Metal Ion Transport and Storage
... • Charged Ions must pass through a Hydrophobic Membrane – Neutral gases (O2, CO2) and low charge density ions (anions) can move directly through the membrane – High charge density cations require help • Once inside the cell, metal ions must be transported to the location of their use, then released ...
... • Charged Ions must pass through a Hydrophobic Membrane – Neutral gases (O2, CO2) and low charge density ions (anions) can move directly through the membrane – High charge density cations require help • Once inside the cell, metal ions must be transported to the location of their use, then released ...
Biomolecules - Biology with Mr. Flores
... and are essential to living organisms. Proteins are composed of the following elements, C – Carbon, H – Hydrogen, O – Oxygen, and N – Nitrogen The monomers of proteins are amino acids, there are 20 different amino acids. ...
... and are essential to living organisms. Proteins are composed of the following elements, C – Carbon, H – Hydrogen, O – Oxygen, and N – Nitrogen The monomers of proteins are amino acids, there are 20 different amino acids. ...
2.1 KEY CONCEPT All living things are based on atoms and their
... Living things consist of atoms of different elements. • An atom is the smallest basic unit of matter. • An element is one type of atom. Hydrogen atom (H) ...
... Living things consist of atoms of different elements. • An atom is the smallest basic unit of matter. • An element is one type of atom. Hydrogen atom (H) ...
Biochemistry Test Review
... Explain pH and illustrate the pH scale. What do acids form in water?/ What do bases form in water? Draw an illustration of diffusion. Describe dynamic equilibrium. What is Brownian motion? What is the difference between Osmosis and Diffusion? Compare a compound and a molecule. Draw the structure of ...
... Explain pH and illustrate the pH scale. What do acids form in water?/ What do bases form in water? Draw an illustration of diffusion. Describe dynamic equilibrium. What is Brownian motion? What is the difference between Osmosis and Diffusion? Compare a compound and a molecule. Draw the structure of ...
Quizon ch5-6-7-8new.doc
... b. facilitated diffusion c. active transport d. endocytosis e. Both the c and d are correct. 2. Proteins which act as catalysts of chemical reactions [in cells] are called: a. enzymes. b. coenzymes. c. reaction cofactors. d. substrates. e. reactants 3. A final product of a metabolic pathway inhibits ...
... b. facilitated diffusion c. active transport d. endocytosis e. Both the c and d are correct. 2. Proteins which act as catalysts of chemical reactions [in cells] are called: a. enzymes. b. coenzymes. c. reaction cofactors. d. substrates. e. reactants 3. A final product of a metabolic pathway inhibits ...
Abstract Coordination Chemistry of Tetra(pyrazolyl)
... ligands that occupy one axial (A) and four equatorial (E) positions about a given transition metal center considering that systems capable of mediating spectacular organic transformations such as alkane oxidation have been identified. The discovery of new systems will improve understanding of C-H bo ...
... ligands that occupy one axial (A) and four equatorial (E) positions about a given transition metal center considering that systems capable of mediating spectacular organic transformations such as alkane oxidation have been identified. The discovery of new systems will improve understanding of C-H bo ...
Chapter 3 (part 2) – Protein Function
... • Enzymes and bound ligand go through a number of intermediate forms of different geometry. They are all called transition states. • The energy that it takes to get to the most unstable transition state is called the activation energy. • Enzymes speed reactions by selectively stabilizing the transi ...
... • Enzymes and bound ligand go through a number of intermediate forms of different geometry. They are all called transition states. • The energy that it takes to get to the most unstable transition state is called the activation energy. • Enzymes speed reactions by selectively stabilizing the transi ...
Give reasons for the following: (i) Bond enthalpy of F2
... of F-F are very large. Thus, the bond dissociation energy of F2 is lower than that of Cl2. (ii) PH3 has lower boiling point than NH3 because NH3 molecule possess ...
... of F-F are very large. Thus, the bond dissociation energy of F2 is lower than that of Cl2. (ii) PH3 has lower boiling point than NH3 because NH3 molecule possess ...
name - Fulton County Schools
... shows two atoms of the same nonmetal (usually in gas form) that have bonded together • Reactive nonmetals will bond with one another if no other substance is available for bonding ...
... shows two atoms of the same nonmetal (usually in gas form) that have bonded together • Reactive nonmetals will bond with one another if no other substance is available for bonding ...
Key: Biomolecule Study Guide 1) In animals, excess carbohydrates
... They must fit the molecules that they interact with (Lock and Key) 10) What does it mean to say an enzyme is “denatured”? It has changed its 3-D shape 11) What are 2 ways to denature an enzyme? Change the temperature or pH ...
... They must fit the molecules that they interact with (Lock and Key) 10) What does it mean to say an enzyme is “denatured”? It has changed its 3-D shape 11) What are 2 ways to denature an enzyme? Change the temperature or pH ...
Übung: Monte Carlo, Molecular Dynamics
... 7. I have built a Bolztmann / knowledge-based score function for proteins using the methodology based on potentials of mean force. It is based on Cα-Cα distances. I do not distinguish between amino acids which are separated by one residue (i,i+2) and those separated by many residues. Why will this b ...
