Covalent Reactions Atoms SHARE electrons
... • Collagen- ligaments, tendons, skin • Many hormones • Actin and Myosin- allow muscles to contract • Hemoglobin- transport oxygen in blood • Antibodies in the blood • Allow movement through cell membrane • Enzymes (speed up chemical reactions) ...
... • Collagen- ligaments, tendons, skin • Many hormones • Actin and Myosin- allow muscles to contract • Hemoglobin- transport oxygen in blood • Antibodies in the blood • Allow movement through cell membrane • Enzymes (speed up chemical reactions) ...
BIOCHEMISTRY STUDY GUIDE Look over Chapter 3 Review on
... What is a functional group and what does it do to a compounds structure and function? How does the structure of phospholipids, linear molecules with a polar end and a nonpolar end, relate to their function in the cell membrane? Know what the monomers of each molecule of life are and examples of each ...
... What is a functional group and what does it do to a compounds structure and function? How does the structure of phospholipids, linear molecules with a polar end and a nonpolar end, relate to their function in the cell membrane? Know what the monomers of each molecule of life are and examples of each ...
Protein structure
... FIGURE 9.13. (A) Two zinc finger motifs in the glucocorticoid receptor. Each circle is one amino acid residue. Four cysteine residues, indicated c, bind each zinc ion. (B) Drawing of the zinc finger domains of a dimerized pair of glucocorticoid hormone receptors interacting with DNA. One zinc finge ...
... FIGURE 9.13. (A) Two zinc finger motifs in the glucocorticoid receptor. Each circle is one amino acid residue. Four cysteine residues, indicated c, bind each zinc ion. (B) Drawing of the zinc finger domains of a dimerized pair of glucocorticoid hormone receptors interacting with DNA. One zinc finge ...
Ion Exchange Chromatography
... • Desalting is carried out before ion exchange either by gel filtration chromatography , by dialysis or by centrifugal filtration. • Elution of bound proteins is achieved by reversing the process of binding and, again, exchanging a counterion for protein. • This is usually carried out by applying a ...
... • Desalting is carried out before ion exchange either by gel filtration chromatography , by dialysis or by centrifugal filtration. • Elution of bound proteins is achieved by reversing the process of binding and, again, exchanging a counterion for protein. • This is usually carried out by applying a ...
Metal-Requiring Enzymes (Molecular Biology)
... are listed in Table 1. Metal ions involved with enzymes that participate in electron transport undergo redox reactions. Thus, the ionic forms of iron, copper, cobalt, and molybdenum can be ...
... are listed in Table 1. Metal ions involved with enzymes that participate in electron transport undergo redox reactions. Thus, the ionic forms of iron, copper, cobalt, and molybdenum can be ...
Extension worksheet – Option C - Cambridge Resources for the IB
... An allosteric, non-competitive inhibitor may combine with an enzyme and cause the shape of the active site to change so that the substrate cannot bind to it. Such inhibitors, if they bind reversibly, can act in end-product inhibition of metabolic reactions. End-product inhibition is an example of ne ...
... An allosteric, non-competitive inhibitor may combine with an enzyme and cause the shape of the active site to change so that the substrate cannot bind to it. Such inhibitors, if they bind reversibly, can act in end-product inhibition of metabolic reactions. End-product inhibition is an example of ne ...
164 Study Guide chem
... Eight), peptide bond, pH (know the formula for this), phospholipids, polar and nonpolar covalent bonds, polysaccharide, proton, ribose, saturated and unsaturated fats, single and double covalent bonds, steroid, triglyceride, weak acid and weak base Be able to recognize (not draw) the general chemica ...
... Eight), peptide bond, pH (know the formula for this), phospholipids, polar and nonpolar covalent bonds, polysaccharide, proton, ribose, saturated and unsaturated fats, single and double covalent bonds, steroid, triglyceride, weak acid and weak base Be able to recognize (not draw) the general chemica ...
Synthesis of Alum Lab
... NH3 is added to Cu2+: Cu(NH3)42+ cupric tetraamine NH3 is added to Ag+: Ag(NH3)2+ silver diamine Conc. OH- is added to Zn(OH)2: Zn(OH)42+ Fe3+ in thiocyanate (SCN-): Fe(SCN)63- complex Co2+ with chlorine: CoCl42Al oxidized in base: Al(OH)4- ...
... NH3 is added to Cu2+: Cu(NH3)42+ cupric tetraamine NH3 is added to Ag+: Ag(NH3)2+ silver diamine Conc. OH- is added to Zn(OH)2: Zn(OH)42+ Fe3+ in thiocyanate (SCN-): Fe(SCN)63- complex Co2+ with chlorine: CoCl42Al oxidized in base: Al(OH)4- ...
Transition metals and complex ions
... Complex ions - Definations • LIGAND – molecule or ion that can donate a pair of electrons to form a coordinate or dative covalent bond • COORDINATE BOND – a bond where both electrons are provided for by the same atom. It is the same as a dative covalent bond but involves transition metals instead. ...
