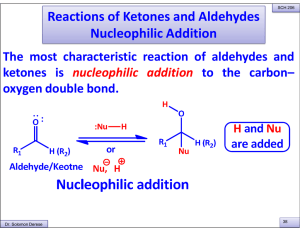
Reactions of Ketones and Aldehydes Nucleophilic Addition
... I. Addition of the organometalics to formaldehyde (CH2=O) forms a 1° alcohol. II. Addition of the organometalics to all other aldehydes forms a 2° alcohol. III.Addition of the organometalics to ketones forms a 3° alcohol. Dr. Solomon Derese ...
... I. Addition of the organometalics to formaldehyde (CH2=O) forms a 1° alcohol. II. Addition of the organometalics to all other aldehydes forms a 2° alcohol. III.Addition of the organometalics to ketones forms a 3° alcohol. Dr. Solomon Derese ...
研 究 業 績 リ ス ト
... H. Matsuzaka, J.-P. Qü, T. Ogino, M. Nishio, Y. Nishibayashi, Y. Ishii, S. Uemura, M. Hidai J. Chem. Soc., Dalton Trans., 4307-4312 (1996). (26) Palladium-catalyzed Homocoupling Reactions of Organic Tellurides Y. Nishibayashi, C. S. Cho, K. Ohe, S. Uemura J. Organomet. Chem., 526, 335-339 (1996). (2 ...
... H. Matsuzaka, J.-P. Qü, T. Ogino, M. Nishio, Y. Nishibayashi, Y. Ishii, S. Uemura, M. Hidai J. Chem. Soc., Dalton Trans., 4307-4312 (1996). (26) Palladium-catalyzed Homocoupling Reactions of Organic Tellurides Y. Nishibayashi, C. S. Cho, K. Ohe, S. Uemura J. Organomet. Chem., 526, 335-339 (1996). (2 ...
Glycosyl amines
... Derivatives of sugars in which the glycosyl moiety is linked to a primary, secondary or a tertiary amino group. If two glycosyl moieties are linked to a secondary amino group, the derivatives are named as bisglycosyl amines. ...
... Derivatives of sugars in which the glycosyl moiety is linked to a primary, secondary or a tertiary amino group. If two glycosyl moieties are linked to a secondary amino group, the derivatives are named as bisglycosyl amines. ...
Chapter 4
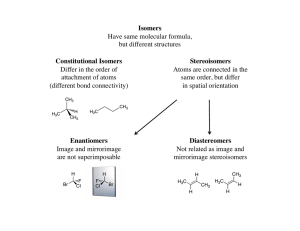
... Therefore if a given solution has 90% of one enantiomer (say R) and 10% of the other enantiomer (S) then the enantiomeric excess is 80% ...
... Therefore if a given solution has 90% of one enantiomer (say R) and 10% of the other enantiomer (S) then the enantiomeric excess is 80% ...
Factors influencing ring closure through olefin metathesis – A
... available, cyclization is the most straightforward way of obtaining rings. Commonly employed cyclization methods involve reactions of cationic, anionic or radical species. Common rings such as 5–7 membered ones are easily available by these methods. However, formation of medium or large rings by the ...
... available, cyclization is the most straightforward way of obtaining rings. Commonly employed cyclization methods involve reactions of cationic, anionic or radical species. Common rings such as 5–7 membered ones are easily available by these methods. However, formation of medium or large rings by the ...
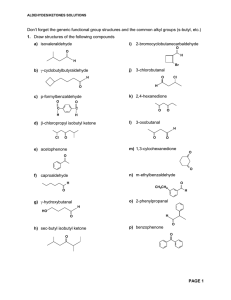
Aldehydes/Ketones Solutions
... mechanisms required but show all reagents and intermediate products formed. More than one step may be necessary a) O O C H ...
... mechanisms required but show all reagents and intermediate products formed. More than one step may be necessary a) O O C H ...
