Chapter 3 Chemical Foundations: Elements, Atoms, and Ions
... 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same ...
... 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same ...
lecture 13
... Modern Atomic Theory and the Law of Conservation of Mass. BALANCING EQUATIONS: The same number of each type of element must occur on the left (BEFORE the reaction) and on the right (AFTER the reaction) ...
... Modern Atomic Theory and the Law of Conservation of Mass. BALANCING EQUATIONS: The same number of each type of element must occur on the left (BEFORE the reaction) and on the right (AFTER the reaction) ...
AP Chemistry Summer Assignment
... 10. A solid white substance A is heated strongly in the absence of air. It decomposes to form a new white substance B and a gas C. The gas has exactly the same properties as the product obtained when carbon is burned in an excess of oxygen. Based on these observations, can we determine whether soli ...
... 10. A solid white substance A is heated strongly in the absence of air. It decomposes to form a new white substance B and a gas C. The gas has exactly the same properties as the product obtained when carbon is burned in an excess of oxygen. Based on these observations, can we determine whether soli ...
Chemical Equation
... particles. • In general: the electrons are shared between the ions. Metals tend to give up their electrons to an incomplete nonmetal. • All Ionic compounds are represented by their empirical formulas. They are always in the smallest whole number ratios. ...
... particles. • In general: the electrons are shared between the ions. Metals tend to give up their electrons to an incomplete nonmetal. • All Ionic compounds are represented by their empirical formulas. They are always in the smallest whole number ratios. ...
Chemistry in Biology
... COMPOUNDS II. Composition of Matter A. Elements—pure substances that cannot be broken down into simpler kinds of matter • Made of one type of atom • More than 100 elements (92 naturally occurring) • 90% of the mass of an organism is composed of 4 elements (oxygen, carbon, hydrogen, and nitrogen) • E ...
... COMPOUNDS II. Composition of Matter A. Elements—pure substances that cannot be broken down into simpler kinds of matter • Made of one type of atom • More than 100 elements (92 naturally occurring) • 90% of the mass of an organism is composed of 4 elements (oxygen, carbon, hydrogen, and nitrogen) • E ...
File
... _____ 14. The activity series of metals can be used to predict products in double-replacement reactions. _____ 15. Carbon dioxide and water are the products of the combustion of hexane (C6H14). _____ 16. A nonmetal can replace another nonmetal from a compound in a single-replacement reaction. ...
... _____ 14. The activity series of metals can be used to predict products in double-replacement reactions. _____ 15. Carbon dioxide and water are the products of the combustion of hexane (C6H14). _____ 16. A nonmetal can replace another nonmetal from a compound in a single-replacement reaction. ...
Essential Standard: 8.P.1 Understand the properties of matter and
... Elements are pure substances that cannot be changed into simpler substances. Elements are composed of one kind of atom. Compounds are pure substances that are composed of two or more types of elements that are chemically combined. Compounds can only be changed into simpler substances called elements ...
... Elements are pure substances that cannot be changed into simpler substances. Elements are composed of one kind of atom. Compounds are pure substances that are composed of two or more types of elements that are chemically combined. Compounds can only be changed into simpler substances called elements ...
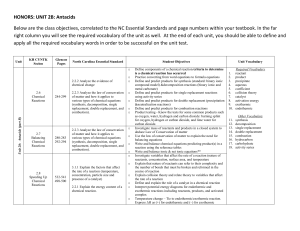
HONORS: UNIT 2B: Antacids Below are the class objectives
... Define components of a chemical reaction/criteria to determine is a chemical reaction has occurred Practice converting from word equations to formula equations Define and predict products for synthesis (standard: binary ionic compound model) &decomposition reactions (binary ionic and metal carbonate ...
... Define components of a chemical reaction/criteria to determine is a chemical reaction has occurred Practice converting from word equations to formula equations Define and predict products for synthesis (standard: binary ionic compound model) &decomposition reactions (binary ionic and metal carbonate ...
Chemical Reactions & Balancing Equations
... – same number of atoms of each type of element on each side What if it isn’t balanced already? ...
... – same number of atoms of each type of element on each side What if it isn’t balanced already? ...
Basics of Chemistry
... Life requires ~25 chemical elements Atomic structure determines behavior of an element ...
... Life requires ~25 chemical elements Atomic structure determines behavior of an element ...
PowerPoint Template
... chemical reaction - Antoine Lavoisier (1 743-1 794) The number of substances may change, but the total amount of matter remains constant. ...
... chemical reaction - Antoine Lavoisier (1 743-1 794) The number of substances may change, but the total amount of matter remains constant. ...
