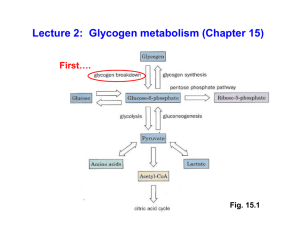
Lecture 2: Glycogen metabolism (Chapter 15)
... UDPG is transferred to the C(4)-OH on one of the non-reducing ends of glycogen, forming an (1 4) glycosidic bond. Note that this step makes -amylose, not the branched structure of glycogen. The ∆G°’ for this reaction is -13.7 kJ/mol, making this reaction spontaneous (exergonic) under the same condit ...
... UDPG is transferred to the C(4)-OH on one of the non-reducing ends of glycogen, forming an (1 4) glycosidic bond. Note that this step makes -amylose, not the branched structure of glycogen. The ∆G°’ for this reaction is -13.7 kJ/mol, making this reaction spontaneous (exergonic) under the same condit ...
Ques#on of the Day: How do you acquire energy?
... “Appears” to be the reverse of photosynthesis ...
... “Appears” to be the reverse of photosynthesis ...
4 ATP - OoCities
... - O2 & glucose enter the body and produce glycolysis & pyruvate - these products enter mitochondria to be breakdown - pyruvate turns to CO2 and H2O to produce ATP - CO2 & ATP diffuse out to cytoplasm &ATP is use for energy ...
... - O2 & glucose enter the body and produce glycolysis & pyruvate - these products enter mitochondria to be breakdown - pyruvate turns to CO2 and H2O to produce ATP - CO2 & ATP diffuse out to cytoplasm &ATP is use for energy ...
Glucose Metabolism Glycolysis Expectations
... – Synthesis of “reducing potential” – Synthesis of 5‐carbon sugars ...
... – Synthesis of “reducing potential” – Synthesis of 5‐carbon sugars ...
Chapter 6
... The baking and winemaking industry have used alcohol fermentation for thousands of years ...
... The baking and winemaking industry have used alcohol fermentation for thousands of years ...
Hemoglobin as the main protein of erythrocytes. Its structure and
... their biological role. Glucose as a source of fat synthesis in liver and adipose tissue. The scheme of carbohydrate transformation into lipids. Cholesterol as a precursor for other steroids. Cholesterol synthesis: reaction sequence up to mevalonic acid formation, the idea about further stages of syn ...
... their biological role. Glucose as a source of fat synthesis in liver and adipose tissue. The scheme of carbohydrate transformation into lipids. Cholesterol as a precursor for other steroids. Cholesterol synthesis: reaction sequence up to mevalonic acid formation, the idea about further stages of syn ...
Phosphoketolase pathway dominates in
... the dry weights of the samples, and, hence, the signal-to-noise levels vary between the spectra. Duplicate samples from different batch cultures were analyzed in this study, and the spectra showed the same results. Enzyme assays. Cell extracts were prepared from cultures harvested in the late expone ...
... the dry weights of the samples, and, hence, the signal-to-noise levels vary between the spectra. Duplicate samples from different batch cultures were analyzed in this study, and the spectra showed the same results. Enzyme assays. Cell extracts were prepared from cultures harvested in the late expone ...
Notes CH 7 - Haiku Learning
... a) Glucose is oxidized because electrons are transferred from it to oxygen. b) The protons follow the electrons to produce water. c) Oxygen atoms on the reactant sided are reduced d) Large drop in potential energy of the compounds on the product side of the equation ...
... a) Glucose is oxidized because electrons are transferred from it to oxygen. b) The protons follow the electrons to produce water. c) Oxygen atoms on the reactant sided are reduced d) Large drop in potential energy of the compounds on the product side of the equation ...
9 and 10 notes with blanks
... Oxidative phosphorylation (accounts for most of the ATP synthesis) Oxidative phosphorylation accounts for almost 90% of the ATP generated by cellular respiration A smaller amount of ATP is formed in glycolysis and the citric acid cycle by substrate-level phosphorylation For each molecule of glucose ...
... Oxidative phosphorylation (accounts for most of the ATP synthesis) Oxidative phosphorylation accounts for almost 90% of the ATP generated by cellular respiration A smaller amount of ATP is formed in glycolysis and the citric acid cycle by substrate-level phosphorylation For each molecule of glucose ...
CELLULAR RESPIRATION - Ms. Tripp
... • The movement of electrons from one molecule to another is an oxidation-reduction reaction (redox). – the loss of electrons from one substance is oxidation – the addition of electrons to another substance is reduction • Cellular respiration uses redox reactions to harvest the chemical energy stored ...
... • The movement of electrons from one molecule to another is an oxidation-reduction reaction (redox). – the loss of electrons from one substance is oxidation – the addition of electrons to another substance is reduction • Cellular respiration uses redox reactions to harvest the chemical energy stored ...
Biochemistry II Test 2Q
... Fatty acid synthase has ___ enzymes and is dimer that contains an ___________ protein. To produce 1 palmitate you need __ AceCoA and __ malonyl CoA thus releasing _____. To produce Malonyl CoA you need an ATP, thus how may ATP are needed in Palmitate synth? 2NADPH are needed per Malonyl, thus how mu ...
... Fatty acid synthase has ___ enzymes and is dimer that contains an ___________ protein. To produce 1 palmitate you need __ AceCoA and __ malonyl CoA thus releasing _____. To produce Malonyl CoA you need an ATP, thus how may ATP are needed in Palmitate synth? 2NADPH are needed per Malonyl, thus how mu ...
