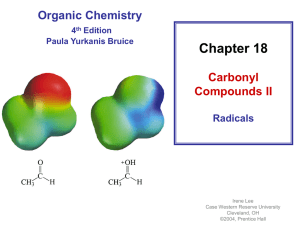
benzylic alcohols
... There is also application in the aliphatic series, for example, pinacolone, available from acetone by reduction and arrangement reacts with bromine in sodium hydroxide solution at below 10oC to give trimethylacetic acid in over 70% yield [I] ...
... There is also application in the aliphatic series, for example, pinacolone, available from acetone by reduction and arrangement reacts with bromine in sodium hydroxide solution at below 10oC to give trimethylacetic acid in over 70% yield [I] ...
Key Practice Exam 3
... 4. Rank in increasing order the boiling points trends you would expect for alcohols, alkanes, ketones and carboxylic acids. Provide rationale for your ranking. Carboxylic acid > alcohol > ketones > alkanes The ranking is based on the ability of these compounds to form hydrogen bonds (their attractiv ...
... 4. Rank in increasing order the boiling points trends you would expect for alcohols, alkanes, ketones and carboxylic acids. Provide rationale for your ranking. Carboxylic acid > alcohol > ketones > alkanes The ranking is based on the ability of these compounds to form hydrogen bonds (their attractiv ...
18 Important and sometimes forgotten) organic transformations
... •Synthesis of anti-diols from alkenes •neighbouring-group participation mechanism prevents the immediate nucleophilic substitution of iodine by a second equivalent of benzoate that would lead to a syn-substituted product. The Ag+ helps to make iodine a better leaving group •The Woodward modification ...
... •Synthesis of anti-diols from alkenes •neighbouring-group participation mechanism prevents the immediate nucleophilic substitution of iodine by a second equivalent of benzoate that would lead to a syn-substituted product. The Ag+ helps to make iodine a better leaving group •The Woodward modification ...
Review 3 - Bonham Chemistry
... 21. Industrially, we often need ethanoic acid. The starting material for this product is usually ethane. Show below a series of reactions that would transform ethane to ethanoic acid. ...
... 21. Industrially, we often need ethanoic acid. The starting material for this product is usually ethane. Show below a series of reactions that would transform ethane to ethanoic acid. ...
2015 CH 420 Take Home Quiz 3 March 24
... an arrow to the more nucleophilic nitrogen atom in the hydrazine substrate. In addition, circle the most electrophilic carbonyl in the 1,3-diketone substrate. ...
... an arrow to the more nucleophilic nitrogen atom in the hydrazine substrate. In addition, circle the most electrophilic carbonyl in the 1,3-diketone substrate. ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... What will be the major product formed on dehydrohalogenation of 2-bromo-2,3dimethylbutane. 06. Name the following alcohols by carbinol and IUPAC system: (i) CH3CHOHCH=CH2 (ii) CH3CH2C(CH3)(OH)CH2CH2CH3 07. COC bond angle in diethyl ether is greater than HOH bond angle in water. Explain. 0 ...
... What will be the major product formed on dehydrohalogenation of 2-bromo-2,3dimethylbutane. 06. Name the following alcohols by carbinol and IUPAC system: (i) CH3CHOHCH=CH2 (ii) CH3CH2C(CH3)(OH)CH2CH2CH3 07. COC bond angle in diethyl ether is greater than HOH bond angle in water. Explain. 0 ...
reactions of the carbonyl group in aldehydes and ketones
... A curly arrow is a symbol used in reaction mechanisms to show the movement of an electron pair in the braking or forming of a covalent bond ...
... A curly arrow is a symbol used in reaction mechanisms to show the movement of an electron pair in the braking or forming of a covalent bond ...
A NEW APROACH TO N-SUBSTITUTED OXAZOLIDINE VIA NITRILIUM ION TRAPPING
... to amides with retention of configuration. This method involved the in situ formation of chlorosulfites followed by a reaction with nitrile complexes of Ti(IV) fluoride. We hypothesize that these amidation reactions involve the intermediacy of nitrilium ions which are subsequently hydrolyzed to form ...