... Complex ions - Definations • LIGAND – molecule or ion that can donate a pair of electrons to form a coordinate or dative covalent bond • COORDINATE BOND – a bond where both electrons are provided for by the same atom. It is the same as a dative covalent bond but involves transition metals instead. ...
How to Assign Oxidation Numbers
... bonded to fluorine (where it may be +1 or +2) and in peroxides where it has an oxidation state of –1 • The sum of the oxidation states of all the atoms in a molecule or ion is equal to the overall charge on the species. ...
... bonded to fluorine (where it may be +1 or +2) and in peroxides where it has an oxidation state of –1 • The sum of the oxidation states of all the atoms in a molecule or ion is equal to the overall charge on the species. ...
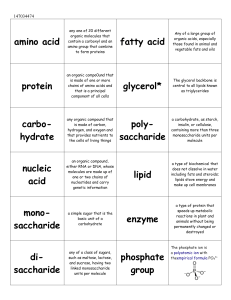
Organic Chemistry and Macromolecules
... made of many bonded glucose units; typically found in cell walls of plants and provides nutritional roughage. • Hemoglobin—a type of protein that is in red blood cells; contains iron and carries oxygen from lungs to body cells. • Biurets Solution—a blue reagent that turns purple in the presence of p ...
... made of many bonded glucose units; typically found in cell walls of plants and provides nutritional roughage. • Hemoglobin—a type of protein that is in red blood cells; contains iron and carries oxygen from lungs to body cells. • Biurets Solution—a blue reagent that turns purple in the presence of p ...
Chapter 7 Acids, bases and ions in aqueous solution
... Common ion effect – If a salt MX is added to an aqueous solution containing the solute MY (the ion Mn+ is common to both salts), the presence of the dissolved Mn+ ions suppresses the dissolution of MX compared with that in pure water. ...
... Common ion effect – If a salt MX is added to an aqueous solution containing the solute MY (the ion Mn+ is common to both salts), the presence of the dissolved Mn+ ions suppresses the dissolution of MX compared with that in pure water. ...
Unit 5 Powerpoint
... Ions of Transition Metals Many form more than one cation with different ionic charges. The charges of the cations of many transition metal ions must be determined by the number of electrons lost. Ex. Fe2+ and Fe3+ ...
... Ions of Transition Metals Many form more than one cation with different ionic charges. The charges of the cations of many transition metal ions must be determined by the number of electrons lost. Ex. Fe2+ and Fe3+ ...
Chapter 2-1 The Nature of Matter
... the structure of malonic acid allows it to bind to the same site on the enzyme (b). But there is no oxidation so no speedy release of products. The inhibition is called competitive because if you increase the ratio of succinic to malonic acid in the mixture, you will gradually restore the rate of ca ...
... the structure of malonic acid allows it to bind to the same site on the enzyme (b). But there is no oxidation so no speedy release of products. The inhibition is called competitive because if you increase the ratio of succinic to malonic acid in the mixture, you will gradually restore the rate of ca ...
Exam 1 - Chemistry Courses: About
... A. ____________ Aminos acids with the appreviations L, I, V, and W are all considered hydrophobic amino acids. B. ____________ Breaking a disulfide bond into two cysteine residues is a hydrolysis reaction. C. ____________ The alpha helix is a right-handed structure which is highly polar. D. ________ ...
... A. ____________ Aminos acids with the appreviations L, I, V, and W are all considered hydrophobic amino acids. B. ____________ Breaking a disulfide bond into two cysteine residues is a hydrolysis reaction. C. ____________ The alpha helix is a right-handed structure which is highly polar. D. ________ ...
BondingBasics11
... 3. You will have to decide how many of each ion you need to make a STABLE IONIC BOND. 4. Record all bonds on your Date Book (data ...
... 3. You will have to decide how many of each ion you need to make a STABLE IONIC BOND. 4. Record all bonds on your Date Book (data ...
Name: Date: AP Chemistry/Chemistry 145 Summer Assignment
... 19. A 11.6-g sample of iron ore, containing Fe3O4 (232 g/mol), is reacted with carbon to form purified iron. The other product of the reaction is carbon dioxide gas. 2.10 g of iron is ...
... 19. A 11.6-g sample of iron ore, containing Fe3O4 (232 g/mol), is reacted with carbon to form purified iron. The other product of the reaction is carbon dioxide gas. 2.10 g of iron is ...
Ecology
... does not dissolve in water including fats and steroids; lipids stove energy and make up cell membranes ...
... does not dissolve in water including fats and steroids; lipids stove energy and make up cell membranes ...
1 | Page Chemistry Lecture #35 Chemistry Lecture #35: Names and
... elements often require the use of Roman numerals since these metals have variable oxidation states. For example, CuO and Cu2O could both be called copper oxide. But the oxidation state of Cu in CuO is +2, and the oxidation state of Cu in Cu2O is +1. ...
... elements often require the use of Roman numerals since these metals have variable oxidation states. For example, CuO and Cu2O could both be called copper oxide. But the oxidation state of Cu in CuO is +2, and the oxidation state of Cu in Cu2O is +1. ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.