Many metal ions (usually transition metals but including a few others
... The anions / molecules are called ligands The coordination number is the number of places on the metal ion where ligands are bound. The bond between the metal ion and the ligand, where the ligand supplies both electrons, is known as a coordinate covalent bond ...
... The anions / molecules are called ligands The coordination number is the number of places on the metal ion where ligands are bound. The bond between the metal ion and the ligand, where the ligand supplies both electrons, is known as a coordinate covalent bond ...
Rules for filling and removal of d-electrons For filling the orbital order
... any complex. This requires being able to recognize all the entities making up the complex and knowing whether the ligands are neutral or anionic, so that you can determine the oxidation state of the metal ion. In many cases the oxidation state for first row transition metal ions will be either (II) ...
... any complex. This requires being able to recognize all the entities making up the complex and knowing whether the ligands are neutral or anionic, so that you can determine the oxidation state of the metal ion. In many cases the oxidation state for first row transition metal ions will be either (II) ...
Basic Chemistry and Biochemistry Unit Review Sheet File
... A. Type of reaction by which complex molecules are synthesized from simple molecules B. A substance composed of similar repeating units. C. Proteins that act as organic catalysts. D. Digestion is accomplished by this type of reaction. E. COOH F. RNA and DNA G. Alcohol found in lipids. H. Monomer of ...
... A. Type of reaction by which complex molecules are synthesized from simple molecules B. A substance composed of similar repeating units. C. Proteins that act as organic catalysts. D. Digestion is accomplished by this type of reaction. E. COOH F. RNA and DNA G. Alcohol found in lipids. H. Monomer of ...
Redox - Plusnet
... number does not necessarily mean that electrons have been lent and borrowed. E.g. in CrO42ˉ the oxidation number of chromium is +6, yet it is covalently bonded to the oxygens and the energy required to remove 6 electrons would be prohibitive. All the +6 tells us is that the electrons probably spend ...
... number does not necessarily mean that electrons have been lent and borrowed. E.g. in CrO42ˉ the oxidation number of chromium is +6, yet it is covalently bonded to the oxygens and the energy required to remove 6 electrons would be prohibitive. All the +6 tells us is that the electrons probably spend ...
New Reaction Chemistries
... (Re)Engineering cofactors - NAD(P)H/FAD/etc CF3 methylation CF3./radical SAM’s/CF3CO2H CF3./Hybrid/chimeric enzymes Modular/Non-covalent ...
... (Re)Engineering cofactors - NAD(P)H/FAD/etc CF3 methylation CF3./radical SAM’s/CF3CO2H CF3./Hybrid/chimeric enzymes Modular/Non-covalent ...
Document
... electrons to a non-metal in an ionic bond electrons are shared in a polar covalent bond. • For example Fe(III) chloride FeCl3 has a melting point of only 306oC - it sublimes when it forms by heating iron with chlorine. By comparison NaCl has a melting point of 806oC, and iron (II) chloride 672oC: he ...
... electrons to a non-metal in an ionic bond electrons are shared in a polar covalent bond. • For example Fe(III) chloride FeCl3 has a melting point of only 306oC - it sublimes when it forms by heating iron with chlorine. By comparison NaCl has a melting point of 806oC, and iron (II) chloride 672oC: he ...
Worksheet 3 - contentextra
... Oxy acid An acid in which the acidic proton is covalently bonded to an oxygen atom. Period A period is a horizontal row of elements in the Periodic Table. Within a Period, the atoms of the elements have the same number of occupied energy levels but an increasing number of electrons in the outer ener ...
... Oxy acid An acid in which the acidic proton is covalently bonded to an oxygen atom. Period A period is a horizontal row of elements in the Periodic Table. Within a Period, the atoms of the elements have the same number of occupied energy levels but an increasing number of electrons in the outer ener ...
Biology Study Guide for Section (Macromolecules) Test
... Monosaccharide- A simple sugar like glucose. Carbohydrate- A type of macromolecule that is produced by plants during photosynthesis. Glycogen- Animal cells store excess sugar as molecules of this. Polysaccharide- Monosaccharides may bond together to form these molecules. Energy-The main function of ...
... Monosaccharide- A simple sugar like glucose. Carbohydrate- A type of macromolecule that is produced by plants during photosynthesis. Glycogen- Animal cells store excess sugar as molecules of this. Polysaccharide- Monosaccharides may bond together to form these molecules. Energy-The main function of ...
A typical hemoglobin molecule consists of a "heme" part and a
... A typical hemoglobin molecule consists of a "heme" part and a protein part=globin. The "heme" part lies in the interior of the hemoglobin molecule. At the center of the heme is an Fe(II), ferrous, atom. Four of the six coordination sites around this atom are occupied by nitrogen atoms from a porphyr ...
... A typical hemoglobin molecule consists of a "heme" part and a protein part=globin. The "heme" part lies in the interior of the hemoglobin molecule. At the center of the heme is an Fe(II), ferrous, atom. Four of the six coordination sites around this atom are occupied by nitrogen atoms from a porphyr ...
Chapter 3 Test Review
... Carbon Hydrogen Oxygen Ratio is 1 Carbon : 2 Hydrogen : 1 Oxygen The large molecules can be broken down into smaller that are exactly alike. ...
... Carbon Hydrogen Oxygen Ratio is 1 Carbon : 2 Hydrogen : 1 Oxygen The large molecules can be broken down into smaller that are exactly alike. ...
Chapter 24: Transition Metals Coordination Compounds Part 1
... The covalently attached ions or molecules are called ligands. The ligand atom which is directly connected to the metal atom/ion is called the ligand donor atom or just the donor atom. So in the above, ammonia is the ligand. If the complex is an ion, like the above, it is written in brackets as shown ...
