Metabolism of amino acids, porphyrins
... Mechanism of the binding of ubiquitin to target protein E1 - ubiquitin-activating enzyme (attachment of ubiquitin to a sulfhydryl group of E1; ATP-driven reaction) E2 - ubiquitin-conjugating enzyme (ubiquitin is shuttled to a sulfhydryl group of E2) E3 - ubiquitin-protein ligase (transfer of ubiqui ...
... Mechanism of the binding of ubiquitin to target protein E1 - ubiquitin-activating enzyme (attachment of ubiquitin to a sulfhydryl group of E1; ATP-driven reaction) E2 - ubiquitin-conjugating enzyme (ubiquitin is shuttled to a sulfhydryl group of E2) E3 - ubiquitin-protein ligase (transfer of ubiqui ...
Gene Section USP15 (ubiquitin specific peptidase 15) Atlas of Genetics and Cytogenetics
... activating, E2 conjugating and E3 ligase enzymes. Ubiquitin contains seven lysine residues (K6, K11, K27, K29, K33, K48 and K63), which allow polyubiquitin chains to assemble through alternative isopeptide bond linkages. In addition, linear ubiquitin chains may be assembled through the amino-terminu ...
... activating, E2 conjugating and E3 ligase enzymes. Ubiquitin contains seven lysine residues (K6, K11, K27, K29, K33, K48 and K63), which allow polyubiquitin chains to assemble through alternative isopeptide bond linkages. In addition, linear ubiquitin chains may be assembled through the amino-terminu ...
Lect21.RegulProtTurnover
... in a cell, and changes in either rate can serve as means to regulate a protein’s concentration in the cell. ...
... in a cell, and changes in either rate can serve as means to regulate a protein’s concentration in the cell. ...
Capturing denaturing proteins * Small Heat Shock Protein substrate
... Elizabeth Vierling and Indu Santhanagopalan Protein aggregation resulting from stress, disease or mutation poses a major threat to all cells. The ubiquitous small heat shock proteins (sHSPs) act as molecular chaperones to prevent irreversible protein aggregation and are significant components of the ...
... Elizabeth Vierling and Indu Santhanagopalan Protein aggregation resulting from stress, disease or mutation poses a major threat to all cells. The ubiquitous small heat shock proteins (sHSPs) act as molecular chaperones to prevent irreversible protein aggregation and are significant components of the ...
Fluorescent High-Throughput Conjugation and Deconjugation
... A similar principle has been applied to the ubiquitin-like proteins including SUMO-1/2/3 and NEDD8. In this particular example, fluorescein labeled SUMO-1 is conjugated to the Sumo activating enzyme (Aos1/Uba2) via a thioester bond in an ATP-dependent reaction. The charged SUMO activating enzyme the ...
... A similar principle has been applied to the ubiquitin-like proteins including SUMO-1/2/3 and NEDD8. In this particular example, fluorescein labeled SUMO-1 is conjugated to the Sumo activating enzyme (Aos1/Uba2) via a thioester bond in an ATP-dependent reaction. The charged SUMO activating enzyme the ...
The Ubiquitin Proteosome pathway
... Several complex processes are mediated via degradation or processing of specific proteins. Aberrations in these systems associates with pathogenic ...
... Several complex processes are mediated via degradation or processing of specific proteins. Aberrations in these systems associates with pathogenic ...
lecture 17
... account for 1-15% of cell volume (most abundant in liver and kidney) Most lysosomal enzymes are transported to lysosomes through recognition by receptors for mannose-6-phosphate. Lysosomal enzymes are synthesized like proteins destined to be secreted or for residence on the plasma membrane but a ...
... account for 1-15% of cell volume (most abundant in liver and kidney) Most lysosomal enzymes are transported to lysosomes through recognition by receptors for mannose-6-phosphate. Lysosomal enzymes are synthesized like proteins destined to be secreted or for residence on the plasma membrane but a ...
GREAT CHANGES IN HEALTH CARE IN THE PAST 40 YEARS
... ubiquitin tagging hypothesis 1980- 1990: Identification of enzymes involved in ubiquitin-mediated protein degradation 1990-present: Roles of the ubiquitin system in the control of cell division ...
