Fe 4 S 4 Cys 4
... • Must avoid oxidation to Fe(III) (Met-hemoglobin) • Neutral His ligand: His-Fe(II)-porphyrin is uncharged: Favourable • P450: Catalyses hydroxylation of hydrophobic substrates. Also 5-coordinate • 1 axial Cys thiolate ligand (negatively charged): Resting state is Fe(III), also uncharged • In cytoch ...
... • Must avoid oxidation to Fe(III) (Met-hemoglobin) • Neutral His ligand: His-Fe(II)-porphyrin is uncharged: Favourable • P450: Catalyses hydroxylation of hydrophobic substrates. Also 5-coordinate • 1 axial Cys thiolate ligand (negatively charged): Resting state is Fe(III), also uncharged • In cytoch ...
PP Ch_ 2-3 Modified - Maria Regina High School
... enzymes (catalysts), some are used for transport around an organism The Shape of a protein is incredibly important ...
... enzymes (catalysts), some are used for transport around an organism The Shape of a protein is incredibly important ...
hard acid
... The word ligand is derived from the Latin verb ‘ligare’ meaning ‘to bind’. In a coordination complex, a central atom or ion is coordinated by one or more molecules or ions (ligands) which act as Lewis bases, forming coordinate bonds with the central atom or ion; the latter acts as a Lewis acid. Atom ...
... The word ligand is derived from the Latin verb ‘ligare’ meaning ‘to bind’. In a coordination complex, a central atom or ion is coordinated by one or more molecules or ions (ligands) which act as Lewis bases, forming coordinate bonds with the central atom or ion; the latter acts as a Lewis acid. Atom ...
Bio II HName list2
... Chapter 3- Biological Molecules Organic compounds Hydrocarbons Functional groups Monomers Polymers Alcohols Enzymes Condensation reaction Hydrolysis Carbohydrate Sugar Monosaccharides Ribose Deoxyribose Glucose Oligosaccharide Sucrose Lactose Polysaccharide Cellulose Starch Glycogen Chitin Lipids Fa ...
... Chapter 3- Biological Molecules Organic compounds Hydrocarbons Functional groups Monomers Polymers Alcohols Enzymes Condensation reaction Hydrolysis Carbohydrate Sugar Monosaccharides Ribose Deoxyribose Glucose Oligosaccharide Sucrose Lactose Polysaccharide Cellulose Starch Glycogen Chitin Lipids Fa ...
PowerPoint Overview for Introduction
... Carbon (18%) is synonymous with life. Its central role is due to the fact that it has four bonding sites that allow for the building of long, complex chains of molecules. Moreover, carbon bonds can be formed and broken with a modest amount of energy, allowing for the dynamic organic chemistry that g ...
... Carbon (18%) is synonymous with life. Its central role is due to the fact that it has four bonding sites that allow for the building of long, complex chains of molecules. Moreover, carbon bonds can be formed and broken with a modest amount of energy, allowing for the dynamic organic chemistry that g ...
Synthesis and Crystal Structure of Nicotinamide Cobalt (II) Complexes
... the body, known as pellagra disease. Victims of pellagra show unusually high serum and urinary copper levels (1). The nicotinic acid derivative N,N-diethylnicotinamide (DENA) is an important respiratory stimulant (2). Transition metal complexes with biochemical molecules show interesting physical an ...
... the body, known as pellagra disease. Victims of pellagra show unusually high serum and urinary copper levels (1). The nicotinic acid derivative N,N-diethylnicotinamide (DENA) is an important respiratory stimulant (2). Transition metal complexes with biochemical molecules show interesting physical an ...
Chapter 14: Complex-Formation Titrations
... Electrogravimetric Determination of Copper and Lead in Brass experiment, ammonia was added to an analyte solution to test for the presence of copper (II), which in the presence of ammonia would produce the copper-ammonia complex, Cu(NH3)42+, a bright blue complex. The donor species, or ligand, must ...
... Electrogravimetric Determination of Copper and Lead in Brass experiment, ammonia was added to an analyte solution to test for the presence of copper (II), which in the presence of ammonia would produce the copper-ammonia complex, Cu(NH3)42+, a bright blue complex. The donor species, or ligand, must ...
Practice Problems for Chapter 4 1. The alkaline earth metals include
... Practice Problems for Chapter 4 ...
... Practice Problems for Chapter 4 ...
Electrontransfer proteins
... - Change in the oxidation state should not be accompanied by changes in the coordination geometry, bond length/bond angles. → the specific coordination geometry should be suitable for both oxidation states of the metal ion. Electron uptake and removal should not result in significant change in the s ...
