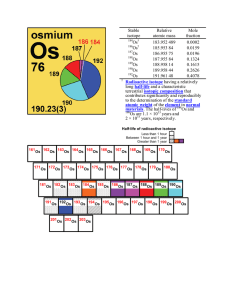
Stable isotope Relative atomic mass Mole fraction Os 183.952 489
... either positive or negative charge (an electron or positron). [return] electron – elementary particle of matter with a negative electric charge and a rest mass of about 9.109 × 10–31 kg. element (chemical element) – a species of atoms; all atoms with the same number of protons in the atomic nucleus. ...
... either positive or negative charge (an electron or positron). [return] electron – elementary particle of matter with a negative electric charge and a rest mass of about 9.109 × 10–31 kg. element (chemical element) – a species of atoms; all atoms with the same number of protons in the atomic nucleus. ...
SCI 3101 Test IV MULTIPLE CHOICE. 1) The sky is blue because air
... C) That would be too much of a coincidence. D) Today's instruments are able to measure the atomic masses to many decimal places. 16) Suppose that a certain atom possesses only four distinct energy levels. Assuming that all transitions between levels are possible, how many spectral lines will this at ...
... C) That would be too much of a coincidence. D) Today's instruments are able to measure the atomic masses to many decimal places. 16) Suppose that a certain atom possesses only four distinct energy levels. Assuming that all transitions between levels are possible, how many spectral lines will this at ...
Lesson 3.1
... space taken up by the nucleus. If the nucleus was the size of a green pea in the middle of a football stadium, the electrons would reach as far away as the top row of seats! ...
... space taken up by the nucleus. If the nucleus was the size of a green pea in the middle of a football stadium, the electrons would reach as far away as the top row of seats! ...
Chemistry: Nuclear Reactions Guided Inquiry + n → + + 3 n +
... STOP – Show your nuclear equation to the teacher. Types of Nuclear Reactions There are 4 general types of nuclear reactions: radioactive decay, fission, fusion and artificial transmutation. Radioactive decay ...
... STOP – Show your nuclear equation to the teacher. Types of Nuclear Reactions There are 4 general types of nuclear reactions: radioactive decay, fission, fusion and artificial transmutation. Radioactive decay ...
Atomic Theory PPT
... atomic number) with different atomic masses (different # of neutrons). C-14 and ...
... atomic number) with different atomic masses (different # of neutrons). C-14 and ...
Chapter 28
... decay into 94Pu, which then decays by α decay into 92U. Uranium then decays in a series of 14 steps to 206Pb. ...
... decay into 94Pu, which then decays by α decay into 92U. Uranium then decays in a series of 14 steps to 206Pb. ...
Notes: Structure of matter
... What do these parts have in common? Which part makes-up the atom's cloud ? How many electrons can the 3 inner most energy levels hold? ...
... What do these parts have in common? Which part makes-up the atom's cloud ? How many electrons can the 3 inner most energy levels hold? ...
Subatomic Particles
... So different numbers of…. Neutrons! How many neutrons does each have? How would we figure that out? Mass Number - Atomic Number = Number of neutrons ...
... So different numbers of…. Neutrons! How many neutrons does each have? How would we figure that out? Mass Number - Atomic Number = Number of neutrons ...
Atomic Math Powerpoint - Parkway C-2
... So different numbers of…. Neutrons! How many neutrons does each have? How would we figure that out? Mass Number - Atomic Number = Number of neutrons ...
... So different numbers of…. Neutrons! How many neutrons does each have? How would we figure that out? Mass Number - Atomic Number = Number of neutrons ...
13.4 The nucleus 3 - Nuclear fission and nuclear fusion
... Fission means breaking apart and fusion means joining together. Nuclear fission refers to the breaking apart of the nucleus of an atom. The best known example of nuclear fission occurs when the nucleus of the uranium isotope 235U captures an extra neutron. This makes the nucleus unstable and it brea ...
... Fission means breaking apart and fusion means joining together. Nuclear fission refers to the breaking apart of the nucleus of an atom. The best known example of nuclear fission occurs when the nucleus of the uranium isotope 235U captures an extra neutron. This makes the nucleus unstable and it brea ...
Which of the following statements correctly describes the
... If an atom has a mass number of 18, what can be said about the number of protons and neutrons it contains? A ...
... If an atom has a mass number of 18, what can be said about the number of protons and neutrons it contains? A ...
Candidate 2 - Elgin Academy
... The alpha particles are constantly released and knock electrons off the atoms in air. This makes these atoms into ions which are attracted to the negatively charged plate and the electrons are attracted to the positively charged plate generating a small current. When smoke enters the chamber the smo ...
... The alpha particles are constantly released and knock electrons off the atoms in air. This makes these atoms into ions which are attracted to the negatively charged plate and the electrons are attracted to the positively charged plate generating a small current. When smoke enters the chamber the smo ...
