CH # 2-3 - SwampBiology
... Proteins are macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen. Proteins are polymers of molecules called amino acids. Proteins perform many varied functions, such as controlling the rate of reactions and regulating cell processes, forming cellular structures, transporting ...
... Proteins are macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen. Proteins are polymers of molecules called amino acids. Proteins perform many varied functions, such as controlling the rate of reactions and regulating cell processes, forming cellular structures, transporting ...
CH # 2-3
... Proteins are macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen. Proteins are polymers of molecules called amino acids. Proteins perform many varied functions, such as controlling the rate of reactions and regulating cell processes, forming cellular structures, transporting ...
... Proteins are macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen. Proteins are polymers of molecules called amino acids. Proteins perform many varied functions, such as controlling the rate of reactions and regulating cell processes, forming cellular structures, transporting ...
Lesson Overview
... Proteins are macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen. Proteins are polymers of molecules called amino acids. Proteins perform many varied functions, such as controlling the rate of reactions and regulating cell processes, forming cellular structures, transporting ...
... Proteins are macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen. Proteins are polymers of molecules called amino acids. Proteins perform many varied functions, such as controlling the rate of reactions and regulating cell processes, forming cellular structures, transporting ...
File
... Where are mitochondria found? What is anaerobic respiration? What is another name for anaerobic respiration? Which produces more energy? Aerobic or anaerobic respiration? ...
... Where are mitochondria found? What is anaerobic respiration? What is another name for anaerobic respiration? Which produces more energy? Aerobic or anaerobic respiration? ...
The chemical building blocks of life Carbon
... also with H, O and N This is because the bonds C-C, C-H, C-O, and C-N have similar energies For instance, N can replace C in ring structures ...
... also with H, O and N This is because the bonds C-C, C-H, C-O, and C-N have similar energies For instance, N can replace C in ring structures ...
Carbon transfer from dissolved organic carbon to the cladoceran
... Hence carbon stable isotopic analysis of their signature PLFA can be used to study the source of carbon they assimilated (PLFA-based stable isotope probing, PLFA-SIP), and is applied in our experiment. Each mesocosm contained 100 L surface water from the shore of Fuxian Lake in China (24°210 2800 –2 ...
... Hence carbon stable isotopic analysis of their signature PLFA can be used to study the source of carbon they assimilated (PLFA-based stable isotope probing, PLFA-SIP), and is applied in our experiment. Each mesocosm contained 100 L surface water from the shore of Fuxian Lake in China (24°210 2800 –2 ...
Carbon Compounds 2-3 Foldable Instructions
... Carbon has 4 valence electrons which enable it to form strong covalent bonds with other atoms. ...
... Carbon has 4 valence electrons which enable it to form strong covalent bonds with other atoms. ...
Study Island Copyright © 2012 Study Island
... 15. Which of the following is true about a compound and its elements? A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are ...
... 15. Which of the following is true about a compound and its elements? A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are ...
Lesson Overview
... In the early 1800s, many chemists called the compounds created by organisms “organic,” believing they were fundamentally different from compounds in nonliving things. We now understand that the principles governing the chemistry of living and nonliving things are the same, but the term “organic chem ...
... In the early 1800s, many chemists called the compounds created by organisms “organic,” believing they were fundamentally different from compounds in nonliving things. We now understand that the principles governing the chemistry of living and nonliving things are the same, but the term “organic chem ...
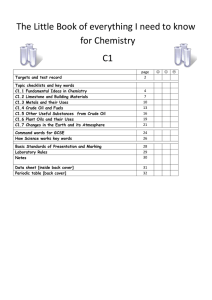
The Big book of C1 chemistry
... Crude oil is a mixture of a very large number of compounds. A mixture consists of two or more elements or compounds not chemically combined together. The chemical properties of each substance in the mixture are unchanged. It is possible to separate the substances in a mixture by physical methods inc ...
... Crude oil is a mixture of a very large number of compounds. A mixture consists of two or more elements or compounds not chemically combined together. The chemical properties of each substance in the mixture are unchanged. It is possible to separate the substances in a mixture by physical methods inc ...
Chemistry 199 - Oregon State chemistry
... The carboxylic acid group on the right is still in tact and will react with an alcohol group from another HOCH2OH molecule. The alcohol group on the left is still in tact and will react with a carboxylic acid group from another HOOCCH2CH2CH2CH2COOH molecule. The polymer may be represented as: O O ...
