Chapter 1 Structure and Bonding
... The rates of reaction depend on the ability of the electron to “tunnel” through the ligands from one metal to the other i. Tunneling = moving through an energy barrier (the ligands) that is normally too high to allow the electron to pass through. This is a quantum mechanical process having to do wit ...
... The rates of reaction depend on the ability of the electron to “tunnel” through the ligands from one metal to the other i. Tunneling = moving through an energy barrier (the ligands) that is normally too high to allow the electron to pass through. This is a quantum mechanical process having to do wit ...
ELECTRON TRANSFER PATHWAYS IN BLUE COPPER
... Cr(II)/(III) couple: While Cr(II) ions are exceptionally strong reductants and exchange their ligands very fast, the Cr(III) ion exchanges its ligands rather slowly [3]. Thus, Cr(II) can coordinate to one or more amino acid residues of the protein while transferring to its active center an electron. ...
... Cr(II)/(III) couple: While Cr(II) ions are exceptionally strong reductants and exchange their ligands very fast, the Cr(III) ion exchanges its ligands rather slowly [3]. Thus, Cr(II) can coordinate to one or more amino acid residues of the protein while transferring to its active center an electron. ...
Metals in Redox Biology - University of Nebraska–Lincoln
... – High accumulation of mitochondrial iron in cells lacking Yfh1 no Sod2 inactivation – Iron is insoluble Fe (III) ...
... – High accumulation of mitochondrial iron in cells lacking Yfh1 no Sod2 inactivation – Iron is insoluble Fe (III) ...
H H H H H N HO O NC[ ]- - Teacher`s Tools® Chemistry
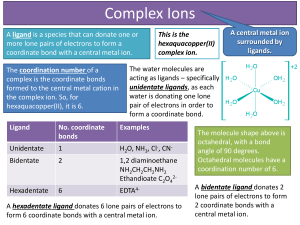
... Transition metals can behave like Lewis Acids (electron pair acceptors) in the formation of what are called complex ions. A coordination compound typically consists of a complex ion and counter ion. The molecules or ions that surround the metal in a complex ion are called ligands. The ligands act li ...
... Transition metals can behave like Lewis Acids (electron pair acceptors) in the formation of what are called complex ions. A coordination compound typically consists of a complex ion and counter ion. The molecules or ions that surround the metal in a complex ion are called ligands. The ligands act li ...
Snímek 1 - physics.muni.cz
... unreactive substrates. Therefore, it uses the negatively charged cysteine ligand which donates electrons to Fe and stabilizes the high oxidation state. One of strategies that proteins employ to modify the redox potential is using different proximal ligands. ...
... unreactive substrates. Therefore, it uses the negatively charged cysteine ligand which donates electrons to Fe and stabilizes the high oxidation state. One of strategies that proteins employ to modify the redox potential is using different proximal ligands. ...
Oxidation Number Rules
... Examples: HCl, hydrogen is +1; NaH, hydrogen is -1. d. The halogens, unless bonded to an element with a higher electronegativity, have an oxidation number of -1. Examples: NaCl, chlorine is -1; HClO4, chlorine is +7. 4. Since chemical compounds are neutral, it stands to reason that the algebraic sum ...
... Examples: HCl, hydrogen is +1; NaH, hydrogen is -1. d. The halogens, unless bonded to an element with a higher electronegativity, have an oxidation number of -1. Examples: NaCl, chlorine is -1; HClO4, chlorine is +7. 4. Since chemical compounds are neutral, it stands to reason that the algebraic sum ...
SUPPLEMENTARY INFORMATION FOR: The structure of a D
... S2A), with the residues coordinating the metal ion and forming the active cleft almost completely conserved between the two proteins (Fig. S2D). The main residues interacting with the sugar ligand in z5688 including the glutamic acid residues E110 and E186, lysines K90 and 108 and arginine R205, are ...