Ch. 21.1 Nuclear Radiation
... – The process continues until unstable isotopes of one element are changed, or transformed, into stable isotopes of a different element. – These stable isotopes are not radioactive. – Nuclear radiation is emitted during radioactive decay. – There are three main types of nuclear radiation: alpha rad ...
... – The process continues until unstable isotopes of one element are changed, or transformed, into stable isotopes of a different element. – These stable isotopes are not radioactive. – Nuclear radiation is emitted during radioactive decay. – There are three main types of nuclear radiation: alpha rad ...
Atomic Structure Notes_BohrRing Activity
... Slides 1-17 are review (we did this last class) Start with slide #18 and confirm your answers for part I and II ...
... Slides 1-17 are review (we did this last class) Start with slide #18 and confirm your answers for part I and II ...
Atomic Structure
... Table is determined by its proton number. All elements in the same group have the same number of valence electrons, which is the same as the Group number. All elements in the same period have the same number of electron shells. ...
... Table is determined by its proton number. All elements in the same group have the same number of valence electrons, which is the same as the Group number. All elements in the same period have the same number of electron shells. ...
Chapter 11 and 12-2 Review/Study Guide for Test
... 5. What happens to the electrons in the atom when it becomes an ion? They are gained or lost to another atom. 6. What is an isotope? When atoms of the same element have different numbers of neutrons from each other. 7. What determines the identity of an element? The number of protons in the nucleus ...
... 5. What happens to the electrons in the atom when it becomes an ion? They are gained or lost to another atom. 6. What is an isotope? When atoms of the same element have different numbers of neutrons from each other. 7. What determines the identity of an element? The number of protons in the nucleus ...
1 CP Chemistry 2013-2014 Chapter 3 Notes Name: Important Vocab
... The observations from the gold foil experiment are: ...
... The observations from the gold foil experiment are: ...
Atoms Are Building Blocks
... Protons and neutrons are found in the nucleus. The nucleus is the center of the atom, and is made of protons and neutrons. Protons and neutrons both have a mass of 1 atomic mass unit (amu). This means an atom with 1 proton and 1 neutron would have 2 amu. Electrons are the smallest of the three parti ...
... Protons and neutrons are found in the nucleus. The nucleus is the center of the atom, and is made of protons and neutrons. Protons and neutrons both have a mass of 1 atomic mass unit (amu). This means an atom with 1 proton and 1 neutron would have 2 amu. Electrons are the smallest of the three parti ...
4.1Atoms and Isotopes
... Tin (Sn) has the most isotopes of any element at 10 Many isotopes are radioactive (unstable nucleus that will eventually break apart and release energy in sometimes harmful forms – eg. Gamma rays) Any isotope with an atomic number greater than 82 is radioactive ...
... Tin (Sn) has the most isotopes of any element at 10 Many isotopes are radioactive (unstable nucleus that will eventually break apart and release energy in sometimes harmful forms – eg. Gamma rays) Any isotope with an atomic number greater than 82 is radioactive ...
Sample pages 1 PDF
... The atomic nucleus Subsequently, different models of the atom were discussed, one of them being the model of Thomson. In this model, the electrons, and an equivalent number of positively charged particles are uniformly distributed throughout the atom. The resulting atom is electrically neutral. Ruth ...
... The atomic nucleus Subsequently, different models of the atom were discussed, one of them being the model of Thomson. In this model, the electrons, and an equivalent number of positively charged particles are uniformly distributed throughout the atom. The resulting atom is electrically neutral. Ruth ...
1 Chapter 4 Atomic Structure 4.1 Defining the Atom Early Models of
... Dalton's Atomic Theory The modern process of discovery regarding atoms began with John _____________ (1766-1844). By using experimental methods. Dalton transformed Democritus's ideas on atoms into a scientific theory. Dalton's atomic theory 1. All elements are composed of tiny indivisible particles ...
... Dalton's Atomic Theory The modern process of discovery regarding atoms began with John _____________ (1766-1844). By using experimental methods. Dalton transformed Democritus's ideas on atoms into a scientific theory. Dalton's atomic theory 1. All elements are composed of tiny indivisible particles ...
mass number - Knittig Science
... A nucleon is a general term to denote a nuclear particle - that is, either a proton or a neutron. The atomic number Z of an element is equal to the number of protons in the nucleus of that element. The mass number A of an element is equal to the total number of nucleons (protons + neutrons). The mas ...
... A nucleon is a general term to denote a nuclear particle - that is, either a proton or a neutron. The atomic number Z of an element is equal to the number of protons in the nucleus of that element. The mass number A of an element is equal to the total number of nucleons (protons + neutrons). The mas ...























