Exam 3 - Organic Chemistry at CU Boulder
... ester. The reaction starts with the ester in an alkoxide/alcohol solution and is worked up with acid to form the neutral β–keto ester product. Show the curved arrow mechanism for the Claisen condensation of ethyl ethanoate treated ethoxide ion. In each step, draw only the species that react in that ...
... ester. The reaction starts with the ester in an alkoxide/alcohol solution and is worked up with acid to form the neutral β–keto ester product. Show the curved arrow mechanism for the Claisen condensation of ethyl ethanoate treated ethoxide ion. In each step, draw only the species that react in that ...
Chapter 20 - people.vcu.edu
... o What would happen if you put the following reagents into a vessel? ...
... o What would happen if you put the following reagents into a vessel? ...
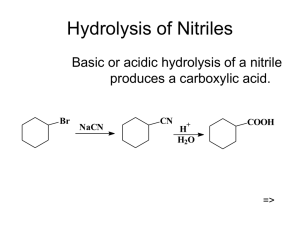
Hydrolysis of Nitriles
... An activated form of the carboxylic acid. Chloride is a good leaving group, so undergoes acyl substitution easily. To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
... An activated form of the carboxylic acid. Chloride is a good leaving group, so undergoes acyl substitution easily. To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034 PART-A
... 25. (a) Explain the addition-elimination reaction mechanism of a α, β-unsaturated ketone. ...
... 25. (a) Explain the addition-elimination reaction mechanism of a α, β-unsaturated ketone. ...
Workshop 9
... mechanisms are well established. In other cases they may be speculative and are likely to change as more data become available. Mechanisms map the path by which the reactants change into products and the movement of electrons that accompanies this change. They also show how reactants come together, ...
... mechanisms are well established. In other cases they may be speculative and are likely to change as more data become available. Mechanisms map the path by which the reactants change into products and the movement of electrons that accompanies this change. They also show how reactants come together, ...
Chapter 9. Addition Reactions of Alkenes
... i. cold, dilute, aqueous alkaline KMnO4 (yields only fair) ii. OsO4 (yields are good) iii. syn-addition stereochemistry i. epoxidation: addition of O to yield epoxides i. typical to use peracids like peracetic acid ii. basic hydrolysis causes net anti-dihydroxylation 2. hydrogenation: addition of H2 ...
... i. cold, dilute, aqueous alkaline KMnO4 (yields only fair) ii. OsO4 (yields are good) iii. syn-addition stereochemistry i. epoxidation: addition of O to yield epoxides i. typical to use peracids like peracetic acid ii. basic hydrolysis causes net anti-dihydroxylation 2. hydrogenation: addition of H2 ...
Synthesis of a Family of Chiral Asymmetric Schiff - Blogs at H-SC
... Department of Chemistry, Hampden-Sydney College, Hampden-Sydney, VA 23943 Introduction Carbon-Carbon bond-forming reactions are essential synthetic methods for organic chemists. Condensation reactions of carbonyl compounds are an important class of such reactions. Chiral organometallic compounds hav ...
... Department of Chemistry, Hampden-Sydney College, Hampden-Sydney, VA 23943 Introduction Carbon-Carbon bond-forming reactions are essential synthetic methods for organic chemists. Condensation reactions of carbonyl compounds are an important class of such reactions. Chiral organometallic compounds hav ...
Carboxylic Acid Questions 1.-Arrange the following in order of (i
... LiAlH4 is a source of H- (a nucleophile) which functions as a reducing agent. ...
... LiAlH4 is a source of H- (a nucleophile) which functions as a reducing agent. ...
Slide 1
... They are stronger acids (pKa = 10) than alcohols, but do not exhibit hydrogen-binding ...
... They are stronger acids (pKa = 10) than alcohols, but do not exhibit hydrogen-binding ...
Study Guide for Exam 4 Chapter 17
... aldehydes and ketones, and the addition of alcohols to aldehydes and ketones. Know that reducing agents such as NaBH4 and LiAlH4 usually initiate reactions by providing hydride ions (H-) that bond with a carbonyl carbon (selective). From their structural formulas, identify hemi-acetals and aceta ...
... aldehydes and ketones, and the addition of alcohols to aldehydes and ketones. Know that reducing agents such as NaBH4 and LiAlH4 usually initiate reactions by providing hydride ions (H-) that bond with a carbonyl carbon (selective). From their structural formulas, identify hemi-acetals and aceta ...
Chapter 21: Carboxylic Acid Derivatives
... however this reaction is easy since the leaving group Cl- is a weaker base than NH2300 ...
... however this reaction is easy since the leaving group Cl- is a weaker base than NH2300 ...
Chapter 20: Carboxylic Acids
... Condensation of acids with amines (20-12) The reaction of carboxylic acids with amines is a simple acid/base reaction forming an ammonium salt. However, under more extreme conditions, when heat is applied it is possible to eliminate water from this salt and form amide. ...
... Condensation of acids with amines (20-12) The reaction of carboxylic acids with amines is a simple acid/base reaction forming an ammonium salt. However, under more extreme conditions, when heat is applied it is possible to eliminate water from this salt and form amide. ...
Organometallic Chemistry
... Chain-end control mechanism: the polymer chain determines the stereospecificity of the final polymer. ...
... Chain-end control mechanism: the polymer chain determines the stereospecificity of the final polymer. ...
