2.6 M - Thierry Karsenti
... If you get 6 items or more correct you can consider that you are doing fine, but if you get less than 4 items correct then you have to work very hard to pass the course. ...
... If you get 6 items or more correct you can consider that you are doing fine, but if you get less than 4 items correct then you have to work very hard to pass the course. ...
Articles Oxidation Numbers in the Study of Metabolism
... the value gives the number of electrons over which ownership has changed compared with the valence state in the pure element. Atoms of pure elements have an oxidation number of zero; they have neither lost nor gained electrons, and the number of protons in the nucleus exactly balances the number of ...
... the value gives the number of electrons over which ownership has changed compared with the valence state in the pure element. Atoms of pure elements have an oxidation number of zero; they have neither lost nor gained electrons, and the number of protons in the nucleus exactly balances the number of ...
Contrasting C3, C4 and CAM Photosynthesis
... fixation and that of C4 and CAM is that the latter systems separate the initial “fixation” or drawing down of CO2 from the true fixation step that is catalysed by RuBisCO. ...
... fixation and that of C4 and CAM is that the latter systems separate the initial “fixation” or drawing down of CO2 from the true fixation step that is catalysed by RuBisCO. ...
Contrasting C3, C4 and CAM Photosynthesis
... fixation and that of C4 and CAM is that the latter systems separate the initial “fixation” or drawing down of CO2 from the true fixation step that is catalysed by RuBisCO. ...
... fixation and that of C4 and CAM is that the latter systems separate the initial “fixation” or drawing down of CO2 from the true fixation step that is catalysed by RuBisCO. ...
From CO2 to cell: energetic expense of creating biomass using the
... Given that dissolved oxygen concentrations can vary from 0 to ∼100 μM over spatial scales of less than 1 cm and temporal scales ranging from seconds to days, oxygen sensitivity is likely to play a role. Additionally, electron donor availabilities are also quite variable; for example, sulfide concent ...
... Given that dissolved oxygen concentrations can vary from 0 to ∼100 μM over spatial scales of less than 1 cm and temporal scales ranging from seconds to days, oxygen sensitivity is likely to play a role. Additionally, electron donor availabilities are also quite variable; for example, sulfide concent ...
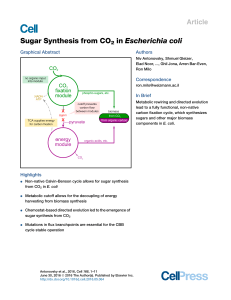
Sugar Synthesis from CO2 in Escherichia coli
... sugar as a feedstock (Galanie et al., 2015; Yim et al., 2011). However, efforts to synthesize sugar from inorganic CO2 by introducing a non-native carbon fixation cycle have never been successful. Carbohydrate biosynthesis through carbon fixation in E. coli would not only open exciting avenues to di ...
... sugar as a feedstock (Galanie et al., 2015; Yim et al., 2011). However, efforts to synthesize sugar from inorganic CO2 by introducing a non-native carbon fixation cycle have never been successful. Carbohydrate biosynthesis through carbon fixation in E. coli would not only open exciting avenues to di ...
pdf link - Aaron Diefendorf
... respectively. Even though both classes of terpenoids have long been recognized as plant biomarkers, their potential use as phylogenetically specific d13C proxies remains largely unexplored. Little is known of how terpenoid abundance and carbon isotope composition vary either with plant phylogenetic p ...
... respectively. Even though both classes of terpenoids have long been recognized as plant biomarkers, their potential use as phylogenetically specific d13C proxies remains largely unexplored. Little is known of how terpenoid abundance and carbon isotope composition vary either with plant phylogenetic p ...
STUDY GUIDE
... MAIN IDEA: Hydrocarbons with multiple bonds are more reactive than alkanes and participate in addition reactions in which atoms from one molecule are added to another molecule. Addition reactions include hydrogenation, halogenation, hydrohalogenation, and hydration. Markovnikov’s rule states that, ...
... MAIN IDEA: Hydrocarbons with multiple bonds are more reactive than alkanes and participate in addition reactions in which atoms from one molecule are added to another molecule. Addition reactions include hydrogenation, halogenation, hydrohalogenation, and hydration. Markovnikov’s rule states that, ...
Chapter 4 Carbon and the Molecular Diversity of Life
... Chapter 4 Carbon and the Molecular Diversity of Life All organisms are composed mostly of chemical structures based on the element carbon. This chapter builds upon information and concepts introduced in Chapters 2 and 3 and extends the descriptions and analysis to more detailed consideration of the ...
... Chapter 4 Carbon and the Molecular Diversity of Life All organisms are composed mostly of chemical structures based on the element carbon. This chapter builds upon information and concepts introduced in Chapters 2 and 3 and extends the descriptions and analysis to more detailed consideration of the ...
Print this article - Journals at the University of Arizona
... same individual. Finding such differences would therefore provide a biomarker for the presence of reservoir carbon and hence allow us to identify the presence of multiple carbon reservoirs in human bone, and to do so without recourse to the dating of associated materials (if possible). This hypothes ...
