Reactions of Hydrocarbons & their functional groups
... 5) REDUCTION REACTION • Carbon atom forms fewer bonds to Oxygen or more bonds to Hydrogen • Aldehydes, ketones and carboxyliic acids can be “reduced” to alcohols • Alkenes and alkynes can be reduced to become alkanes • Occurs in the presence of reducing agents such as LiAlH4, and H2/Pt where Hydrog ...
... 5) REDUCTION REACTION • Carbon atom forms fewer bonds to Oxygen or more bonds to Hydrogen • Aldehydes, ketones and carboxyliic acids can be “reduced” to alcohols • Alkenes and alkynes can be reduced to become alkanes • Occurs in the presence of reducing agents such as LiAlH4, and H2/Pt where Hydrog ...
doc CHEM 222 Lab exam with Answers
... temperature and then allowing them to come back out of solution. 2.__T___ The purpose of refluxing is to carry out a reaction at the boiling point of the solvent. 3.__F___ All chemical reactions must take place in solution. 4.__T___ When a carbene is formed in the presence of an alkene, a cyclopropa ...
... temperature and then allowing them to come back out of solution. 2.__T___ The purpose of refluxing is to carry out a reaction at the boiling point of the solvent. 3.__F___ All chemical reactions must take place in solution. 4.__T___ When a carbene is formed in the presence of an alkene, a cyclopropa ...
CHEM 2412
... carboxylic acids: odor, boiling points, solubility, pH, intermolecular forces; Relative acidity of substituted carboxylic acids; Acid-base reactions; Properties of carboxylate salts; Preparations of carboxylic acids: Oxidation of primary alcohols, Oxidative cleavage of alkenes and alkynes, Oxidation ...
... carboxylic acids: odor, boiling points, solubility, pH, intermolecular forces; Relative acidity of substituted carboxylic acids; Acid-base reactions; Properties of carboxylate salts; Preparations of carboxylic acids: Oxidation of primary alcohols, Oxidative cleavage of alkenes and alkynes, Oxidation ...
E2 reactions
... Decide whether the following substrates could react by E1 or E2 (and by SN1 or SN2). Br ...
... Decide whether the following substrates could react by E1 or E2 (and by SN1 or SN2). Br ...
Chem 30B Spring 2004 QUIZ #1 KEY Weds April 14th / 30
... BONUS QUESTION: What is the product of the Pinacol rearrangement shown below? Write your answer (just the structure) clearly in the box provided on the cover sheet to this quiz. There will be NO partial credit – either your structure is right or wrong. The next blank page can be used for working thr ...
... BONUS QUESTION: What is the product of the Pinacol rearrangement shown below? Write your answer (just the structure) clearly in the box provided on the cover sheet to this quiz. There will be NO partial credit – either your structure is right or wrong. The next blank page can be used for working thr ...
Organic Tutorial 1st Year MT03
... Peter Sykes,“A Guidebook to Mechanism in Organic Chemistry”, and Eames & Peach “Stereochemistry at a Glance”. Notes and Questions a) Summary on not more than 6 sides. This should outline the possible mechanisms and the evidence on which they are based, in particular the evidence for inversion during ...
... Peter Sykes,“A Guidebook to Mechanism in Organic Chemistry”, and Eames & Peach “Stereochemistry at a Glance”. Notes and Questions a) Summary on not more than 6 sides. This should outline the possible mechanisms and the evidence on which they are based, in particular the evidence for inversion during ...
Organic Tutorial 1st Year HT01
... to cover another major function of carbonyl compounds: enolisation and subsequent reaction. A proton a to a carbonyl centre is acidic (we can delocalise the charge on to the electronegative oxygen) and so that this C-H can be deprotonated and then further functionalised by reaction with an electroph ...