CHEMICAL REACTIONS
... – Chemical reactions occur when bonds between the outermost parts of atoms are formed or broken – Chemical reactions involve changes in matter, the making of new materials with new properties, and energy changes. – Symbols represent elements, formulas describe compounds, chemical equations describe ...
... – Chemical reactions occur when bonds between the outermost parts of atoms are formed or broken – Chemical reactions involve changes in matter, the making of new materials with new properties, and energy changes. – Symbols represent elements, formulas describe compounds, chemical equations describe ...
PUC Schools - cloudfront.net
... 53. Carbon can form a large variety of compounds from simple to highly complex due to its ability to a) form ions with four different charges b) form four covalent bonds c) share electrons with itself d) form ionic bonds with many elements ...
... 53. Carbon can form a large variety of compounds from simple to highly complex due to its ability to a) form ions with four different charges b) form four covalent bonds c) share electrons with itself d) form ionic bonds with many elements ...
CHAPTER 2: THE ATOMS AND MOLECULES OF ANCIENT EARTH
... 2. Redox reactions are the most common chemical reactions in biology. 3. Reduction of carbon was a key step in chemical evolution. a. Carbon is the most versatile molecule found in biological tissues. (1) Each carbon atom can form four bonds with other molecules. (2) Carbon atoms form the skeleton o ...
... 2. Redox reactions are the most common chemical reactions in biology. 3. Reduction of carbon was a key step in chemical evolution. a. Carbon is the most versatile molecule found in biological tissues. (1) Each carbon atom can form four bonds with other molecules. (2) Carbon atoms form the skeleton o ...
e c n i
... The activation energy is the energy needed by a system to initiate the reaction. It is the minimum energy needed for a specific chemical reaction to occur. Once achieved, the reaction continues until reactants are ...
... The activation energy is the energy needed by a system to initiate the reaction. It is the minimum energy needed for a specific chemical reaction to occur. Once achieved, the reaction continues until reactants are ...
Ahmed Fazary_Click Chemistry
... carbon - hetero atom bonds C-X-C rather than carbon carbon bonds. In addition, enzymes ensure that chemical processes can overcome large enthalpy hurdles by division into a series of reactions each with a small energy step. Mimicking nature in organic synthesis of new pharmaceuticals is essential gi ...
... carbon - hetero atom bonds C-X-C rather than carbon carbon bonds. In addition, enzymes ensure that chemical processes can overcome large enthalpy hurdles by division into a series of reactions each with a small energy step. Mimicking nature in organic synthesis of new pharmaceuticals is essential gi ...
Physical Science
... It’s like printing a word in a different font, it’s the same word just looks different! ...
... It’s like printing a word in a different font, it’s the same word just looks different! ...
Name - rwebbchem
... 2. You have a solution of an unknown ionic compound. The unknown solution contains either Br-, CO32-, or NO3- ions. Separate samples of the unknown solution are treated with AgNO3, Pb(NO3)2, and BaCl2. Precipitates form in all three cases. Which of the following could be the anion of the unknown sal ...
... 2. You have a solution of an unknown ionic compound. The unknown solution contains either Br-, CO32-, or NO3- ions. Separate samples of the unknown solution are treated with AgNO3, Pb(NO3)2, and BaCl2. Precipitates form in all three cases. Which of the following could be the anion of the unknown sal ...
CHM 130 Final Exam Review Chapter 1 Scientific method Theory
... Activity series Solubility rules Electrolytes Oxidation and reduction, the agents Writing products Chapter 16 Increasing the rate of a reaction Energy profiles Chapter 9 The mole and Avogadro’s number Molar mass Converting between grams and moles and atoms/molecules Molar volume Converting between l ...
... Activity series Solubility rules Electrolytes Oxidation and reduction, the agents Writing products Chapter 16 Increasing the rate of a reaction Energy profiles Chapter 9 The mole and Avogadro’s number Molar mass Converting between grams and moles and atoms/molecules Molar volume Converting between l ...
Fall.2008.Week9.Lesson.2 - reich
... • 1-Berylium chloride and aluminum react together. What is the reaction type? Balance the chemical reaction. • 2-Magnesium chloride and sodium phosphate undergo a double displacement reaction. Go through all the steps to show the net ionic equation. • 3- When you cook with a propane grill you burn p ...
... • 1-Berylium chloride and aluminum react together. What is the reaction type? Balance the chemical reaction. • 2-Magnesium chloride and sodium phosphate undergo a double displacement reaction. Go through all the steps to show the net ionic equation. • 3- When you cook with a propane grill you burn p ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.