... ubiquitin tagging hypothesis 1980- 1990: Identification of enzymes involved in ubiquitin-mediated protein degradation 1990-present: Roles of the ubiquitin system in the control of cell division ...
Ubiquitin Found to Mark Pathogen-Containing Vacuoles
... vacuoles for destruction by using a molecule called ubiquitin, commonly known as the "kiss of death." The finding could lead to new therapeutic strategies to boost the immune system's response to the pathogens responsible for a long list of human ailments, including tuberculosis, salmonella, chlamyd ...
... vacuoles for destruction by using a molecule called ubiquitin, commonly known as the "kiss of death." The finding could lead to new therapeutic strategies to boost the immune system's response to the pathogens responsible for a long list of human ailments, including tuberculosis, salmonella, chlamyd ...
Post-translation
... Proteins build up all living things: plants, animals and therefore us humans. In the past few decades biochemistry has come a long way towards explaining how the cell produces all its various proteins(at least five Nobel Prizes have been awarded in this area). But as to the breaking down of protein ...
... Proteins build up all living things: plants, animals and therefore us humans. In the past few decades biochemistry has come a long way towards explaining how the cell produces all its various proteins(at least five Nobel Prizes have been awarded in this area). But as to the breaking down of protein ...
Moonlighting and pleiotropy among regulators of the degradation
... distinct cellular pathways. These features are hard to discriminate, especially in orphan proteins that carry out alone several functions. However, multisubunit enzymes can be good model since a mutation in single subunit might cause to both complexdependent and unique (complex-independent), phenoty ...
... distinct cellular pathways. These features are hard to discriminate, especially in orphan proteins that carry out alone several functions. However, multisubunit enzymes can be good model since a mutation in single subunit might cause to both complexdependent and unique (complex-independent), phenoty ...
Post-translational Modification by Ubiquitin and
... Residues 22-97 of Sumo-1 is 18% identical and 48% homologous to human ubiquitin Sumo-2 is a 95 aa protein 46% identical and 66% homologous to Sumo-1 Sumo-3 is a 103 aa protein which is 97% identical to Sumo-2 in the ubiquitin homology domain ...
... Residues 22-97 of Sumo-1 is 18% identical and 48% homologous to human ubiquitin Sumo-2 is a 95 aa protein 46% identical and 66% homologous to Sumo-1 Sumo-3 is a 103 aa protein which is 97% identical to Sumo-2 in the ubiquitin homology domain ...
Introduction
... was histone 2a and the other protein was identified as ubiquitin, a 76-residue ubiquitously (hence its name) expressed protein of unknown function that was described (as a free protein) by Gideon Goldstein and colleagues in 1975. In 1980, Keith Wilkinson, Michael Urban and Arthur Haas showed that AP ...
... was histone 2a and the other protein was identified as ubiquitin, a 76-residue ubiquitously (hence its name) expressed protein of unknown function that was described (as a free protein) by Gideon Goldstein and colleagues in 1975. In 1980, Keith Wilkinson, Michael Urban and Arthur Haas showed that AP ...
Leukaemia Section t(7;21)(p22;q22) Atlas of Genetics and Cytogenetics in Oncology and Haematology
... USP42 (ubiquitin specific protease 42), belongs to the ubiquitin specific protease family. Ubiquitins are highly conserved proteins. Ubiquitins target proteins for degradation in the proteasome. Some USPs, however, act in the opposite reaction. These ubiquitin specific proteases (cysteine proteases) ...
... USP42 (ubiquitin specific protease 42), belongs to the ubiquitin specific protease family. Ubiquitins are highly conserved proteins. Ubiquitins target proteins for degradation in the proteasome. Some USPs, however, act in the opposite reaction. These ubiquitin specific proteases (cysteine proteases) ...
Abstract I. DLC1 encodes a RhoA GTPase
... decrease in NSCLC anchorage-dependent and -independent proliferation. In summary, we identify a posttranslational mechanism for loss of DLC1 and a linkage between CRL4A-FBXW5-associated oncogenesis and regulation of RhoA signaling. II. Defining the full complement of substrates for each ubiquitin li ...
... decrease in NSCLC anchorage-dependent and -independent proliferation. In summary, we identify a posttranslational mechanism for loss of DLC1 and a linkage between CRL4A-FBXW5-associated oncogenesis and regulation of RhoA signaling. II. Defining the full complement of substrates for each ubiquitin li ...
