* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
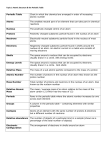
Download C2.1 Key Terms Atomic number: The number of protons in the
Survey
Document related concepts
Transcript
Atomic number: The number of protons in the nucleus of an atom. Atoms: The smallest part of an element that can take part in chemical reactions. Electronic configuration: The arrangement of electrons in shells around the nucleus of an atom. Electrons: Negative particles of negligible mass and charge -1 (relative to a proton). Energy levels: In chemistry, space around a nucleus that can be occupied by electrons, usually drawn as a circle. Also referred to as shells. Groups: Columns in the periodic table, containing elements with similar properties. Isotopes: Atoms of an element with the same number of protons and electrons but with a different number of neutrons. Mass number: The total number of protons and neutrons in the nucleus of an atom. Neutrons: Particles found in the nucleus, having zero charge and mass of 1 (relative to a proton). Nucleus: In chemistry, the positively charged centre of an atom. Periodic table: Chart in which the chemical elements are arranged in order of increasing atomic number. Periods: Rows in the periodic table in which the atomic number increases by one from one element to the next. Protons: Positive particles found in the nucleus, having a charge of +1 and a mass of 1. Relative abundance: The number of objects of a particular kind in a sample shown as a percentage of the total number of objects in the sample. Relative atomic mass: The mean mass of an atom relevant to the mass of an atom of carbon-12, which is assigned a mass of 12. Relative charge: The electrical charge of a subatomic particle compared to the electrical charge of a proton. Shells: In chemistry, space around a nucleus that can be occupied by electrons, usually drawn as a circle. Also referred to as energy levels.