... same individual. Finding such differences would therefore provide a biomarker for the presence of reservoir carbon and hence allow us to identify the presence of multiple carbon reservoirs in human bone, and to do so without recourse to the dating of associated materials (if possible). This hypothes ...
Impact of carbon sources on growth and oxalate - The Keep
... was also stimulated by pyruvate. Other carbon sources appeared to be stimulatory but the range of growth often observed for a given isolate was large, thus making comparisons within isolates difficult. In ...
... was also stimulated by pyruvate. Other carbon sources appeared to be stimulatory but the range of growth often observed for a given isolate was large, thus making comparisons within isolates difficult. In ...
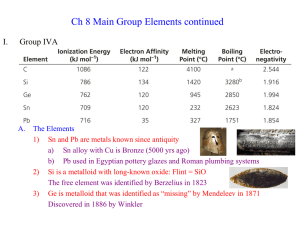
P-BLOCK ELEMENTS
... (f ) Higher ionisation enthalpy Heavier elements do not form p-p bond because their atomic orbitals are very large and diffuse to have effective overlapping. 5.2.4 Allotropes of Carbon ...
... (f ) Higher ionisation enthalpy Heavier elements do not form p-p bond because their atomic orbitals are very large and diffuse to have effective overlapping. 5.2.4 Allotropes of Carbon ...
(cobalamin)-dependent enzymes
... tailoring. Indeed, it has the most complex structure of any biological cofactor, requiring more than 20 genes for its biosynthesis [6]. At the heart of the coenzyme is the cobalt atom that is chelated by a macrocyclic ring called a corrin. The corrin ring is smaller than a porphyrin ring due to the ...
... tailoring. Indeed, it has the most complex structure of any biological cofactor, requiring more than 20 genes for its biosynthesis [6]. At the heart of the coenzyme is the cobalt atom that is chelated by a macrocyclic ring called a corrin. The corrin ring is smaller than a porphyrin ring due to the ...
Assigning and Using Oxidation Numbers in Biochemistry Lecture
... most important. These processes can be analyzed quantitatively by assigning redox numbers to these heteroatoms using the rules given above. The reduction of a disulfide bond to two thiol groups is a 2-electron process. An example of this is the reduction of glutathione disulfide (GCH2SSCH2G) to two ...
... most important. These processes can be analyzed quantitatively by assigning redox numbers to these heteroatoms using the rules given above. The reduction of a disulfide bond to two thiol groups is a 2-electron process. An example of this is the reduction of glutathione disulfide (GCH2SSCH2G) to two ...
The evolution of photosynthesis.again?
... enzymes and comparing them, leading to the conclusion that carbon fixation in general is basic to life and has arisen multiple times. Certainly, the evolutionary pressure to evolve new pathways for carbon fixation would have been present early in evolution. From knowledge about planetary systems and ...
... enzymes and comparing them, leading to the conclusion that carbon fixation in general is basic to life and has arisen multiple times. Certainly, the evolutionary pressure to evolve new pathways for carbon fixation would have been present early in evolution. From knowledge about planetary systems and ...
Growth of E. coli BL21 in minimal media with different
... Abstract Escherichia coli strain BL21 is commonly used as a host strain for protein expression and purification. For structural analysis, proteins are frequently isotopically labeled with deuterium (2H), 13C, or 15N by growing E. coli cultures in a medium containing the appropriate isotope. When lar ...
... Abstract Escherichia coli strain BL21 is commonly used as a host strain for protein expression and purification. For structural analysis, proteins are frequently isotopically labeled with deuterium (2H), 13C, or 15N by growing E. coli cultures in a medium containing the appropriate isotope. When lar ...
InorgCh8.2
... 1) Discovery in 1766 by Cavendish a) Nonreactive, leftover fraction of air (after N2, O2, CO2 all reacted) b) Not separated from each other 2) He discovered in 1868 by Locklear and Frankland a) New emission line observed during a solar eclipse b) Helios = Greek for “sun” c) Rare on Earth (Dexter, Ka ...
... 1) Discovery in 1766 by Cavendish a) Nonreactive, leftover fraction of air (after N2, O2, CO2 all reacted) b) Not separated from each other 2) He discovered in 1868 by Locklear and Frankland a) New emission line observed during a solar eclipse b) Helios = Greek for “sun” c) Rare on Earth (Dexter, Ka ...
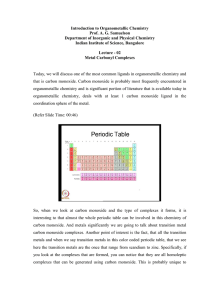
Pdf - Text of NPTEL IIT Video Lectures
... We will see in a moment why this is the case; all of them are capable of forming homoleptic or M C O n complexes. So, these are complexes, which have M C O n and only 1 ligand is formed in the coordination sphere of the metal. Let us just take a look at the systems that we have here on this table, y ...
... We will see in a moment why this is the case; all of them are capable of forming homoleptic or M C O n complexes. So, these are complexes, which have M C O n and only 1 ligand is formed in the coordination sphere of the metal. Let us just take a look at the systems that we have here on this table, y ...
