. It is for this reason that life is commonly categorized on the basis of both energy and carbon sources (Figure 1). Phototrophs obtain their ...
... discussion on microbial diversity. In addition to requiring energy, all known forms of life are carbon-based and require carbon as their primary macronutrient. It is for this reason that life is commonly categorized on the basis of both energy and carbon sources (Figure 1). Phototrophs obtain their ...
cycle - realfuture.org
... discussion on microbial diversity. In addition to requiring energy, all known forms of life are carbon-based and require carbon as their primary macronutrient. It is for this reason that life is commonly categorized on the basis of both energy and carbon sources (Figure 1). Phototrophs obtain their ...
... discussion on microbial diversity. In addition to requiring energy, all known forms of life are carbon-based and require carbon as their primary macronutrient. It is for this reason that life is commonly categorized on the basis of both energy and carbon sources (Figure 1). Phototrophs obtain their ...
Are you ready for S279?
... The amount of maths in S279 is significant, and essential. Throughout the course, graphs are commonly used to show the relationship between two variables. Angles are measured in degrees and the sine, cosine and tangent of angles are also used at various points throughout the course. Several algebrai ...
... The amount of maths in S279 is significant, and essential. Throughout the course, graphs are commonly used to show the relationship between two variables. Angles are measured in degrees and the sine, cosine and tangent of angles are also used at various points throughout the course. Several algebrai ...
Biocompatibility of synthetic and bio-material fusion
... ligands with weak to intermediate field strength and multidenticities; (iii) design of inert complexes, and (iv) development of multi-nuclear complexes. Keywords: Biocompatibility, carbon transformation, drug delivery, sequestration, synthetic–natural fusion. SYNTHETIC and biological materials can b ...
... ligands with weak to intermediate field strength and multidenticities; (iii) design of inert complexes, and (iv) development of multi-nuclear complexes. Keywords: Biocompatibility, carbon transformation, drug delivery, sequestration, synthetic–natural fusion. SYNTHETIC and biological materials can b ...
L24_Krebs
... • Fluoroacetate is a strong inhibitor of the cycle – Creates fluorocitrate which inhibits citrate utilisation – Very toxic and used as poison(1080) ...
... • Fluoroacetate is a strong inhibitor of the cycle – Creates fluorocitrate which inhibits citrate utilisation – Very toxic and used as poison(1080) ...
2-3
... Why is carbon so interesting that a whole branch of chemistry should be set aside just to study carbon compounds? There are two reasons for this. First, carbon atoms have four valence electrons, allowing them to form strong covalent bonds with many other elements. Carbon can bond with many elements, ...
... Why is carbon so interesting that a whole branch of chemistry should be set aside just to study carbon compounds? There are two reasons for this. First, carbon atoms have four valence electrons, allowing them to form strong covalent bonds with many other elements. Carbon can bond with many elements, ...
File - Mrs. Pisciotta`s Biology Classes
... Why is carbon so interesting that a whole branch of chemistry should be set aside just to study carbon compounds? There are two reasons for this. First, carbon atoms have four valence electrons, allowing them to form strong covalent bonds with many other elements. Carbon can bond with many elements, ...
... Why is carbon so interesting that a whole branch of chemistry should be set aside just to study carbon compounds? There are two reasons for this. First, carbon atoms have four valence electrons, allowing them to form strong covalent bonds with many other elements. Carbon can bond with many elements, ...
Ch 3 Notes
... • Carbon atoms can readily form four covalent bonds with other atoms including other carbon atoms. The carbon bonds allow the carbon atoms to form a wide variety of simple and complex organic compounds. ...
... • Carbon atoms can readily form four covalent bonds with other atoms including other carbon atoms. The carbon bonds allow the carbon atoms to form a wide variety of simple and complex organic compounds. ...
mb_ch03
... • Carbon atoms can readily form four covalent bonds with other atoms including other carbon atoms. The carbon bonds allow the carbon atoms to form a wide variety of simple and complex organic compounds. ...
... • Carbon atoms can readily form four covalent bonds with other atoms including other carbon atoms. The carbon bonds allow the carbon atoms to form a wide variety of simple and complex organic compounds. ...
Grade 11 Unit 8 - Amazon Web Services
... completely to CO2 and H2O, large amounts of pollution occur from the C, CO, and unburned hydrocarbons. Industrial cities that power their plants with low-grade soft coal have especially large amounts of pollution from the incomplete combustion of the soft coal. When coal is heated to a high tempera ...