NAME Chem 204 Spring, 1990 Final Exam FINAL EXAM: Chaps. 1
... Supplies: Common Solvents of all kinds: non-polar; hydroxylic; dipolar, aprotic Inorganic Reagents, including SOCl2, CO2, Mg, Na, K, Li, Al, NaN3, PX3, CrO3, ZnCl2, D2O, AgBr, AgNO3, RCN, NaSH, NaOH, NH3, H2SO4, HX (Cl, Br, I), NaSCN, NaH, KSCN, KCN, NaSH, NaOH, NH3, H2SO4, HX(Cl,Br,or I), NH2OSO3H, ...
... Supplies: Common Solvents of all kinds: non-polar; hydroxylic; dipolar, aprotic Inorganic Reagents, including SOCl2, CO2, Mg, Na, K, Li, Al, NaN3, PX3, CrO3, ZnCl2, D2O, AgBr, AgNO3, RCN, NaSH, NaOH, NH3, H2SO4, HX (Cl, Br, I), NaSCN, NaH, KSCN, KCN, NaSH, NaOH, NH3, H2SO4, HX(Cl,Br,or I), NH2OSO3H, ...
Reactions
... Alcohols can be oxidized to aldehydes or ketones using a modified form of CrO3 called PCC for pyridinium chlorochromate, (C5H6NCrO3Cl). It is a milder reagent and if you use it carefully you can stop the reaction at the intermediate ...
... Alcohols can be oxidized to aldehydes or ketones using a modified form of CrO3 called PCC for pyridinium chlorochromate, (C5H6NCrO3Cl). It is a milder reagent and if you use it carefully you can stop the reaction at the intermediate ...
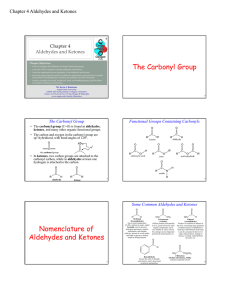
The Carbonyl Group - Angelo State University
... • Draw structural formulas for the following molecules: – 4-chloro-2-phenylpentanal ...
... • Draw structural formulas for the following molecules: – 4-chloro-2-phenylpentanal ...
The Carbonyl Group Nomenclature of Aldehydes and Ketones
... • In molecules which have both OH and C=O groups on different carbon atoms, an intramolecular addition reaction can occur, producing a cyclic ...
... • In molecules which have both OH and C=O groups on different carbon atoms, an intramolecular addition reaction can occur, producing a cyclic ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... CC bonds (from 37.0% to 52.0%). Notably, these changes include loss of CO and C−O functions from the GO surface (epoxide, hydroxyl) during the reaction, as well as the reduction of anhydride and carboxylic acid functional groups on the GO surface (from 13.6% to 7.2%). (7) Film conductivity measure ...
... CC bonds (from 37.0% to 52.0%). Notably, these changes include loss of CO and C−O functions from the GO surface (epoxide, hydroxyl) during the reaction, as well as the reduction of anhydride and carboxylic acid functional groups on the GO surface (from 13.6% to 7.2%). (7) Film conductivity measure ...
Applied Catalysis A: General, 374 (1-2) 201
... [27–30] catalysts in combination with trialkylphosphine ligands have been shown to produce alcohols; however, cobalt-based catalysts exhibit low alcohol selectivity because they promote the hydrogenation of the alkene to the corresponding alkane. Studies by Cole-Hamilton and coworkers have demonstra ...
... [27–30] catalysts in combination with trialkylphosphine ligands have been shown to produce alcohols; however, cobalt-based catalysts exhibit low alcohol selectivity because they promote the hydrogenation of the alkene to the corresponding alkane. Studies by Cole-Hamilton and coworkers have demonstra ...
幻灯片 1
... would be given the formula RO. But Williamson, by his ether synthesis, showed that mixed ethers, with two different alkyl groups, could be prepared. Ethers thus has to have the water-type formula ROR', and oxygen had the equivalent weight of 8 but the atomic weight of 16. By this type of argument he ...
... would be given the formula RO. But Williamson, by his ether synthesis, showed that mixed ethers, with two different alkyl groups, could be prepared. Ethers thus has to have the water-type formula ROR', and oxygen had the equivalent weight of 8 but the atomic weight of 16. By this type of argument he ...
Ion Exchange Resins: Catalyst Recovery and Recycle
... rhodium complexes involved in the catalytic cycle are anionic suggested this system to be an excellent candidate for noncovalent, ionic immobilization,107 and, indeed, this was one of the earliest applications of molecular catalysts supported on ion exchange resins.108,109 Most importantly, Chiyoda/ ...
