Unit 5 and 6 revsion - Deans Community High School
... (a) Name the acidic gas formed when air in a car engine is sparked. (b) Name the acidic gas formed when coal with a high sulphur content is burned. (c) Name the toxic gas formed when methane is burned in a limited supply of air. (d) A catalytic converter fitted to a car exhaust reduces the amount of ...
... (a) Name the acidic gas formed when air in a car engine is sparked. (b) Name the acidic gas formed when coal with a high sulphur content is burned. (c) Name the toxic gas formed when methane is burned in a limited supply of air. (d) A catalytic converter fitted to a car exhaust reduces the amount of ...
PowerPoint for Cornell Notes
... • We learned about Physical and Chemical properties and changes. – Physical properties were those like mass, volume, density, melting point, boiling point, conductivity, malleability, state/phase of matter etc. • Changes within these properties can be undone and the properties of the substances are ...
... • We learned about Physical and Chemical properties and changes. – Physical properties were those like mass, volume, density, melting point, boiling point, conductivity, malleability, state/phase of matter etc. • Changes within these properties can be undone and the properties of the substances are ...
AP CHEMISTRY
... and that helped John Dalton develop his Atomic Theory in the early 1800s. Learning Objectives To understand three laws about mass that were important for development of atomic theory. Success Criteria Be able to state, describe the usefulness of, and use the laws named in the New Concepts sectio ...
... and that helped John Dalton develop his Atomic Theory in the early 1800s. Learning Objectives To understand three laws about mass that were important for development of atomic theory. Success Criteria Be able to state, describe the usefulness of, and use the laws named in the New Concepts sectio ...
classification of chemical reactions
... Chemical change change in matter that produces new substances Example: rusting of iron burning of wood Physical change a change that does not produce a new substance a change in appearance or state Example: chopping wood ...
... Chemical change change in matter that produces new substances Example: rusting of iron burning of wood Physical change a change that does not produce a new substance a change in appearance or state Example: chopping wood ...
What is Matter PowerPoint
... • Remember mass is measured in grams and taking up space is a measurement of volume (which is a derived unit) • Matter is composed of tiny particles that are always in constant motion • Examples: Salt, wood, atom, insect • Matter can be described as either a pure substance or a mixture. ...
... • Remember mass is measured in grams and taking up space is a measurement of volume (which is a derived unit) • Matter is composed of tiny particles that are always in constant motion • Examples: Salt, wood, atom, insect • Matter can be described as either a pure substance or a mixture. ...
objectives chm 1025 - Miami Dade College
... The student will demonstrate an ability to understand several of the intricacies of the periodic table by: a. Showing how to obtain an element’s average atomic mass and atomic number from the periodic table. b. Using the structure of the periodic table to classify elements (e.g., metal, nonmetal, me ...
... The student will demonstrate an ability to understand several of the intricacies of the periodic table by: a. Showing how to obtain an element’s average atomic mass and atomic number from the periodic table. b. Using the structure of the periodic table to classify elements (e.g., metal, nonmetal, me ...
Matter and Atoms
... masses B. Nature contains a variety of isotopes C. Isotopes used to find atomic mass of element ...
... masses B. Nature contains a variety of isotopes C. Isotopes used to find atomic mass of element ...
The Periodic Table - Harlan Independent Schools
... anymore, it used to be used in glow-in-thedark paints. The other elements are found in many items including fireworks, batteries, flashbulbs, and special alloys. The lighter alkaline earth metals such as magnesium and calcium are very important in animal and plant physiology. You all know that calci ...
... anymore, it used to be used in glow-in-thedark paints. The other elements are found in many items including fireworks, batteries, flashbulbs, and special alloys. The lighter alkaline earth metals such as magnesium and calcium are very important in animal and plant physiology. You all know that calci ...
File
... _____ 6. If two or more compounds are composed of the same two elements, then the ratio of the masses of the second element that is combined with a certain mass of the first element is always a ratio of small whole numbers. This statement is called the law of a. definite proportions. b. conservation ...
... _____ 6. If two or more compounds are composed of the same two elements, then the ratio of the masses of the second element that is combined with a certain mass of the first element is always a ratio of small whole numbers. This statement is called the law of a. definite proportions. b. conservation ...
section_2_review_set
... 1. What is the claim to fame for the proton? 2. What is the claim to fame for the electron? 3. What is the claim to fame for the neutron? 4. What is the mass of each of the following particles?: proton; neutron; electron. 5. What is the charge for each of the following particles?: proton; neutron; e ...
... 1. What is the claim to fame for the proton? 2. What is the claim to fame for the electron? 3. What is the claim to fame for the neutron? 4. What is the mass of each of the following particles?: proton; neutron; electron. 5. What is the charge for each of the following particles?: proton; neutron; e ...
Dr. Ali Ebneshahidi © 2016 Ebneshahidi
... Since all enzymes are made of globular proteins, and proteins are made of amino acids linked by peptide bonds, enzymes can be affected or denatured very easily. Factors that could affect or denature enzymes include heat, radiation , electricity, certain chemical substances, and extreme pH. © 201 ...
... Since all enzymes are made of globular proteins, and proteins are made of amino acids linked by peptide bonds, enzymes can be affected or denatured very easily. Factors that could affect or denature enzymes include heat, radiation , electricity, certain chemical substances, and extreme pH. © 201 ...