... completely to CO2 and H2O, large amounts of pollution occur from the C, CO, and unburned hydrocarbons. Industrial cities that power their plants with low-grade soft coal have especially large amounts of pollution from the incomplete combustion of the soft coal. When coal is heated to a high tempera ...
A2 revision
... chemiosmosis. Also, NADP will not be reduced so no NADPH would be produced. This means that the light-independent stage (Calvin cycle) will not happen, so there will be no fixation of carbon dioxide into sugars or other organic molecules such as fatty acids or amino acids. As a result the plant will ...
... chemiosmosis. Also, NADP will not be reduced so no NADPH would be produced. This means that the light-independent stage (Calvin cycle) will not happen, so there will be no fixation of carbon dioxide into sugars or other organic molecules such as fatty acids or amino acids. As a result the plant will ...
Carbon Isotope Fractionation by Autotrophic Bacteria with Three
... bon cycle as early as 3.5 x 109, if not 3.8 x 109 years ago. In the past decade novel autotrophic C 0 2 fixation pathways have been unravelled occurring mostly in strictly anaerobic prokaryotes; so far. the reductive pentose phosphate cycle (Calvin cycle) could not be found in this group ([2 ], for ...
... bon cycle as early as 3.5 x 109, if not 3.8 x 109 years ago. In the past decade novel autotrophic C 0 2 fixation pathways have been unravelled occurring mostly in strictly anaerobic prokaryotes; so far. the reductive pentose phosphate cycle (Calvin cycle) could not be found in this group ([2 ], for ...
Chapter 2 3EPchanges
... the study of compounds that contain bonds between carbon atoms Inorganic chemistry the study of all other compounds. ...
... the study of compounds that contain bonds between carbon atoms Inorganic chemistry the study of all other compounds. ...
Fundamental of Organic chemistry
... <2> They usually react more slowly than inorganic compounds. Energy in the form of heat is usually required. <3> The reactions are seldom complete reacted. Purification is usually required to obtain the desired product. <4> Organic compounds are often exist in groups or families (Homologous series) ...
... <2> They usually react more slowly than inorganic compounds. Energy in the form of heat is usually required. <3> The reactions are seldom complete reacted. Purification is usually required to obtain the desired product. <4> Organic compounds are often exist in groups or families (Homologous series) ...
2–3 Carbon Compounds
... Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Carbon atoms have four valence electrons that can join with the electrons from other atoms to form strong covalent bonds. Living organisms are made of molecules that consist of carbon (C) and other elements (H, ...
... Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Carbon atoms have four valence electrons that can join with the electrons from other atoms to form strong covalent bonds. Living organisms are made of molecules that consist of carbon (C) and other elements (H, ...
Carbon

Carbon (from Latin: carbo ""coal"") is a chemical element with symbol C and atomic number 6. On the Periodic table, it is the first (row 2) of six elements in column (group) 14, which have in common the composition of their outer electron shell. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. There are three naturally occurring isotopes, with 12C and 13C being stable, while 14C is radioactive, decaying with a half-life of about 5,730 years. Carbon is one of the few elements known since antiquity.Carbon is the 15th most abundant element in the Earth's crust, and the fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. It is present in all forms of carbon-based life, and in the human body carbon is the second most abundant element by mass (about 18.5%) after oxygen. This abundance, together with the unique diversity of organic compounds and their unusual polymer-forming ability at the temperatures commonly encountered on Earth, make this element the chemical basis of all known life.The atoms of carbon can be bonded together in different ways: allotropes of carbon. The best known are graphite, diamond, and amorphous carbon. The physical properties of carbon vary widely with the allotropic form. For example, graphite is opaque and black, while diamond is highly transparent. Graphite is soft enough to form a streak on paper (hence its name, from the Greek word ""γράφω"" which means ""to write""), while diamond is the hardest naturally-occurring material known. Graphite is a very good conductor, while diamond has a very low electrical conductivity. Under normal conditions, diamond, carbon nanotubes, and graphene have the highest thermal conductivities of all known materials. All carbon allotropes are solids under normal conditions, with graphite being the most thermodynamically stable form. They are chemically resistant and require high temperature to react even with oxygen.The most common oxidation state of carbon in inorganic compounds is +4, while +2 is found in carbon monoxide and other transition metal carbonyl complexes. The largest sources of inorganic carbon are limestones, dolomites and carbon dioxide, but significant quantities occur in organic deposits of coal, peat, oil and methane clathrates. Carbon forms a vast number of compounds, more than any other element, with almost ten million compounds described to date, which in turn are a tiny fraction of such compounds that are theoretically possible under standard conditions.