... to amides with retention of configuration. This method involved the in situ formation of chlorosulfites followed by a reaction with nitrile complexes of Ti(IV) fluoride. We hypothesize that these amidation reactions involve the intermediacy of nitrilium ions which are subsequently hydrolyzed to form ...
Slide 1
... If the nucleophile that adds to the aldehyde or ketone is an O or an N, a nucleophilic addition–elimination reaction will occur ...
... If the nucleophile that adds to the aldehyde or ketone is an O or an N, a nucleophilic addition–elimination reaction will occur ...
J. Indian. Chem. Soc. 1999, 76, 631-639
... N-protected amino acid allylic esters can easily be deprotonated by LDA at -78 °C and transmetallated by addition of metal salts. Chelated metal enolates, which undergo Claisen rearrangements upon warming up to room temperature, giving rise to unsaturated amino acids, are formed with many different ...
... N-protected amino acid allylic esters can easily be deprotonated by LDA at -78 °C and transmetallated by addition of metal salts. Chelated metal enolates, which undergo Claisen rearrangements upon warming up to room temperature, giving rise to unsaturated amino acids, are formed with many different ...
Document
... • Imine formation is also a nucleophilic addition. • There is a different end result here, though as elimination of water occurs. • The initial reaction is attack of the amine on the carbonyl to give the alkoxide intermediate as normal. • Following protonation of the alkoxide and loss of the proton ...
... • Imine formation is also a nucleophilic addition. • There is a different end result here, though as elimination of water occurs. • The initial reaction is attack of the amine on the carbonyl to give the alkoxide intermediate as normal. • Following protonation of the alkoxide and loss of the proton ...
Exam 1
... Acid catalyzed ester & amide hydrolysis, Fisher esterification, trans esterification Hydroxide promoted ester hydrolysis Nucleophilic addition to aldehyde/ketone with Grignard reagents Determine reaction mechanisms from reaction outcome (see Mechanism work sheet). Be able to recognize the following ...
... Acid catalyzed ester & amide hydrolysis, Fisher esterification, trans esterification Hydroxide promoted ester hydrolysis Nucleophilic addition to aldehyde/ketone with Grignard reagents Determine reaction mechanisms from reaction outcome (see Mechanism work sheet). Be able to recognize the following ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034 PART-A
... 05. Why group transfer reactions are neither cycloaddition nor sigmatropic rearrangement reactions? 06. Does hydroboration of alkene follow Markonikov’s addition? Justify your answer with suitable example. 07. How is catalytic hydrogenation different from dissolving metal reduction? Give reasons. 08 ...
... 05. Why group transfer reactions are neither cycloaddition nor sigmatropic rearrangement reactions? 06. Does hydroboration of alkene follow Markonikov’s addition? Justify your answer with suitable example. 07. How is catalytic hydrogenation different from dissolving metal reduction? Give reasons. 08 ...
Preface - Wiley Online Library
... unusual class of reactions that involve the loss of small molecular fragments from a ring in service of uniting separate carbon atoms in that ring to form alkenes. These so called “extrusion reactions” are more commonly known for the loss of a single molecular fragment as in, e.g. the Ramberg-B¨ackl ...
... unusual class of reactions that involve the loss of small molecular fragments from a ring in service of uniting separate carbon atoms in that ring to form alkenes. These so called “extrusion reactions” are more commonly known for the loss of a single molecular fragment as in, e.g. the Ramberg-B¨ackl ...
AMINO ACIDS Ethan Secor, John N. Gitua (Mentor)
... The titanium complex was prepared in purified tetrahydrofuran using titanium(IV) isopropoxide and butyl lithium at low temperatures maintained by mixing acetone and dry ice. The imine was added to this mixture. The reaction mixture was gradually warmed to room temperature. Carbon dioxide was introdu ...
... The titanium complex was prepared in purified tetrahydrofuran using titanium(IV) isopropoxide and butyl lithium at low temperatures maintained by mixing acetone and dry ice. The imine was added to this mixture. The reaction mixture was gradually warmed to room temperature. Carbon dioxide was introdu ...
1. Rank the following compounds in order of decreasing acidity (1
... 6. What is the carbon nucleophile that attacks molecular bromine in the acid-catalyzed α-bromination of a ketone? (a) an enolate (b) a carbocation (c) an acetylide (d) an enol 7. The Hell-Volhard-Zelinsky reaction involves: (a) the α-bromination of carboxylic acids (b) the α-bromination of ketones ( ...