CARBONYL COMPOUNDS ALDEHYDES AND KETONES
... The most common oxidation reaction of carbonyl compounds is the oxidation of aldehydes to carboxylic acids. A variety of oxidizing agents can be used, including CrO3, Na2Cr2O7, K2Cr2O7 and KMnO4. Aldehydes are also oxidized selectively in the presence of other functional groups using silver(I) oxide ...
... The most common oxidation reaction of carbonyl compounds is the oxidation of aldehydes to carboxylic acids. A variety of oxidizing agents can be used, including CrO3, Na2Cr2O7, K2Cr2O7 and KMnO4. Aldehydes are also oxidized selectively in the presence of other functional groups using silver(I) oxide ...
Preparation and Reaction of Carboxylic Acids - IDC
... In the second procedure the electrophilic halide is first transformed into a strongly nucleophilic metal derivative, and this adds to carbon dioxide (an electrophile). The initial product is a salt of the carboxylic acid, which must then be released by treatment with strong aqueous acid. ...
... In the second procedure the electrophilic halide is first transformed into a strongly nucleophilic metal derivative, and this adds to carbon dioxide (an electrophile). The initial product is a salt of the carboxylic acid, which must then be released by treatment with strong aqueous acid. ...
CH19 Aldehydes and Ketones
... Slightly different mechanisms in acid or base. Neutral vs. Negatively charged Nucleophile ...
... Slightly different mechanisms in acid or base. Neutral vs. Negatively charged Nucleophile ...
This is the first exam with targeted syntheses that you
... Most of the reactions of aldehydes and ketones in these chapters are nucleophilic addition reactions. The oxygen in C=O polarizes the bond. Therefore, while electrophilic addition (electrophile first, followed by nucleophile) was favored for the comparatively non-polar, electron-rich alkene, carbony ...
... Most of the reactions of aldehydes and ketones in these chapters are nucleophilic addition reactions. The oxygen in C=O polarizes the bond. Therefore, while electrophilic addition (electrophile first, followed by nucleophile) was favored for the comparatively non-polar, electron-rich alkene, carbony ...
Exam 3 Review
... What is the name for this alcohol / ether / aldehyde / ketone? Is an alcohol 1°, 2°, or 3°? Describe hydrogen bonding in alcohols, and compare alcohol polarity to ether polarity. What are the acid/base properties of alcohols? Rank these compounds in order of acidity. How are Grignard reagents prepar ...
... What is the name for this alcohol / ether / aldehyde / ketone? Is an alcohol 1°, 2°, or 3°? Describe hydrogen bonding in alcohols, and compare alcohol polarity to ether polarity. What are the acid/base properties of alcohols? Rank these compounds in order of acidity. How are Grignard reagents prepar ...
Chapter 8_part 1
... The Rate Determining step Step 1 is the rate determined step Formation of carbocation ...
... The Rate Determining step Step 1 is the rate determined step Formation of carbocation ...
carboxylic acids - La Salle University
... This is the work-up step, a simple acid/base reaction. Protonation of the carboxylate oxygen creates the carboxylic acid product from the intermediate complex. ...
... This is the work-up step, a simple acid/base reaction. Protonation of the carboxylate oxygen creates the carboxylic acid product from the intermediate complex. ...
CHAPTER 9 Further Reactions of Alcohols and the Chemistry of
... Conversion of Alcohols to Alkyl sulfonates creates a good leaving group for subsequent displacement by an anionic nucleophile ...
... Conversion of Alcohols to Alkyl sulfonates creates a good leaving group for subsequent displacement by an anionic nucleophile ...
Elimination reactions under acidic conditions
... 1. Provide a mechanism for these elimination reactions of alcohols under acidic conditions. ...
... 1. Provide a mechanism for these elimination reactions of alcohols under acidic conditions. ...
Lecture 9a - University of California, Los Angeles
... used in synthetic organic chemistry The main problem is that the use of protective groups usually adds two (or more) steps to the reaction sequence This generates additional waste It also decreases atom economy (=atoms used that are ...
... used in synthetic organic chemistry The main problem is that the use of protective groups usually adds two (or more) steps to the reaction sequence This generates additional waste It also decreases atom economy (=atoms used that are ...
Test 3 Test Skills/Competencies
... f. Amines (reversible aminol and imine formation, including cyclic aminols and imines, and the reverse reaction involving imine hydrolysis) Mechanisms: Be able to draw mechanisms for carbonyl reactions listed above, including the reverse reaction, including those involving rings. Major mechanisms in ...
... f. Amines (reversible aminol and imine formation, including cyclic aminols and imines, and the reverse reaction involving imine hydrolysis) Mechanisms: Be able to draw mechanisms for carbonyl reactions listed above, including the reverse reaction, including those involving rings. Major mechanisms in ...
Wolff rearrangement

The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo [2+2] cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical retention of the migrating group. However, the Wolff rearrangement has limitations due to the highly reactive nature of α-diazocarbonyl compounds, which can undergo a variety of competing reactions.The Wolff rearrangement can be induced via thermolysis, photolysis, or transition metal catalysis. In this last case, the reaction is sensitive to the transition metal; silver (I) oxide or other Ag(I) catalysts work well and are generally used. The Wolff rearrangement has been used in many total syntheses; the most common use is trapping the ketene intermediate with nucleophiles to form carboxylic acid derivatives. The Arndt-Eistert homologation is a specific example of this use, wherein a carboxylic acid may be elongated by a methylene unit. Another common use is in ring-contraction methods; if the α-diazo ketone is cyclic, the Wolff rearrangement results in a ring-contracted product. The Wolff rearrangement works well in generating ring-strained systems, where other reactions may fail.