... The carboxylic acid group on the right is still in tact and will react with an alcohol group from another HOCH2OH molecule. The alcohol group on the left is still in tact and will react with a carboxylic acid group from another HOOCCH2CH2CH2CH2COOH molecule. The polymer may be represented as: O O ...
The Calvin Cycle
... •Requires ATP and NADPH (reducing power) •Requires 9 ATP and 6 NADPH (which are regenerated by light reactions) ...
... •Requires ATP and NADPH (reducing power) •Requires 9 ATP and 6 NADPH (which are regenerated by light reactions) ...
Free Response – due Friday, Oct 2 – typed – single
... monosaccharide, lipid, fatty acid, semi-permeable membrane, protein, amino acid, nucleic acid, nucleotide, genetic, energy. ...
... monosaccharide, lipid, fatty acid, semi-permeable membrane, protein, amino acid, nucleic acid, nucleotide, genetic, energy. ...
fo-Balancing Chemical Notes
... In a chemical reaction, atoms are NOT created or destroyed. What changes in a chemical reaction is the connections (bonds) between atoms. In the first reaction shown above, two hydrogen atoms (H) start out bonded to each other. During the course of the chemical reaction, this H-H bond breaks and a n ...
... In a chemical reaction, atoms are NOT created or destroyed. What changes in a chemical reaction is the connections (bonds) between atoms. In the first reaction shown above, two hydrogen atoms (H) start out bonded to each other. During the course of the chemical reaction, this H-H bond breaks and a n ...
Prentice Hall Biology
... chemical bonds that join atoms in compounds. The elements or compounds that enter into a chemical reaction are called reactants. The elements or compounds produced by a chemical reaction are called products. As wood burns, molecules of cellulose are broken down and combine with oxygen to form carbon ...
... chemical bonds that join atoms in compounds. The elements or compounds that enter into a chemical reaction are called reactants. The elements or compounds produced by a chemical reaction are called products. As wood burns, molecules of cellulose are broken down and combine with oxygen to form carbon ...
N.9 – Metabolic Changes of Drugs and Related
... Oxidation at Aliphatic and Alicyclic Carbon Atoms Alkyl or aliphatic carbon centers are subject to mixed function oxidation. Metabolic oxidation at the terminal methyl group often is referred to as ω-oxidation, and oxidation of the penultimate carbon atom (i.e., next-to-the-last carbon) is called ...
... Oxidation at Aliphatic and Alicyclic Carbon Atoms Alkyl or aliphatic carbon centers are subject to mixed function oxidation. Metabolic oxidation at the terminal methyl group often is referred to as ω-oxidation, and oxidation of the penultimate carbon atom (i.e., next-to-the-last carbon) is called ...
II.I Corhon compounds lI.2 Hydrocorhons
... Hydrocarborrs are compounds whose molecular structures c:ontainonly hydrogen and carbon The simplest hydrocarbons are the alkanes-hydro carbons that contain only single coualentbonds.The smallest organic molecule is the alkane called methane (cHn;. Methane, a gas at standard temperature and pressure ...
... Hydrocarborrs are compounds whose molecular structures c:ontainonly hydrogen and carbon The simplest hydrocarbons are the alkanes-hydro carbons that contain only single coualentbonds.The smallest organic molecule is the alkane called methane (cHn;. Methane, a gas at standard temperature and pressure ...
formula - eduBuzz.org
... • What is the rule for working out the number of hydrogen atoms in an alkane molecule? • What is a homologous series, and why are the alkanes an example of one? • What is the general formula for the alkanes? • Why are the products of combustion the same for every alkane? ...
... • What is the rule for working out the number of hydrogen atoms in an alkane molecule? • What is a homologous series, and why are the alkanes an example of one? • What is the general formula for the alkanes? • Why are the products of combustion the same for every alkane? ...
DO NOW
... wear, the cosmetics you use and the gasoline that fuels your car. Carbon is the sixth most abundant element in the universe. ...
... wear, the cosmetics you use and the gasoline that fuels your car. Carbon is the sixth most abundant element in the universe. ...
Chapter 3 Biochemistry Section 1 – Carbon Compounds Section 2
... 6. Describe how the three major types of lipids differ in structure from one another. 7. What are the functions of the two types of nucleic acids. ...