1 category question correct answer your answer
... A) Glycogen metabolism traps glucose within liver cells and increases storage of glucose in the form of glycogen. These processes decrease blood glucose levels. A) Glycogen metabolism traps glucose within liver cells and increases storage of glucose in the form of glycogen. These processes decrease ...
... A) Glycogen metabolism traps glucose within liver cells and increases storage of glucose in the form of glycogen. These processes decrease blood glucose levels. A) Glycogen metabolism traps glucose within liver cells and increases storage of glucose in the form of glycogen. These processes decrease ...
Nehru Arts Science and College Reaccredited with “A” Grade by
... 19. The polysaccharide found in the exoskeleton of invertebrates is (1) Pectin (2) Chitin (3) Cellulose (4) Chondroitin sulphate 20. The most abundant carbohydrate found in nature is (1) Starch (2) Glycogen (3) Cellulose (4) Chitin 21. Iodine gives a blue colour with (1) Starch (2) Glycogen (3) Cell ...
... 19. The polysaccharide found in the exoskeleton of invertebrates is (1) Pectin (2) Chitin (3) Cellulose (4) Chondroitin sulphate 20. The most abundant carbohydrate found in nature is (1) Starch (2) Glycogen (3) Cellulose (4) Chitin 21. Iodine gives a blue colour with (1) Starch (2) Glycogen (3) Cell ...
Sustained nonoxidative glucose utilization and depletion of
... tion dogs received a priming bolus of 20 &i of L-[1-‘4C]lactic acid (New England Nuclear Corp.; specific activity 55 mCi/mmol) followed by constant intravenous infusion at a rate of 25 &i/h. After an equilibration period of 25 min (16), arterial and venous samples were withdrawn as outlined to deter ...
... tion dogs received a priming bolus of 20 &i of L-[1-‘4C]lactic acid (New England Nuclear Corp.; specific activity 55 mCi/mmol) followed by constant intravenous infusion at a rate of 25 &i/h. After an equilibration period of 25 min (16), arterial and venous samples were withdrawn as outlined to deter ...
1 The diagram below represents a biological process 5
... polysaccharide 4) ADP 19. Lipase, maltase, and protease are members of a group of catalysts known as 1) enzymes 3) carbohydrates 2) hormones 4) fats 20. Hydrogen peroxide (H2O2) is a toxic by-product of cellular metabolism in aerobic organisms. The reaction below occurs within the cells to prevent t ...
... polysaccharide 4) ADP 19. Lipase, maltase, and protease are members of a group of catalysts known as 1) enzymes 3) carbohydrates 2) hormones 4) fats 20. Hydrogen peroxide (H2O2) is a toxic by-product of cellular metabolism in aerobic organisms. The reaction below occurs within the cells to prevent t ...
Chapter 1 Notes
... Polysaccharides: polymers w/ few hundred to a few thousand monomers -fcn. of a polysaccharide is determined by monomers and positions of glycosidic linkages ...
... Polysaccharides: polymers w/ few hundred to a few thousand monomers -fcn. of a polysaccharide is determined by monomers and positions of glycosidic linkages ...
HMP SHUNT & URONIC ACID PATHWAY
... • Essential Pentosuria: one of Garrod’s tetrad [alkaptonuria, albinism, pentosuria, cystinuria- inborn error of metabolism]: *1 in 2500 births due to deficiency of xylitol dehydrogenase → L-xylulose excreted in urine gives + benedict’s test-not harmful. *Diffentiated from DM by + Bials test [orcinol ...
... • Essential Pentosuria: one of Garrod’s tetrad [alkaptonuria, albinism, pentosuria, cystinuria- inborn error of metabolism]: *1 in 2500 births due to deficiency of xylitol dehydrogenase → L-xylulose excreted in urine gives + benedict’s test-not harmful. *Diffentiated from DM by + Bials test [orcinol ...
APchapter5notes
... Polysaccharides: polymers w/ few hundred to a few thousand monomers -fcn. of a polysaccharide is determined by monomers and positions of glycosidic linkages ...
... Polysaccharides: polymers w/ few hundred to a few thousand monomers -fcn. of a polysaccharide is determined by monomers and positions of glycosidic linkages ...
Respiration - WordPress.com
... The Metabolic Process responsible for the majority of ATP Production within living organisms in order to produce energy. It involves the breaking down of Glucose molecules to form ATP and other products. Consists of Four Stages in Aerobic respiration and only One Stage in Anaerobic Respiration. ...
... The Metabolic Process responsible for the majority of ATP Production within living organisms in order to produce energy. It involves the breaking down of Glucose molecules to form ATP and other products. Consists of Four Stages in Aerobic respiration and only One Stage in Anaerobic Respiration. ...
Glucose

Glucose is a sugar with the molecular formula C6H12O6. The name ""glucose"" (/ˈɡluːkoʊs/) comes from the Greek word γλευκος, meaning ""sweet wine, must"". The suffix ""-ose"" is a chemical classifier, denoting a carbohydrate. It is also known as dextrose or grape sugar. With 6 carbon atoms, it is classed as a hexose, a sub-category of monosaccharides. α-D-glucose is one of the 16 aldose stereoisomers. The D-isomer (D-glucose) occurs widely in nature, but the L-isomer (L-glucose) does not. Glucose is made during photosynthesis from water and carbon dioxide, using energy from sunlight. The reverse of the photosynthesis reaction, which releases this energy, is a very important source of power for cellular respiration. Glucose is stored as a polymer, in plants as starch and in animals as glycogen.