Student CSE paper
... ubiquitin/proteasome pathway involves two major steps: ubiquitination followed by degradation. A de-ubiquitination step also plays an important role in this pathway 2 . Ubiquitin (Ub) is a small peptide, consisting of 76 amino acids and is abundant in all eukaryotic cells 2. Covalent attachment of u ...
... ubiquitin/proteasome pathway involves two major steps: ubiquitination followed by degradation. A de-ubiquitination step also plays an important role in this pathway 2 . Ubiquitin (Ub) is a small peptide, consisting of 76 amino acids and is abundant in all eukaryotic cells 2. Covalent attachment of u ...
Document
... molecules to a protein substrate. 2. Degradation of the tagged protein by the 26s proteasome. (ubiquitin is recycled) ...
... molecules to a protein substrate. 2. Degradation of the tagged protein by the 26s proteasome. (ubiquitin is recycled) ...
Key concepts_Protein processing and modification
... Phosphorylation occurs primarily at serine and threonine residues. Acetylation of lysine residues is mostly used to modulate interactions between proteins or between proteins and nucleic acids. Glycosylation involves the addition of saccharide or polysaccharide moieties to serine or threonine, in O- ...
... Phosphorylation occurs primarily at serine and threonine residues. Acetylation of lysine residues is mostly used to modulate interactions between proteins or between proteins and nucleic acids. Glycosylation involves the addition of saccharide or polysaccharide moieties to serine or threonine, in O- ...
Ubiquitin
... - one of its functions: it directs protein recycling - can attach to proteins and label them for destruction. - discovery won the Nobel Prize for chemistry in 2004 ...
... - one of its functions: it directs protein recycling - can attach to proteins and label them for destruction. - discovery won the Nobel Prize for chemistry in 2004 ...
Ubiquitin

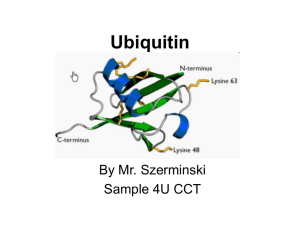
Ubiquitin is a small (8.5 kDa) regulatory protein that has been found in almost all tissues (ubiquitously) of eukaryotic organisms. It was discovered in 1975 by Goldstein and further characterized throughout the 1970s and 1980s. There are four genes in the human genome that produce ubiquitin: UBB, UBC, UBA52 and RPS27A.The addition of ubiquitin to a substrate protein is called ubiquitination or ubiquitylation. Ubiquitination can affect proteins in many ways: It can signal for their degradation via the proteasome, alter their cellular location, affect their activity, and promote or prevent protein interactions. Ubiquitination is carried out in three main steps: activation, conjugation, and ligation, performed by ubiquitin-activating enzymes (E1s), ubiquitin-conjugating enzymes (E2s), and ubiquitin ligases (E3s), respectively. The result of this sequential cascade binds ubiquitin to lysine residues on the protein substrate via an isopeptide bond or to the amino group of the protein's N-terminus via a peptide bond.The protein modifications can be either a single ubiquitin protein (monoubiquitination) or a chain of ubiquitin (polyubiquitination). The ubiquitination bonds are always formed with one of the seven lysine residues from the ubiquitin molecule. These 'linking' lysines are represented by a ""K"" (which is the one-letter amino acid notation of lysine) and a number, referring to its position in the ubiquitin molecule. First, a ubiquitin molecule is bonded by its C-terminus to a specific lysine residue (e.g. K48, K29, K63,...) on the target protein. Poly-ubiquitination occurs when the C-terminus of another ubiquitin, will be linked again to a lysine residue (for example again K48 or K29) on the previously added ubiquitin molecule, forming a chain. This process repeats several times, leading to the addition of several ubiquitins. Only poly-ubiquitination on defined lysines, mostly on K48 and K29, is related to degradation with the proteasome (referred to as the ""molecular kiss of death""), while other polyubiquitinations (e.g. on K63, K11, K6) and monoubiquitinations may regulate processes such as endocytic trafficking, inflammation, translation and DNA repair.Lysine 48-linked chains have been much-studied. They are the forms of chains that signal proteins to the proteasome, which destroys and recycles proteins. This discovery won the Nobel Prize for chemistry in 2004.