... 6. Describe how the three major types of lipids differ in structure from one another. 7. What are the functions of the two types of nucleic acids. ...
Slide 1
... Carbon atoms have four valence electrons, allowing them to form strong covalent bonds with many other elements, including hydrogen, oxygen, phosphorus, sulfur, and nitrogen. Living organisms are made up of molecules that consist of carbon and these other elements. Organic compounds-contain carbon (l ...
... Carbon atoms have four valence electrons, allowing them to form strong covalent bonds with many other elements, including hydrogen, oxygen, phosphorus, sulfur, and nitrogen. Living organisms are made up of molecules that consist of carbon and these other elements. Organic compounds-contain carbon (l ...
Empirical is the
... the empirical formula of urea ( Remember this is where you will use the AMU on the periodic table to convert to moles, then you divide by the lowest value to set it to one, then divide the others by that number to find the ratios between the other ones: [ 1.121 g N ( 1 mole of N/ 14.00 g) = 0.008 mo ...
... the empirical formula of urea ( Remember this is where you will use the AMU on the periodic table to convert to moles, then you divide by the lowest value to set it to one, then divide the others by that number to find the ratios between the other ones: [ 1.121 g N ( 1 mole of N/ 14.00 g) = 0.008 mo ...
Use of Reduced Carbon Compounds
... With sources of the basic chemical building blocks: C, N, O, S, & P most bacteria can synthesize all 20 commonly appearing amino acids the 5 nucleic acid bases as well as the lipids and simple sugars --- this broad spectrum synthetic ability is what has freed the more complex life forms from much of ...
... With sources of the basic chemical building blocks: C, N, O, S, & P most bacteria can synthesize all 20 commonly appearing amino acids the 5 nucleic acid bases as well as the lipids and simple sugars --- this broad spectrum synthetic ability is what has freed the more complex life forms from much of ...
qp13 - Smart Edu Hub
... 24 Metal X reacts violently with water. Metal Y reacts slowly with steam. Metal Z does not react with dilute hydrochloric acid. What is the correct order of reactivity of these metals, most reactive first? A ...
... 24 Metal X reacts violently with water. Metal Y reacts slowly with steam. Metal Z does not react with dilute hydrochloric acid. What is the correct order of reactivity of these metals, most reactive first? A ...
Carbon

Carbon (from Latin: carbo ""coal"") is a chemical element with symbol C and atomic number 6. On the Periodic table, it is the first (row 2) of six elements in column (group) 14, which have in common the composition of their outer electron shell. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. There are three naturally occurring isotopes, with 12C and 13C being stable, while 14C is radioactive, decaying with a half-life of about 5,730 years. Carbon is one of the few elements known since antiquity.Carbon is the 15th most abundant element in the Earth's crust, and the fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. It is present in all forms of carbon-based life, and in the human body carbon is the second most abundant element by mass (about 18.5%) after oxygen. This abundance, together with the unique diversity of organic compounds and their unusual polymer-forming ability at the temperatures commonly encountered on Earth, make this element the chemical basis of all known life.The atoms of carbon can be bonded together in different ways: allotropes of carbon. The best known are graphite, diamond, and amorphous carbon. The physical properties of carbon vary widely with the allotropic form. For example, graphite is opaque and black, while diamond is highly transparent. Graphite is soft enough to form a streak on paper (hence its name, from the Greek word ""γράφω"" which means ""to write""), while diamond is the hardest naturally-occurring material known. Graphite is a very good conductor, while diamond has a very low electrical conductivity. Under normal conditions, diamond, carbon nanotubes, and graphene have the highest thermal conductivities of all known materials. All carbon allotropes are solids under normal conditions, with graphite being the most thermodynamically stable form. They are chemically resistant and require high temperature to react even with oxygen.The most common oxidation state of carbon in inorganic compounds is +4, while +2 is found in carbon monoxide and other transition metal carbonyl complexes. The largest sources of inorganic carbon are limestones, dolomites and carbon dioxide, but significant quantities occur in organic deposits of coal, peat, oil and methane clathrates. Carbon forms a vast number of compounds, more than any other element, with almost ten million compounds described to date, which in turn are a tiny fraction of such compounds that are theoretically possible under standard conditions.