
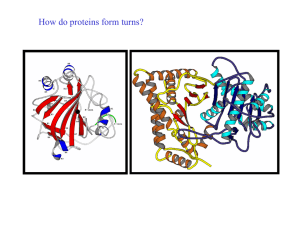
How do proteins form turns? - UF Macromolecular Structure Group
... The loop regions connecting alpha-helical segments can have important functions ...
... The loop regions connecting alpha-helical segments can have important functions ...
013368718X_CH28_437
... atom of calcium (atomic number 20) bonds to two atoms of chlorine (atomic number 17). Fill in the number of protons and electrons in each ion. ...
... atom of calcium (atomic number 20) bonds to two atoms of chlorine (atomic number 17). Fill in the number of protons and electrons in each ion. ...
C h e m g u i d e ... COMPLEX IONS - INTRODUCTION
... a) Use the diagram to explain what is meant by the term ligands. b) What is the essential feature of a molecule or ion which can serve as a ligand? c) What sort of bonding is there between the ligand and the metal ion? d) What is the coordination number of the aluminium in this ion? e) Explain what ...
... a) Use the diagram to explain what is meant by the term ligands. b) What is the essential feature of a molecule or ion which can serve as a ligand? c) What sort of bonding is there between the ligand and the metal ion? d) What is the coordination number of the aluminium in this ion? e) Explain what ...
1 [L 5 FeO 2 ] - physics.muni.cz
... Water solubility of oxygen at 25oC and pressure = 1 bar is at 40 mg/L water. This is not enough to guarantee the oxygen supply to mitochondria by mere diffusion. Cells of aerobic organisms use therefore oxygen transporters. ...
... Water solubility of oxygen at 25oC and pressure = 1 bar is at 40 mg/L water. This is not enough to guarantee the oxygen supply to mitochondria by mere diffusion. Cells of aerobic organisms use therefore oxygen transporters. ...
problem set 3 2016
... 2. Balance the following equations using the half-reaction method a. Fe(s) + HCl(aq) HFeCl4(aq) + H2(g) b. IO3-(aq) + I-(aq) I33. Gold metal will not dissolve in either concentrated nitric acid or concentrated hydrochloric acid. It will dissolve, however, in aqua regia, a mixture of the two conc ...
... 2. Balance the following equations using the half-reaction method a. Fe(s) + HCl(aq) HFeCl4(aq) + H2(g) b. IO3-(aq) + I-(aq) I33. Gold metal will not dissolve in either concentrated nitric acid or concentrated hydrochloric acid. It will dissolve, however, in aqua regia, a mixture of the two conc ...
Biochemistry
... •Fats and oils – store long term energy Found in animal fats, vegetable oil, petroleum. ...
... •Fats and oils – store long term energy Found in animal fats, vegetable oil, petroleum. ...
Ch. 6 Vocabulary
... Products – the substances formed during a reaction Activation Energy – the minimum amount of energy needed for reactants to form products in a chemical reaction Catalyst – a substance that lowers the activation energy needed to start a chemical reaction Enzymes – a protein and biological catalysts t ...
... Products – the substances formed during a reaction Activation Energy – the minimum amount of energy needed for reactants to form products in a chemical reaction Catalyst – a substance that lowers the activation energy needed to start a chemical reaction Enzymes – a protein and biological catalysts t ...
Chemistry Revision
... its outer shell) to be come positively charged by three. It has done this because it still has the original number of protons (13) which are positively charged. What is the difference between 12 C and 13 C? What is one way the atoms will be physically different? What charge do isotopes have? Why? ...
... its outer shell) to be come positively charged by three. It has done this because it still has the original number of protons (13) which are positively charged. What is the difference between 12 C and 13 C? What is one way the atoms will be physically different? What charge do isotopes have? Why? ...
Lecture 2
... Submit a list of projects that you judged with the associated grade-levels. A brief description of your impression of the projects would be helpful. (Be in SUB Ballroom by 5:00 PM for dinner and instructions.) If you are not a judge, you may attend the Science Expo and write a detailed summary of ...
