* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Organometallic Chemistry
Spin crossover wikipedia , lookup
Metalloprotein wikipedia , lookup
Sol–gel process wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
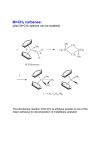
Metal carbonyl wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Coordination complex wikipedia , lookup
Hydroformylation wikipedia , lookup
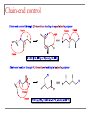
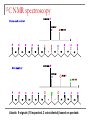
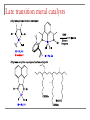
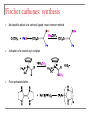
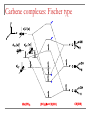
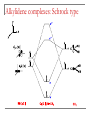
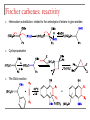
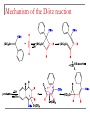
Tacticity isotactic • Isotactic and syndiotactic polymers are crystalline, atactic is amorphous. • NMR spectroscopy is a syndiotactic atactic powerful tool for studying polymer stereochemistry. Triad Tacticity • Ziegler/Natta catalysts Dyad Tacticity allow control of tacticity. m •Tacticity of polymer is determined by % m or r dyads. (Perfectly isotactic polypropylene has 100% m dyads) m m isotactic(mm) isotactic (meso, m) m r r atactic (mr) syndiotactic (racemic, r) r r syndiotactic (rr) Site vs. chain-end control Site control mechanism: the catalytic center determines the stereospecificity of the polymer. Chain-end control mechanism: the polymer chain determines the stereospecificity of the final polymer. Chain-end control 13C NMR spectroscopy Atactic: 9 signals (10 expected, 2 coincidental) based on pentads Late transition metal catalysts M=C complexes Textbook H: Chapter 13.1 – 13.2 Textbook A: Chapter 9 2005 Nobel prize in chemistry "for the development of the metathesis method in organic synthesis" Richard Schrock Massachusetts Institute of Technology (MIT) Cambridge, MA, USA Yves Chauvin Institut Français du Pétrole Rueil-Malmaison, France Robert Grubbs California Institute of Technology (Caltech) Pasadena, CA, USA http://nobelprize.org/chemistry/laureates/2005/index.html Outline Carbene complexes Bonding Structural and spectroscopic features Synthesis Reactivity Olefin metathesis Mechanism Reaction overview Grubbs catalysts Stereochemistry Catalyst decomposition Alkyne metathesis Reactions related to olefin metathesis ROMP RCM M=C complexes: comparison Property Fischer-type (carbene) Schrock-type (alkylidene) Nature of carbene C Typical R groups Typical metal Typical ligands also attached to the metal Electrophilic p donor (e.g. OR) Mo(0), Fe(O), Cr(0) Good p acceptor (e.g. CO) Nucleophilic Alkyl, H Ti(IV), Ta(V), W(VI) Good s or p donor (Cl, Cp, Alkyl) Fischer carbenes: synthesis Nucleophilic attack at a carbonyl ligand: most common method Activation of a neutral acyl complex From activated olefins Schrock carbenes: synthesis a-abstraction: induced by steric bulk Rearrangement of coordinated ligands From activated olefins M=C complexes: comparison Property Fischer-type (carbene) Schrock-type (alkylidene) Nature of carbene C Typical R groups Typical metal Typical ligands also attached to the metal Electrophilic p donor (e.g. OR) Mo(0), Fe(O), Cr(0) Good p acceptor (e.g. CO) Nucleophilic Alkyl, H Ti(IV), Ta(V), W(VI) Good s or p donor (Cl, Cp, Alkyl) Carbene complexes: Fischer type Alkylidene complexes: Schrock type Structural and spectroscopic features Fischer type Schrock type NMR: 5 – 15 ppm 13C NMR: 220 – 260 ppm JCH typical: 150 – 160 Hz JCH agostic: 90 – 120 Hz 1H MCC angle: 160 – 170° MC distance: longer than M=C, but shorter than M-C CX distance: shorter than C-X Fischer carbenes: reactivity Heteroatom substitution: related to the aminolysis of esters to give amides Cyclopropanation The Dötz reaction Mechanism of the Dötz reaction Schrock carbenes: reactions with electrophiles Other reactions Olefin metathesis Mechanism: Such cycloaddition reactions between two alkenes to give cyclobutanes are symmetry forbidden and occur only photochemically. However, the presence of dorbitals on the metal alkylidene fragment breaks this symmetry and the reaction is quite facile. Normally, the products are statistical, unless the reaction can be driven in some way or the two alkenes have different reactivities. Reference: Chem. & Eng. News 2002, Dec 23, 34-38 http://nobelprize.org/nobel_prizes/chemistry/laureates/2005/animation.html Timeline