... to cover another major function of carbonyl compounds: enolisation and subsequent reaction. A proton a to a carbonyl centre is acidic (we can delocalise the charge on to the electronegative oxygen) and so that this C-H can be deprotonated and then further functionalised by reaction with an electroph ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 1. Classify the following groups into +I and –I groups: i) CH3 ii) NH2 iii) NO2 iv) Cl 2. Give the differences between mesomeric effect and inductive effect. 3. Define torsional strain and torsional energy. 4. Give the eclipsed and staggered conformations of n-butane. 5. Give the IUPAC names and the ...
... 1. Classify the following groups into +I and –I groups: i) CH3 ii) NH2 iii) NO2 iv) Cl 2. Give the differences between mesomeric effect and inductive effect. 3. Define torsional strain and torsional energy. 4. Give the eclipsed and staggered conformations of n-butane. 5. Give the IUPAC names and the ...
File
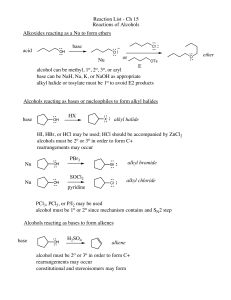
... • 1. Dehydration - elimination of water • water is eliminated from adjacent carbon atoms and a second bond is formed between the 2 carbon atoms • uses a large amount of alcohol and a small amount of acid (H2SO4) as the catalyst • order of reactivity: – tertiary > secondary > primary ...
... • 1. Dehydration - elimination of water • water is eliminated from adjacent carbon atoms and a second bond is formed between the 2 carbon atoms • uses a large amount of alcohol and a small amount of acid (H2SO4) as the catalyst • order of reactivity: – tertiary > secondary > primary ...
Etherification of monosaccharide with isobutene: A - chem
... some bio-feedstock compounds. A recent example is etherification of glycerol and ethylene glycol with isobutene over acidic catalysts. [1] [2] The reaction is straightforward thanks to the facile formation of tertiary carbocation from isobutene, which subsequently reacts with the alcohols. ...
... some bio-feedstock compounds. A recent example is etherification of glycerol and ethylene glycol with isobutene over acidic catalysts. [1] [2] The reaction is straightforward thanks to the facile formation of tertiary carbocation from isobutene, which subsequently reacts with the alcohols. ...
Slide 1
... presence of base or by heating a mixture of the reactants at high temperatures ranging from 150-220°C in the absence of catalyst.(1) ...
... presence of base or by heating a mixture of the reactants at high temperatures ranging from 150-220°C in the absence of catalyst.(1) ...
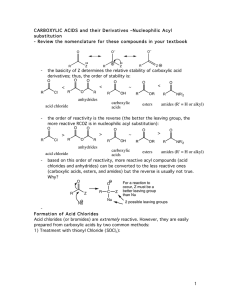
carboxylic acids esters amides (R
... be used to acetylate functional groups such as alcohols and amines. Acetylation can modify both the chemistry and biological activity of a compound. In the case of aspirin, for example, acetylation of the relatively acidic phenol alcohol of salicylic acid leads to a compound that doesn’t dissolve yo ...
... be used to acetylate functional groups such as alcohols and amines. Acetylation can modify both the chemistry and biological activity of a compound. In the case of aspirin, for example, acetylation of the relatively acidic phenol alcohol of salicylic acid leads to a compound that doesn’t dissolve yo ...
Aldehydes and Ketones Both contain the functional group C O
... Of the two reducing agents, sodium borohydride is the milder reagent and is the one of preference for aldehydes and ketones since it is specific for these two functional groups. Lithium aluminum hydride will reduce many types of compounds very quickly. Reduction to hydrocarbons (we saw this used to ...
... Of the two reducing agents, sodium borohydride is the milder reagent and is the one of preference for aldehydes and ketones since it is specific for these two functional groups. Lithium aluminum hydride will reduce many types of compounds very quickly. Reduction to hydrocarbons (we saw this used to ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... approach. State whether the reaction is feasible by thermal or photochemical means. 13. Predict the products in the following pericyclic reactions and explain the mechanism with proper stereochemistry. a) hv 4+2 addition b) H Sigmatropic reaction 120 C ...