Pdf - Text of NPTEL IIT Video Lectures
... compounds. Since, the chemistry is extensive; we had to split this into 2 lectures. In the first lecture, we covered the fact that most metal carbonyl compounds are in fact most metal carbonyl compounds are in fact typically 18 electron systems. So, they tend to be 18 electron complexes. They also a ...
... compounds. Since, the chemistry is extensive; we had to split this into 2 lectures. In the first lecture, we covered the fact that most metal carbonyl compounds are in fact most metal carbonyl compounds are in fact typically 18 electron systems. So, they tend to be 18 electron complexes. They also a ...
Chapter 15: Aldehyde and Ketones In this chapter, we discuss the
... major way. A carbon oxygen double bond is more polar, and a carboncarbon single bond is less polar. The electronegativity (Section 5.9) of oxygen (3.5) is much greater than that of carbon (2.5). Hence the carbonoxygen double bond has more electrons and is more polarized, the oxygen atom acquiring a ...
... major way. A carbon oxygen double bond is more polar, and a carboncarbon single bond is less polar. The electronegativity (Section 5.9) of oxygen (3.5) is much greater than that of carbon (2.5). Hence the carbonoxygen double bond has more electrons and is more polarized, the oxygen atom acquiring a ...
Mixotrophic and photoheterotrophic metabolism in
... evolution rates by two- to threefold compared with nitrogen-fixing conditions, while the changes of the quantum yields of PSII were much less significant (10– 30 %). Gene expression in the carbon fixation pathway was also determined (Fig. 4). RT-PCR results indicated that two key enzymes in the Calv ...
... evolution rates by two- to threefold compared with nitrogen-fixing conditions, while the changes of the quantum yields of PSII were much less significant (10– 30 %). Gene expression in the carbon fixation pathway was also determined (Fig. 4). RT-PCR results indicated that two key enzymes in the Calv ...
Principles of Drug Action I, Winter 2005 ISOMERISM AND
... number of the next adjacent atom determines the priority. If two or more atoms connected to the second atom are the same, the third atom determines the priority, etc. 3. All atoms except hydrogen are formally given a valence of 4. When the actual valence is less than 4 (N, O), phantom atoms are assi ...
... number of the next adjacent atom determines the priority. If two or more atoms connected to the second atom are the same, the third atom determines the priority, etc. 3. All atoms except hydrogen are formally given a valence of 4. When the actual valence is less than 4 (N, O), phantom atoms are assi ...
Organic Molecules
... structures containing thousands of atoms! Although carbon is present in all organic compounds, other elements such as hydrogen (H), oxygen (O), nitrogen (N), sulfur (S) and phosphorus (P) are also common in these molecules. Until the early nineteenth century, chemists had managed to make many simple ...
... structures containing thousands of atoms! Although carbon is present in all organic compounds, other elements such as hydrogen (H), oxygen (O), nitrogen (N), sulfur (S) and phosphorus (P) are also common in these molecules. Until the early nineteenth century, chemists had managed to make many simple ...
Carbon

Carbon (from Latin: carbo ""coal"") is a chemical element with symbol C and atomic number 6. On the Periodic table, it is the first (row 2) of six elements in column (group) 14, which have in common the composition of their outer electron shell. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. There are three naturally occurring isotopes, with 12C and 13C being stable, while 14C is radioactive, decaying with a half-life of about 5,730 years. Carbon is one of the few elements known since antiquity.Carbon is the 15th most abundant element in the Earth's crust, and the fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. It is present in all forms of carbon-based life, and in the human body carbon is the second most abundant element by mass (about 18.5%) after oxygen. This abundance, together with the unique diversity of organic compounds and their unusual polymer-forming ability at the temperatures commonly encountered on Earth, make this element the chemical basis of all known life.The atoms of carbon can be bonded together in different ways: allotropes of carbon. The best known are graphite, diamond, and amorphous carbon. The physical properties of carbon vary widely with the allotropic form. For example, graphite is opaque and black, while diamond is highly transparent. Graphite is soft enough to form a streak on paper (hence its name, from the Greek word ""γράφω"" which means ""to write""), while diamond is the hardest naturally-occurring material known. Graphite is a very good conductor, while diamond has a very low electrical conductivity. Under normal conditions, diamond, carbon nanotubes, and graphene have the highest thermal conductivities of all known materials. All carbon allotropes are solids under normal conditions, with graphite being the most thermodynamically stable form. They are chemically resistant and require high temperature to react even with oxygen.The most common oxidation state of carbon in inorganic compounds is +4, while +2 is found in carbon monoxide and other transition metal carbonyl complexes. The largest sources of inorganic carbon are limestones, dolomites and carbon dioxide, but significant quantities occur in organic deposits of coal, peat, oil and methane clathrates. Carbon forms a vast number of compounds, more than any other element, with almost ten million compounds described to date, which in turn are a tiny fraction of such compounds that are theoretically possible under standard conditions.