... Submit a list of projects that you judged with the associated grade-levels. A brief description of your impression of the projects would be helpful. (Be in SUB Ballroom by 5:00 PM for dinner and instructions.) If you are not a judge, you may attend the Science Expo and write a detailed summary of ...
Naming Ionic Compounds Many Ionic compounds contain
... Polyatomic ions exist as a unit, so never change the subscript o If you have to balance an ionic compound with a polyatomic ion then ( )and a subscript must be written Ex. Ca (+2) and PO4 (-3) →Ca3(PO4)2 and named Calcium phosphate o Most polyatomic ions are oxyanions Oxyanion is a polyatomic ...
... Polyatomic ions exist as a unit, so never change the subscript o If you have to balance an ionic compound with a polyatomic ion then ( )and a subscript must be written Ex. Ca (+2) and PO4 (-3) →Ca3(PO4)2 and named Calcium phosphate o Most polyatomic ions are oxyanions Oxyanion is a polyatomic ...
Biologist Date ______ CHAPTER 2 STUDY GUIDE 2.1 What are
... 7. What is an acid? What ion do they contain more of? What numbers on the pH scale are acids? ...
... 7. What is an acid? What ion do they contain more of? What numbers on the pH scale are acids? ...
effective oxidation states applied to endohedral - IQCC
... rationalized on the basis of the OS of the metal center. OS are intrinsically related to the electronic distribution, but electronic or spin populations are only a pointer of the atom's OS. We have most recently shown that the so-called effective atomic orbitals (eff-AO's) can be utilized, treating ...
... rationalized on the basis of the OS of the metal center. OS are intrinsically related to the electronic distribution, but electronic or spin populations are only a pointer of the atom's OS. We have most recently shown that the so-called effective atomic orbitals (eff-AO's) can be utilized, treating ...
An_explanation_of_the_pH_scale
... ion is an atom or a group of atoms that carries a positive or a negative charge as a result of having lost or gained one or more electrons. A free electron or other subatomic-charged particle is also referred to as an ion. ...
... ion is an atom or a group of atoms that carries a positive or a negative charge as a result of having lost or gained one or more electrons. A free electron or other subatomic-charged particle is also referred to as an ion. ...
Chemistry of Life - Haughton Science
... together in protein molecules dipeptide bond = two connected amino acids polypeptide bond = 3 or more connected amino acids ...
... together in protein molecules dipeptide bond = two connected amino acids polypeptide bond = 3 or more connected amino acids ...
Transition Metals & Complex ions
... a) One copper (II) ion & four chloride ions b) One iron (III) ion & five water molecules & a chloride ion. c) One copper (II) ion & four ammonia molecules & two hydroxide ions d) One cobalt (III) ion & four hydroxide ions ...
... a) One copper (II) ion & four chloride ions b) One iron (III) ion & five water molecules & a chloride ion. c) One copper (II) ion & four ammonia molecules & two hydroxide ions d) One cobalt (III) ion & four hydroxide ions ...
CHEM1102 Worksheet 12: Coordination Chemistry Model 1: The
... The sum of the charges of the metal cation and its ligands adds up to give the charge of the complex ion. If the complex ion is charged, this is balanced by counter ions. The number of valence electrons on an atom is equal to its group number. In a cation, the oxidation number is equal to the number ...
... The sum of the charges of the metal cation and its ligands adds up to give the charge of the complex ion. If the complex ion is charged, this is balanced by counter ions. The number of valence electrons on an atom is equal to its group number. In a cation, the oxidation number is equal to the number ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.




![1 [L 5 FeO 2 ] - physics.muni.cz](http://s1.studyres.com/store/data/000263887_1-9a7fea8feae8a4c4c33cd53b2038de6b-300x300.png)

