... approach. State whether the reaction is feasible by thermal or photochemical means. 13. Predict the products in the following pericyclic reactions and explain the mechanism with proper stereochemistry. a) hv 4+2 addition b) H Sigmatropic reaction 120 C ...
Quiz 3 – Aldehydes and Ketones 1 Which of the following reactions
... 7 You have two C6H10O ketones, I and II. Both are optically active, but I is racemized by treatment with base and II is not. Wolff-Kishner reduction of both ketones gives the same achiral hydrocarbon, formula C6H12. What reasonable structures may be assigned to I and II? A) I is 3-methyl-4-penten-2- ...
... 7 You have two C6H10O ketones, I and II. Both are optically active, but I is racemized by treatment with base and II is not. Wolff-Kishner reduction of both ketones gives the same achiral hydrocarbon, formula C6H12. What reasonable structures may be assigned to I and II? A) I is 3-methyl-4-penten-2- ...
اســـم المـــدرس: د
... ] The reaction of Butanol with HCl is faster than the reaction of t-butyl alcohol ...
... ] The reaction of Butanol with HCl is faster than the reaction of t-butyl alcohol ...
Synthesis of an Ester: Fischer Esterification The ester is synthesised
... The yield of the reaction is also dependent on the position of the equilibrium (i.e. it not lying completely in favour of products) A number of methods can be employed to drive the reaction completely to product, including: - Removing one of the products (ester or water) from the reaction, as it is ...
... The yield of the reaction is also dependent on the position of the equilibrium (i.e. it not lying completely in favour of products) A number of methods can be employed to drive the reaction completely to product, including: - Removing one of the products (ester or water) from the reaction, as it is ...
Nucleophilic Substitution Reaction
... There are a few reactions in which both the groups are lost from the same carbon atom. These are called -elimination reactions and the most common example of this type is the generation of dichlorocarbene from chloroform. In analogy with substitution reaction,b-elimination reactions are divided into ...
... There are a few reactions in which both the groups are lost from the same carbon atom. These are called -elimination reactions and the most common example of this type is the generation of dichlorocarbene from chloroform. In analogy with substitution reaction,b-elimination reactions are divided into ...
Carboxylic Acid Derivatives
... If it is A AC2 (with Nu), the intermediate and reactant (the protonated starting material) are both positively charged. Electronegative groups would destabilize both. However, the effect may be greater on the reactant because the positive charge is transferred to the carbonyl carbon by both an induc ...
... If it is A AC2 (with Nu), the intermediate and reactant (the protonated starting material) are both positively charged. Electronegative groups would destabilize both. However, the effect may be greater on the reactant because the positive charge is transferred to the carbonyl carbon by both an induc ...
1 - Wikispaces
... It is also produced when methyl ethanoate reacts with an acid such as hydrochloric acid. (i) ...
... It is also produced when methyl ethanoate reacts with an acid such as hydrochloric acid. (i) ...
Wolff rearrangement

The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo [2+2] cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical retention of the migrating group. However, the Wolff rearrangement has limitations due to the highly reactive nature of α-diazocarbonyl compounds, which can undergo a variety of competing reactions.The Wolff rearrangement can be induced via thermolysis, photolysis, or transition metal catalysis. In this last case, the reaction is sensitive to the transition metal; silver (I) oxide or other Ag(I) catalysts work well and are generally used. The Wolff rearrangement has been used in many total syntheses; the most common use is trapping the ketene intermediate with nucleophiles to form carboxylic acid derivatives. The Arndt-Eistert homologation is a specific example of this use, wherein a carboxylic acid may be elongated by a methylene unit. Another common use is in ring-contraction methods; if the α-diazo ketone is cyclic, the Wolff rearrangement results in a ring-contracted product. The Wolff rearrangement works well in generating ring-strained systems, where other reactions may fail.