... - Change in the oxidation state should not be accompanied by changes in the coordination geometry, bond length/bond angles. → the specific coordination geometry should be suitable for both oxidation states of the metal ion. Electron uptake and removal should not result in significant change in the s ...
Lecture 20.The d-Block Elements.VII
... Manganese(IV) oxide is the starting point for making most other manganese compounds. Potassium permanganate, KMnO4, is an important oxidizing agent that is used in both analytical and organic chemistry laboratories, and in water treatment. ...
... Manganese(IV) oxide is the starting point for making most other manganese compounds. Potassium permanganate, KMnO4, is an important oxidizing agent that is used in both analytical and organic chemistry laboratories, and in water treatment. ...
transition metals
... o Bonds in the reactants are weakened (allowing reaction to takes place) o Desorption of products from the surface of the catalyst Bonding to the surface of the catalyst must be strong enough to weaken bonds and allow reaction to take place but weak enough to allow desorption after the reaction The ...
... o Bonds in the reactants are weakened (allowing reaction to takes place) o Desorption of products from the surface of the catalyst Bonding to the surface of the catalyst must be strong enough to weaken bonds and allow reaction to take place but weak enough to allow desorption after the reaction The ...
What do you know about light?
... same! – The composition of a molecule of water. The chemical formula tells us that a water molecule s made up of 3 atoms; 2 hydrogen and 1 oxygen. ...
... same! – The composition of a molecule of water. The chemical formula tells us that a water molecule s made up of 3 atoms; 2 hydrogen and 1 oxygen. ...
Exam 1
... The general name for the type of reaction that occurs when two nucleotides are separated is hydrolysis. ...
... The general name for the type of reaction that occurs when two nucleotides are separated is hydrolysis. ...
called “organic molecules”
... linking amino acids together in a chain called a “polypeptide” •Each link is created by the dehydration reaction between the amino group of one amino acid and the carboxyl group of the next amino acid in the chain. ...
... linking amino acids together in a chain called a “polypeptide” •Each link is created by the dehydration reaction between the amino group of one amino acid and the carboxyl group of the next amino acid in the chain. ...
Organic Compounds
... Building of Structural parts of cell Non- Polar - Does not dissolve in water ...
... Building of Structural parts of cell Non- Polar - Does not dissolve in water ...
Biochemistry- Ch 11. Carbohydrates
... Formation of a Mannose 6phosphate Marker I-cell Disease: deficient in the phosphotransferase. ...
... Formation of a Mannose 6phosphate Marker I-cell Disease: deficient in the phosphotransferase. ...
Calculating & Naming Compounds
... type of element: metals, non metals, etc valence electrons: for lewis diagrams oxidation #: criss-cross method ...
... type of element: metals, non metals, etc valence electrons: for lewis diagrams oxidation #: criss-cross method ...
Welcome to AP Chemistry!
... Oxidation Number: A number assigned to an atom in a molecular compound or molecular ion that indicates the general distribution of electrons among the bonded atoms. 1. The oxidation number of any uncombined element is O. 2. The oxidation number of a monatomic ion equal the charge on the ion. 3. The ...
... Oxidation Number: A number assigned to an atom in a molecular compound or molecular ion that indicates the general distribution of electrons among the bonded atoms. 1. The oxidation number of any uncombined element is O. 2. The oxidation number of a monatomic ion equal the charge on the ion. 3. The ...
Macromolecules Worksheet
... lipids_ 6. Of what kind of organic compound are oils, waxes, and fats an example? nucleotides_ 7. These are the individual subunits that make up DNA and RNA. polypeptide or protein_ 8. What is a long chain of amino acids called? covalent_ 9. What type of bond involves the sharing of electrons? deoxy ...
... lipids_ 6. Of what kind of organic compound are oils, waxes, and fats an example? nucleotides_ 7. These are the individual subunits that make up DNA and RNA. polypeptide or protein_ 8. What is a long chain of amino acids called? covalent_ 9. What type of bond involves the sharing of electrons? deoxy ...
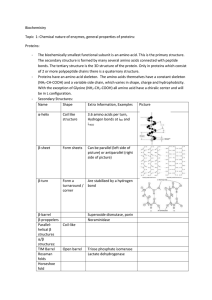
Biochemistry Topic 1: Chemical nature of enzymes, general
... Proteins can be separated according to their size, charge at a specific pH, MW, solubility or affinity to other substances Proteins have domains (parts with specific functions) The isoelectric point - pI – is the pH at which the net charge of the protein equals zero. At this point the Protein will l ...
... Proteins can be separated according to their size, charge at a specific pH, MW, solubility or affinity to other substances Proteins have domains (parts with specific functions) The isoelectric point - pI – is the pH at which the net charge of the protein equals zero. At this point the Protein will l ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.