... S2A), with the residues coordinating the metal ion and forming the active cleft almost completely conserved between the two proteins (Fig. S2D). The main residues interacting with the sugar ligand in z5688 including the glutamic acid residues E110 and E186, lysines K90 and 108 and arginine R205, are ...
Chapter 12 (Complexometric Titration)
... • the ability of multidentate ligands to form more stable metal complexes than those formed by similar monodentate ligands • results from the formation of 5membered "ring" with metal and two atoms on the ligand ...
... • the ability of multidentate ligands to form more stable metal complexes than those formed by similar monodentate ligands • results from the formation of 5membered "ring" with metal and two atoms on the ligand ...
transition metals
... 6.) Using the ideas of Lewis and resonance structures, determine which of the following ligands can participate in linkage isomerism (meaning can they cause a compound to be a linkage isomer). DRAW the resonance structures in order to explain! Remember that a linkage isomer can bond to the metal ion ...
... 6.) Using the ideas of Lewis and resonance structures, determine which of the following ligands can participate in linkage isomerism (meaning can they cause a compound to be a linkage isomer). DRAW the resonance structures in order to explain! Remember that a linkage isomer can bond to the metal ion ...
Metal Regulation and Signalling - Zn Proteins
... And limit possible number of conformations, make the correct structure. ...
... And limit possible number of conformations, make the correct structure. ...
Problem 2
... Problem 10. We can use either Rasmol or SwissPDB to examine the structure. I used Swiss PDB first, displaying the heme in spacefilling model, and then using the mode that allows me to pick an atom and display all residues with XX distance of it. This is the ninth button from the left, with an eye, ...
... Problem 10. We can use either Rasmol or SwissPDB to examine the structure. I used Swiss PDB first, displaying the heme in spacefilling model, and then using the mode that allows me to pick an atom and display all residues with XX distance of it. This is the ninth button from the left, with an eye, ...
Chemistry 2000 B Spring 2005 Answers to the Second Problem Set
... (a) The coordination number of iron The coordination number is six (from the six oxygens in water) (b) The oxidation state of iron Fe(II) (c) The number of unpaired electrons, assuming high spin This is not a free ion, but an octahedral aqua complex, so we must consider the splitting of the d orbita ...
... (a) The coordination number of iron The coordination number is six (from the six oxygens in water) (b) The oxidation state of iron Fe(II) (c) The number of unpaired electrons, assuming high spin This is not a free ion, but an octahedral aqua complex, so we must consider the splitting of the d orbita ...
HL Answers to First row d-block elements questions
... 4. In [Fe(H2O)6]3+ the 3d sub-level is split into two by the water ligands. A d electron can absorb energy as it is promoted from the lower to the higher split level. The colour of the light transmitted is the complementary colour to the light absorbed. In [Fe(CN)6]3- the cyanide ligands cause the a ...
... 4. In [Fe(H2O)6]3+ the 3d sub-level is split into two by the water ligands. A d electron can absorb energy as it is promoted from the lower to the higher split level. The colour of the light transmitted is the complementary colour to the light absorbed. In [Fe(CN)6]3- the cyanide ligands cause the a ...
Mahua Ghosh - SN Bose National Centre for Basic Sciences
... basic histidine (H) 32 of A2 helix and the hydrophobic residues, namely, isoleucine (I) 59, W60 and I95 of the ...
... basic histidine (H) 32 of A2 helix and the hydrophobic residues, namely, isoleucine (I) 59, W60 and I95 of the ...
C h e m g u i d e ... CARBOXYLIC ACIDS: REDUCTION
... 1. Carboxylic acids can be reduced using lithium tetrahydridoaluminate, LiAlH4, which contains the [AlH4]- ion. a) Carboxylic acids are reduced to alcohols in this way. What kind of alcohols? b) Describe the bonding between the aluminium and the four hydrogens in the [AlH4]- ion. c) Writing the redu ...