... 6. What is the carbon nucleophile that attacks molecular bromine in the acid-catalyzed α-bromination of a ketone? (a) an enolate (b) a carbocation (c) an acetylide (d) an enol 7. The Hell-Volhard-Zelinsky reaction involves: (a) the α-bromination of carboxylic acids (b) the α-bromination of ketones ( ...
Carboxylic Acid
... You know Alkanes and Benzenes, and Alkynes and Alkenes, Amines and Alcohols, Aldehydes and Ketones...... But do you recall, the most famous functional group of all Carboxylic Acid, Has a carbonyl and a hydroxyl It loves to donate protons Then an anion is formed Of all the other acids It’s the most c ...
... You know Alkanes and Benzenes, and Alkynes and Alkenes, Amines and Alcohols, Aldehydes and Ketones...... But do you recall, the most famous functional group of all Carboxylic Acid, Has a carbonyl and a hydroxyl It loves to donate protons Then an anion is formed Of all the other acids It’s the most c ...
Chap Thirteen: Alcohols
... inversion of configuration incomplete iii. SN2 reaction With phosphorus trihalides PBr3 or PCl3 or PCl5 or P° and I2 to form alkyl halides o Mechanism/ no rearrangement/ inversion of configuration e. Alkyl tosylates (sulfonate esters) by reaction of ROH with sulfonyl chlorides i. Mechanism/ retentio ...
... inversion of configuration incomplete iii. SN2 reaction With phosphorus trihalides PBr3 or PCl3 or PCl5 or P° and I2 to form alkyl halides o Mechanism/ no rearrangement/ inversion of configuration e. Alkyl tosylates (sulfonate esters) by reaction of ROH with sulfonyl chlorides i. Mechanism/ retentio ...
Outline_CH13_Klein
... inversion of configuration incomplete iii. SN2 reaction With phosphorus trihalides PBr3 or PCl3 or PCl5 or P° and I2 to form alkyl halides o Mechanism/ no rearrangement/ inversion of configuration e. Alkyl tosylates (sulfonate esters) by reaction of ROH with sulfonyl chlorides i. Mechanism/ retentio ...
... inversion of configuration incomplete iii. SN2 reaction With phosphorus trihalides PBr3 or PCl3 or PCl5 or P° and I2 to form alkyl halides o Mechanism/ no rearrangement/ inversion of configuration e. Alkyl tosylates (sulfonate esters) by reaction of ROH with sulfonyl chlorides i. Mechanism/ retentio ...
CHE 322
... 3. (8) Give the complete mechanism that shows why the reaction of butanal with a cyclic 2° amine followed by heating with acid produces an enamine that is nucleophilic at butanal’s former α-carbon. ...
... 3. (8) Give the complete mechanism that shows why the reaction of butanal with a cyclic 2° amine followed by heating with acid produces an enamine that is nucleophilic at butanal’s former α-carbon. ...
$doc.title
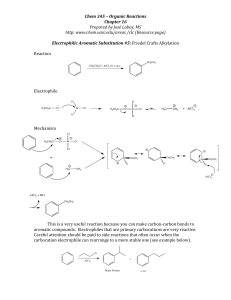
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
Wolff rearrangement

The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo [2+2] cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical retention of the migrating group. However, the Wolff rearrangement has limitations due to the highly reactive nature of α-diazocarbonyl compounds, which can undergo a variety of competing reactions.The Wolff rearrangement can be induced via thermolysis, photolysis, or transition metal catalysis. In this last case, the reaction is sensitive to the transition metal; silver (I) oxide or other Ag(I) catalysts work well and are generally used. The Wolff rearrangement has been used in many total syntheses; the most common use is trapping the ketene intermediate with nucleophiles to form carboxylic acid derivatives. The Arndt-Eistert homologation is a specific example of this use, wherein a carboxylic acid may be elongated by a methylene unit. Another common use is in ring-contraction methods; if the α-diazo ketone is cyclic, the Wolff rearrangement results in a ring-contracted product. The Wolff rearrangement works well in generating ring-strained systems, where other reactions may fail.