Unit Description - Honors Chemistry
... Use the Aufbau Principle, the Pauli Exclusion Principle and Hund’s Rule to write the electron configurations and orbital diagrams of the elements (5.3) Relate valence electrons to Lewis (electron dot) structures (5.3) Describe the ground-state arrangement of electrons in atoms of any element u ...
... Use the Aufbau Principle, the Pauli Exclusion Principle and Hund’s Rule to write the electron configurations and orbital diagrams of the elements (5.3) Relate valence electrons to Lewis (electron dot) structures (5.3) Describe the ground-state arrangement of electrons in atoms of any element u ...
Worksheet
... Part I: Match the name on the right with the correct statement on the left. You will use some names more than once. 1. __________ His model of the atom is the Electron Cloud Model. ...
... Part I: Match the name on the right with the correct statement on the left. You will use some names more than once. 1. __________ His model of the atom is the Electron Cloud Model. ...
The Dalton Thompson 1889 Rutherford Niels Bohr Moseley
... The famous ‘gold foil experiment’ by Rutherford proved the ‘Plum Pudding’ model wrong as not all alpha particles passed through the gold atoms, some were deflected. The positively charged alpha particle hit a positive substance which caused it to be deflected, therefore atoms must have a nucleus whe ...
... The famous ‘gold foil experiment’ by Rutherford proved the ‘Plum Pudding’ model wrong as not all alpha particles passed through the gold atoms, some were deflected. The positively charged alpha particle hit a positive substance which caused it to be deflected, therefore atoms must have a nucleus whe ...
chpt 11 and 12 notes with answers
... Each element has a symbol made from its name; either English or Latin root Rows are the periods that show a pattern of change in physical and chemical properties are you move across left to right Vertical columns are groups/families ◦ Similar chemical/physical prosperities ...
... Each element has a symbol made from its name; either English or Latin root Rows are the periods that show a pattern of change in physical and chemical properties are you move across left to right Vertical columns are groups/families ◦ Similar chemical/physical prosperities ...
The Periodic Table - Mrs Molchany`s Webpage
... Electron affinity does not change greatly as we move down a group. Electron affinity should become more positive (less energy released). Reason: Moving down a group the average distance between the added electron and the nucleus steadily increases, causing the electron-nucleus attraction to decrease ...
... Electron affinity does not change greatly as we move down a group. Electron affinity should become more positive (less energy released). Reason: Moving down a group the average distance between the added electron and the nucleus steadily increases, causing the electron-nucleus attraction to decrease ...
Ch 3: Atomic Structure - Teach-n-Learn-Chem
... Excited Atoms and the Fourth of July 1. What is light, and how do various colors of light differ? 2. What is going on at the level of atoms and molecules when fireworks produce colored light? 3. How does the instability of copper chloride at high temperatures ineterfere with its ability to emit blue ...
... Excited Atoms and the Fourth of July 1. What is light, and how do various colors of light differ? 2. What is going on at the level of atoms and molecules when fireworks produce colored light? 3. How does the instability of copper chloride at high temperatures ineterfere with its ability to emit blue ...
Atomic structure unit powerpoint
... elements with similar properties were vertically aligned with each other. In making such alignments Mendeleev was able to determine that several, as yet unidentified, elements should exist (the elements with masses 44, 68 and 72 are examples). He went on to make predictions about the properties of t ...
... elements with similar properties were vertically aligned with each other. In making such alignments Mendeleev was able to determine that several, as yet unidentified, elements should exist (the elements with masses 44, 68 and 72 are examples). He went on to make predictions about the properties of t ...
Ch 3: Atomic Structure - Teach-n-Learn-Chem
... Use the periodic table to write the electron configurations and orbital diagrams for various atoms. ATOMIC NUMBER AND MASS NUMBER Each element has an atomic number Atomic number - the number of protons in the nucleus of an atom Isotope - one or two or more atoms having the same number of protons bu ...
... Use the periodic table to write the electron configurations and orbital diagrams for various atoms. ATOMIC NUMBER AND MASS NUMBER Each element has an atomic number Atomic number - the number of protons in the nucleus of an atom Isotope - one or two or more atoms having the same number of protons bu ...
1st Semester Review - Moore Public Schools
... 35. Practice writing at least 20 formulas given the name of compounds, and writing 20 names given the correct formulas. Have someone else make the formulas/ names for you to do. 36. Practice writing Lewis Structures, and identifying the electron domain geometry, molecular geometry, intermolecular fo ...
... 35. Practice writing at least 20 formulas given the name of compounds, and writing 20 names given the correct formulas. Have someone else make the formulas/ names for you to do. 36. Practice writing Lewis Structures, and identifying the electron domain geometry, molecular geometry, intermolecular fo ...
C2.1 Key Terms Atomic number: The number of protons in the
... The number of protons in the nucleus of an atom. Atoms: The smallest part of an element that can take part in chemical reactions. Electronic configuration: The arrangement of electrons in shells around the nucleus of an atom. Electrons: Negative particles of negligible mass and charge -1 (relative t ...