... 1. Carboxylic acids can be reduced using lithium tetrahydridoaluminate, LiAlH4, which contains the [AlH4]- ion. a) Carboxylic acids are reduced to alcohols in this way. What kind of alcohols? b) Describe the bonding between the aluminium and the four hydrogens in the [AlH4]- ion. c) Writing the redu ...
problem set: atomic structure
... Write clear, concise answers for the following questions. Write no more than two sentences for any question. Include electron configuration, orbital filling diagrams, and mathematical formulas where they will support your answers. 1)Explain each of the following observations about ionization energie ...
... Write clear, concise answers for the following questions. Write no more than two sentences for any question. Include electron configuration, orbital filling diagrams, and mathematical formulas where they will support your answers. 1)Explain each of the following observations about ionization energie ...
Word Doc - Biochemistry
... number of residues is necessary to perform a particular biochemical function, and around 40-50 residues appears to be the lower limit for a functional domain size. Protein sizes range from this lower limit to several hundred residues in multi-functional proteins. Very large aggregates can be formed ...
... number of residues is necessary to perform a particular biochemical function, and around 40-50 residues appears to be the lower limit for a functional domain size. Protein sizes range from this lower limit to several hundred residues in multi-functional proteins. Very large aggregates can be formed ...
Ch 12 Electrolysis in water
... 2 Cl- Cl2+ 2 e Water may be oxidized to oxygen 2 H2O O2 + 4 H+ + 4 e Chlorine, bromine and iodine will oxidize before oxygen. That is it. ...
... 2 Cl- Cl2+ 2 e Water may be oxidized to oxygen 2 H2O O2 + 4 H+ + 4 e Chlorine, bromine and iodine will oxidize before oxygen. That is it. ...
File
... Carbon monoxide, CO, bonds to haemoglobin with a higher affinity than oxygen. If a person inhales CO, a ligand exchange reaction occurs and the CO prevents the transport of O2. This leads to drowsiness, lack of consciousness and can be fatal. ...
... Carbon monoxide, CO, bonds to haemoglobin with a higher affinity than oxygen. If a person inhales CO, a ligand exchange reaction occurs and the CO prevents the transport of O2. This leads to drowsiness, lack of consciousness and can be fatal. ...
Concepts in Transition Metal Chemistry – Questions
... Which of the following combinations of σ-bonding orbitals will form a bonding combination with a 4s orbital on a metal ion? Assume the 4s orbital has a positive sign and the metal ion is at the centre of the combination. ...
... Which of the following combinations of σ-bonding orbitals will form a bonding combination with a 4s orbital on a metal ion? Assume the 4s orbital has a positive sign and the metal ion is at the centre of the combination. ...
Simple Synthesis and Decomposition Reactions Worksheet
... Simple Synthesis and Decomposition Reactions Worksheet ...
... Simple Synthesis and Decomposition Reactions Worksheet ...
Oxidation-Reduction (Redox) Reactions
... In a compound or as an ion, alkali metals will always be +1. In a compound or an ion, alkaline metals will always be +2. (The other atoms in the compound have to adjust their oxidation states to give the correct overall charge. e.g. NaH) O is –2 (unless two Os bonded to each other) (Everything else ...
... In a compound or as an ion, alkali metals will always be +1. In a compound or an ion, alkaline metals will always be +2. (The other atoms in the compound have to adjust their oxidation states to give the correct overall charge. e.g. NaH) O is –2 (unless two Os bonded to each other) (Everything else ...
Metalloprotein

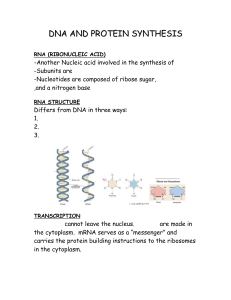
Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.


![H H H H H N HO O NC[ ]- - Teacher`s Tools® Chemistry](http://s1.studyres.com/store/data/017018154_1-68467b392d8cadb27de319df72045839-300x300.png)