... 7. I have built a Bolztmann / knowledge-based score function for proteins using the methodology based on potentials of mean force. It is based on Cα-Cα distances. I do not distinguish between amino acids which are separated by one residue (i,i+2) and those separated by many residues. Why will this b ...
Macromolecules Worksheet
... ____________________ 5. This is the name given to an amino acid added to a dipeptide. ____________________ 6. Of what kind of organic compound are oils, waxes, and fats an example? ____________________ 7. These are the individual subunits that make up DNA and RNA. ____________________ 8. What is a l ...
... ____________________ 5. This is the name given to an amino acid added to a dipeptide. ____________________ 6. Of what kind of organic compound are oils, waxes, and fats an example? ____________________ 7. These are the individual subunits that make up DNA and RNA. ____________________ 8. What is a l ...
Print › Biochemistry | Quizlet
... lowering the activation energy needed to start the reaction hydrogen bond: weak electrostatic bond formed by the attraction of opposite charges between a hydrogen atom and an oxygen , fluorine, or nitrogen atom ion: atom that is negatively or positively charged because it has lost or gained one or m ...
... lowering the activation energy needed to start the reaction hydrogen bond: weak electrostatic bond formed by the attraction of opposite charges between a hydrogen atom and an oxygen , fluorine, or nitrogen atom ion: atom that is negatively or positively charged because it has lost or gained one or m ...
Honors Chemistry II Review 1. Express the following in scientific
... Which reactant is limiting if there are 80kg of water to be removed and 65kg of Li2O available? How many kg of the excess reactant remain? 20. After lithium hydroxide is produced aboard the space shuttle, it is used to remove exhaled carbon dioxide from the air supply according to the following equa ...
... Which reactant is limiting if there are 80kg of water to be removed and 65kg of Li2O available? How many kg of the excess reactant remain? 20. After lithium hydroxide is produced aboard the space shuttle, it is used to remove exhaled carbon dioxide from the air supply according to the following equa ...
Macromolecules Notes
... ____________________ 5. This is the name given to an amino acid added to a dipeptide. ____________________ 6. Of what kind of organic compound are oils, waxes, and fats an example? ____________________ 7. These are the individual subunits that make up DNA and RNA. ____________________ 8. What is a l ...
... ____________________ 5. This is the name given to an amino acid added to a dipeptide. ____________________ 6. Of what kind of organic compound are oils, waxes, and fats an example? ____________________ 7. These are the individual subunits that make up DNA and RNA. ____________________ 8. What is a l ...
4. Transition Metals - Cathkin High School
... Transition metals exhibit variable oxidation states because they can not only lose their 4s electrons but some or all of their 3d electrons. ...
... Transition metals exhibit variable oxidation states because they can not only lose their 4s electrons but some or all of their 3d electrons. ...
Experiment 11: Acidity and Basicity
... The carbon dioxide in our breath reacts with the NaOH solution, which results to a change in its color. Iron is more reactive than copper. The presence of iodine was confirmed by the addition of starch. ...
... The carbon dioxide in our breath reacts with the NaOH solution, which results to a change in its color. Iron is more reactive than copper. The presence of iodine was confirmed by the addition of starch. ...
Chapter 4: The Chemical Basis of Life
... o Forms by the attraction of the oily parts of lipid molecules for each other and by the attraction of the other parts of the lipid molecules for the surrounding water ...
... o Forms by the attraction of the oily parts of lipid molecules for each other and by the attraction of the other parts of the lipid molecules for the surrounding water ...
EXAM 1-A fall 2004.doc
... b. oxygen, carbon, helium, nitrogen e. nitrogen, carbon, helium, sulfur c. oxygen, nitrogen, carbon, sulfur ___ ...
... b. oxygen, carbon, helium, nitrogen e. nitrogen, carbon, helium, sulfur c. oxygen, nitrogen, carbon, sulfur ___ ...
Midterm Review Project Ch 5
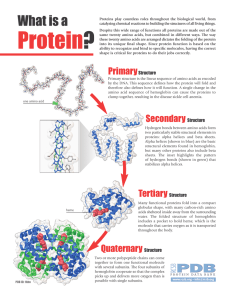
... amino acid: amino group, carboxyl group, an alpha carbon and an R group, the variable group that differentiates amino acids (the group’s properties, whether it is polar or not, determine if amino acid is hydrophilic or not) primary structure: amino acid sequence; secondary structure: coils and folds ...
... amino acid: amino group, carboxyl group, an alpha carbon and an R group, the variable group that differentiates amino acids (the group’s properties, whether it is polar or not, determine if amino acid is hydrophilic or not) primary structure: amino acid sequence; secondary structure: coils and folds ...
an introduction to bioinorganic chemistry
... involving several critical biochemical pathways. Several of these enzymes are noted here. Superoxide dismutase, which metabolize the potentially damaging superoxide anion. Lysyl oxidase is a monoamine oxidase required for cross-linking collagen and elastin, the structural macromolecules of connectiv ...
... involving several critical biochemical pathways. Several of these enzymes are noted here. Superoxide dismutase, which metabolize the potentially damaging superoxide anion. Lysyl oxidase is a monoamine oxidase required for cross-linking collagen and elastin, the structural macromolecules of connectiv ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.