HERE
... 2) One difference between mixtures and pure substances is that A) mixtures can be physically separated. B) mixtures are made of one type of atom. C) pure substances have no chemical bonds. D) pure substances can be physically separated. 3) When two or more substances combine, but each keeps its own ...
... 2) One difference between mixtures and pure substances is that A) mixtures can be physically separated. B) mixtures are made of one type of atom. C) pure substances have no chemical bonds. D) pure substances can be physically separated. 3) When two or more substances combine, but each keeps its own ...
Science-M2-Basic-Che..
... other’s questions is a great confidence builder, as they see that they have the skills to help each other. Finally, have each group come up to the board and lead the class through the process. Using the periodic chart to find the number of protons, neutrons and electrons for each element: protons = ...
... other’s questions is a great confidence builder, as they see that they have the skills to help each other. Finally, have each group come up to the board and lead the class through the process. Using the periodic chart to find the number of protons, neutrons and electrons for each element: protons = ...
AP Chemistry Syllabus - Tuloso
... descriptive facts, including the chemistry involved in environmental and societal issues, should not be isolated from the principles being studied but should be taught throughout the course to illustrate and illuminate the principles. The following areas should be covered: A. Chemical reactivity and ...
... descriptive facts, including the chemistry involved in environmental and societal issues, should not be isolated from the principles being studied but should be taught throughout the course to illustrate and illuminate the principles. The following areas should be covered: A. Chemical reactivity and ...
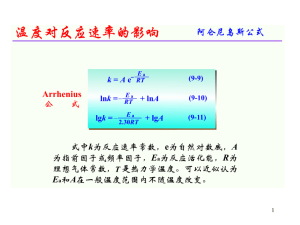
Lecture 7
... Like group 1 compounds, much of the reactivity is due to the reactions of the anions, but unlike group 1, the cations here have twice as much charge and so are more polarizing. This polarizing power is especially noticeable at the top of the group: the beryllium ion is very small and polarizes water ...
... Like group 1 compounds, much of the reactivity is due to the reactions of the anions, but unlike group 1, the cations here have twice as much charge and so are more polarizing. This polarizing power is especially noticeable at the top of the group: the beryllium ion is very small and polarizes water ...
ch15-Atmospheric Chemistry
... – Vinyl chloride used to make polyvinylchloride plastic, carcinogen – Trichloroethylene widely used as a dry cleaning solvent • PCBs are notorious water pollutants – Once widely used in electrical applications, as hydraulic fluids, other – Highly persistent and bioaccumulative, manufacture now banne ...
... – Vinyl chloride used to make polyvinylchloride plastic, carcinogen – Trichloroethylene widely used as a dry cleaning solvent • PCBs are notorious water pollutants – Once widely used in electrical applications, as hydraulic fluids, other – Highly persistent and bioaccumulative, manufacture now banne ...
green chemistry
... 1999 EPA Green Chemistry Award for noncompetitive for treating epilepsy and neurodegenerative diseases was given to Lilly Research Labs for producing Talampanol. This example will explain that how one can minimize the waste, save energy and produce cost ...
... 1999 EPA Green Chemistry Award for noncompetitive for treating epilepsy and neurodegenerative diseases was given to Lilly Research Labs for producing Talampanol. This example will explain that how one can minimize the waste, save energy and produce cost ...
1.1 to 1.4
... - are made up of two or more different compounds and or elements close together. They do not have a constant composition and proportions may vary. Ex. air, coffee, soft drinks. ...
... - are made up of two or more different compounds and or elements close together. They do not have a constant composition and proportions may vary. Ex. air, coffee, soft drinks. ...
Chapter 8
... • A formula equation uses symbols & formulas to represent the identities & relative amounts of reactants & products. • There are also word equations which use chemical names instead of formulas. • See p. 246 Table Reactants (s) → Products (g) Original Substances ...
... • A formula equation uses symbols & formulas to represent the identities & relative amounts of reactants & products. • There are also word equations which use chemical names instead of formulas. • See p. 246 Table Reactants (s) → Products (g) Original Substances ...
StudyGuide_Biochemistry
... 25. How many different amino acids are there? With only so few types of amino acids, how are so many different proteins possible? 26. How many amino acids can the body make? How do we get the remaining amino acids? 27. When will the body use proteins for energy? 28. Describe the 5 types of proteins. ...
... 25. How many different amino acids are there? With only so few types of amino acids, how are so many different proteins possible? 26. How many amino acids can the body make? How do we get the remaining amino acids? 27. When will the body use proteins for energy? 28. Describe the 5 types of proteins. ...
AEED Sustainability - June 17, 2004
... engineering (OSU, PSU) environmental science (PNNL) • The current Oregon business community is supportive and in need: trained, stable workforce technical expertise and consultation (e.g., CAMCOR) • Sustainability represents a critical mechanism for attraction to and growth of the Oregon business co ...
... engineering (OSU, PSU) environmental science (PNNL) • The current Oregon business community is supportive and in need: trained, stable workforce technical expertise and consultation (e.g., CAMCOR) • Sustainability represents a critical mechanism for attraction to and growth of the Oregon business co ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.