... The number of protons in the nucleus of an atom. Atoms: The smallest part of an element that can take part in chemical reactions. Electronic configuration: The arrangement of electrons in shells around the nucleus of an atom. Electrons: Negative particles of negligible mass and charge -1 (relative t ...
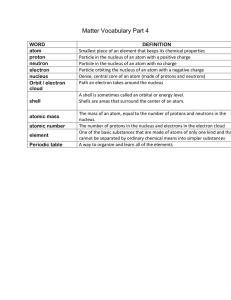
Matter Vocab Part 4
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
Chemistry: The Nature of Matter
... o 2nd shell has a little more energy and holds 8 electrons o 3rd shell has even more energy, etc. ____________________________________________________________ ____________________________________________________________ ____________________________________________________________ Electron config ...
... o 2nd shell has a little more energy and holds 8 electrons o 3rd shell has even more energy, etc. ____________________________________________________________ ____________________________________________________________ ____________________________________________________________ Electron config ...
At. Theory Timeline - Advanced Placement Chemistry
... 2. The atoms of a given element are identical in average atomic mass and properties; atoms of different elements are different in some fundamental way (or ways). 3. Chemical compounds are formed when atoms combine with each other. A given compound always has the same relative numbers and types of at ...
... 2. The atoms of a given element are identical in average atomic mass and properties; atoms of different elements are different in some fundamental way (or ways). 3. Chemical compounds are formed when atoms combine with each other. A given compound always has the same relative numbers and types of at ...
CHAPTER 2: ATOMS, IONS, AND COMPOUNDS
... water, fire, and earth. Aristotle (384-321 B.C.): accepted Empedocles idea and added a fifth element, heavenly ether, which is perfect, eternal, and incorruptible. Aristotle’s idea of five basic elements was accepted for 2000 years. John Dalton (1766-1844), an English chemist and physicist, establis ...
... water, fire, and earth. Aristotle (384-321 B.C.): accepted Empedocles idea and added a fifth element, heavenly ether, which is perfect, eternal, and incorruptible. Aristotle’s idea of five basic elements was accepted for 2000 years. John Dalton (1766-1844), an English chemist and physicist, establis ...
Semester 1 Final Exam Study Guide
... 42. Write the noble gas shortcut electron configuration for calcium. ...
... 42. Write the noble gas shortcut electron configuration for calcium. ...
Chapter 18: Atoms and Elements
... Understand how elements are organized in the periodic table. Use the periodic table to identify the atomic number and mass numbers of each element. Calculate the numbers of protons and neutrons in each stable isotope of an element. ...
... Understand how elements are organized in the periodic table. Use the periodic table to identify the atomic number and mass numbers of each element. Calculate the numbers of protons and neutrons in each stable isotope of an element. ...
Unit 4 – Atomic Structure Study Guide
... The atom remains in the excited state as long there is energy to absorb When there is no longer energy to absorb, the electron loses energy, which it emits usually as heat or light, and the atom returns to the ground state 15. Describe the shape of and maximum number of electrons in the s and p ...
... The atom remains in the excited state as long there is energy to absorb When there is no longer energy to absorb, the electron loses energy, which it emits usually as heat or light, and the atom returns to the ground state 15. Describe the shape of and maximum number of electrons in the s and p ...
Any substance that cannot be decomposed into
... arranged in layers or shells. An atom can have as many as seven shells, each of which holds only a certain number of electrons. The shells, in sequence from the closest to the furthest from the nucleus, hold a maximum of 2, 8, 18, 32, 50, 72, and 98 electrons each. The lightest element, hydrogen, ha ...
... arranged in layers or shells. An atom can have as many as seven shells, each of which holds only a certain number of electrons. The shells, in sequence from the closest to the furthest from the nucleus, hold a maximum of 2, 8, 18, 32, 50, 72, and 98 electrons each. The lightest element, hydrogen, ha ...
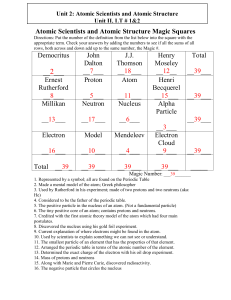
Atomic Scientists and Atomic Structure Magic Squares
... 5. The positive particle in the nucleus of an atom. (Not a fundamental particle) 6. The tiny positive core of an atom; contains protons and neutrons. 7. Credited with the first atomic theory model of the atom which had four main postulates. 8. Discovered the nucleus using his gold foil experiment. 9 ...
... 5. The positive particle in the nucleus of an atom. (Not a fundamental particle) 6. The tiny positive core of an atom; contains protons and neutrons. 7. Credited with the first atomic theory model of the atom which had four main postulates. 8. Discovered the nucleus using his gold foil experiment. 9 ...
Year 9 Science Revision Unit: Elements NGA PUMOTU O
... 1. Each column in a periodic table is called a group. Elements in a group have similar chemical reactions. 2. Each row in a periodic table is called a period. As you move across a period, the number of electrons increases until the electron shell is full at the end of the period. Distinguishing betw ...
... 1. Each column in a periodic table is called a group. Elements in a group have similar chemical reactions. 2. Each row in a periodic table is called a period. As you move across a period, the number of electrons increases until the electron shell is full at the end of the period. Distinguishing betw ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.