... The covalently attached ions or molecules are called ligands. The ligand atom which is directly connected to the metal atom/ion is called the ligand donor atom or just the donor atom. So in the above, ammonia is the ligand. If the complex is an ion, like the above, it is written in brackets as shown ...
which line the 3-fold and 4-fold axes, through which iron may
... a protein cage encompassing an «iron-core» of the mineral ferrihydrite complexed with phosphate [1]. The space for this mineral is a sphere of ca. 80 A diameter allowing the storage of up to about 4500 Fe 3 + atoms. The cage, apoferritin, is a nearly spherical protein shell of thickness about 25 A. ...
... a protein cage encompassing an «iron-core» of the mineral ferrihydrite complexed with phosphate [1]. The space for this mineral is a sphere of ca. 80 A diameter allowing the storage of up to about 4500 Fe 3 + atoms. The cage, apoferritin, is a nearly spherical protein shell of thickness about 25 A. ...
General Properties of Transition Metals
... • Transition metals can form complexes because their ions have a high charge density: o they have quite a large nuclear charge but are relatively small; o the 3d electrons are not so effective (as 2s or 2p electrons) at shielding the effect of the ionic charge which really comes from the nucleus. ...
... • Transition metals can form complexes because their ions have a high charge density: o they have quite a large nuclear charge but are relatively small; o the 3d electrons are not so effective (as 2s or 2p electrons) at shielding the effect of the ionic charge which really comes from the nucleus. ...
A Practice Reactions Quiz -
... A) Write complete balanced equations for the following reactions. B) Label each reaction as either SYN, DEC, SR, DR, or COMB. C) Place a star next to any reaction which required knowledge of oxidation numbers. D) Finally, find the two reactions below which do not actually take place. Write “NR” and ...
... A) Write complete balanced equations for the following reactions. B) Label each reaction as either SYN, DEC, SR, DR, or COMB. C) Place a star next to any reaction which required knowledge of oxidation numbers. D) Finally, find the two reactions below which do not actually take place. Write “NR” and ...
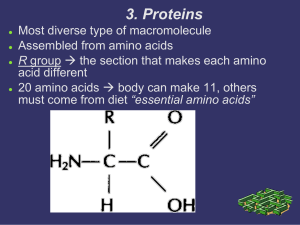
Proteins have a higher order of folding known as tertiary structure
... You should know that hydrogen bonds between the polypeptide backbone are responsible for maintaining a protein’s secondary structure. However, many amino acids have side chains which contain a hydrogen atom attached to an oxygen or nitrogen, and can therefore form hydrogen bonds. For example, serine ...
... You should know that hydrogen bonds between the polypeptide backbone are responsible for maintaining a protein’s secondary structure. However, many amino acids have side chains which contain a hydrogen atom attached to an oxygen or nitrogen, and can therefore form hydrogen bonds. For example, serine ...
Beta sheets are twisted
... loop regions connecting alpha-helical segments can have important functions e.g. EF-hand and DNA-binding EF hand loop ~ 12 residues polar and hydrophobic a.a. conserved positions Glycine is invariant at the sixth position The calcium ion is octahedrally coordinated by carboxyl side chains, main chai ...
... loop regions connecting alpha-helical segments can have important functions e.g. EF-hand and DNA-binding EF hand loop ~ 12 residues polar and hydrophobic a.a. conserved positions Glycine is invariant at the sixth position The calcium ion is octahedrally coordinated by carboxyl side chains, main chai ...
lecture1 - Unaab.edu.ng
... (iii) Electron transfer – metal-containing electron transfer agents such as ferrodoxins (Fe) and many copper-containing “blue proteins” are involved in electron transfer chemistry that goes on in the biological systems. (iv) Metalloenzymes and metallocoenzymes have metal ions at their active sites. ...
... (iii) Electron transfer – metal-containing electron transfer agents such as ferrodoxins (Fe) and many copper-containing “blue proteins” are involved in electron transfer chemistry that goes on in the biological systems. (iv) Metalloenzymes and metallocoenzymes have metal ions at their active sites. ...
Polyatomic Ions Writing Formulas / Naming Compounds
... • Write the formula for the metal. Add the oxidation number from the periodic table. Ca+2 • Write the formula for the polyatomic ion from the ion chart. Add its oxidation number. PO4-3 ...
... • Write the formula for the metal. Add the oxidation number from the periodic table. Ca+2 • Write the formula for the polyatomic ion from the ion chart. Add its oxidation number. PO4-3 ...
InorgCh425
... The rates of reaction depend on the ability of the electron to “tunnel” through the ligands from one metal to the other i. Tunneling = moving through an energy barrier (the ligands) that is normally too high to allow the electron to pass through. This is a quantum mechanical process having to do wit ...
... The rates of reaction depend on the ability of the electron to “tunnel” through the ligands from one metal to the other i. Tunneling = moving through an energy barrier (the ligands) that is normally too high to allow the electron to pass through. This is a quantum mechanical process having to do wit ...
NAME: IDU DOREEN MATRIC NO: 14/SCI03/011 COURSE
... Metal/Ligand Substitution Metalloenzymes frequently contain metal cofactors that are spectroscopically useless. For example, zinc enzymes are diamagnetic and colourless – electron spin resonance or electronic absorption spectroscopy are therefore uninformative. However, information about the enviro ...
... Metal/Ligand Substitution Metalloenzymes frequently contain metal cofactors that are spectroscopically useless. For example, zinc enzymes are diamagnetic and colourless – electron spin resonance or electronic absorption spectroscopy are therefore uninformative. However, information about the enviro ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.