... rhodium complexes involved in the catalytic cycle are anionic suggested this system to be an excellent candidate for noncovalent, ionic immobilization,107 and, indeed, this was one of the earliest applications of molecular catalysts supported on ion exchange resins.108,109 Most importantly, Chiyoda/ ...
Organic Chemistry Fifth Edition
... Nitrosation of N-Alkylarylamines Similar to secondary alkylamines; Gives N-nitroso amines ...
... Nitrosation of N-Alkylarylamines Similar to secondary alkylamines; Gives N-nitroso amines ...
Modern Synthetic Methods for Copper-Mediated C(aryl
... detail the remarkable simplicity of the new reaction conditions. A mixture of the phenol (1 equiv), aryl boronic acid (2–3 equiv), anhydrous Cu(OAc)2 (1–2 equiv), and Et3N (2– 3 equiv) in dichloromethane were stirred at room temperature for 1–2 days and then the product was isolated in good yield af ...
... detail the remarkable simplicity of the new reaction conditions. A mixture of the phenol (1 equiv), aryl boronic acid (2–3 equiv), anhydrous Cu(OAc)2 (1–2 equiv), and Et3N (2– 3 equiv) in dichloromethane were stirred at room temperature for 1–2 days and then the product was isolated in good yield af ...
CHEM 203 Material
... Why "formal" charge? Rigorously speaking, the + 1 charge present in NH4 is delocalized all over the molecule, i.e., each atom (N and 4 H's in this case) bears a share thereof. For simplicity, however, it is convenient to think of it as if it were localized on the N atom. That's why one calls it a fo ...
... Why "formal" charge? Rigorously speaking, the + 1 charge present in NH4 is delocalized all over the molecule, i.e., each atom (N and 4 H's in this case) bears a share thereof. For simplicity, however, it is convenient to think of it as if it were localized on the N atom. That's why one calls it a fo ...
HIGHLY SELECTIVE RHODIUM–CATALYZED C–H BORYLATIONS IN
... synthesis of new carbon–carbon bonds has increased the demand for efficient routes to organoboron starting materials. C–H borylation (activation) has provided an interesting approach to alleviate the requirement for prefunctionalized molecules such as aryl halides to obtain these desired organoboron ...
... synthesis of new carbon–carbon bonds has increased the demand for efficient routes to organoboron starting materials. C–H borylation (activation) has provided an interesting approach to alleviate the requirement for prefunctionalized molecules such as aryl halides to obtain these desired organoboron ...
Iodomethylzinc_iodid.. - Groupe Charette
... generating iodomethylzinc reagents, which can be categorized into three general classes: type 1, the oxidative addition of a dihalomethane to zinc metal, as typified by the original Simmons– Smith procedure;7,8 type 2, the reaction of a zinc(II) salt with a diazoalkane, first reported by Wittig and ...
... generating iodomethylzinc reagents, which can be categorized into three general classes: type 1, the oxidative addition of a dihalomethane to zinc metal, as typified by the original Simmons– Smith procedure;7,8 type 2, the reaction of a zinc(II) salt with a diazoalkane, first reported by Wittig and ...
Reactions You Should Know When You Begin Organic II
... Adding agent can be symmetrical or asymmetrical. Ex. H2 vs. HCl Symmetrical: H2, C12, Br2, and I2 (I2 slow and readily reversible) Asymmetrical: HCl, RBr, HOH (H2O) Addition of symmetrical agents may be anti or syn depending on mechanism or catalyst. Addition of asymmetrical agents follows Markovnik ...
... Adding agent can be symmetrical or asymmetrical. Ex. H2 vs. HCl Symmetrical: H2, C12, Br2, and I2 (I2 slow and readily reversible) Asymmetrical: HCl, RBr, HOH (H2O) Addition of symmetrical agents may be anti or syn depending on mechanism or catalyst. Addition of asymmetrical agents follows Markovnik ...
Baylis–Hillman reaction

The Baylis–Hillman reaction is a carbon-carbon bond forming reaction between the α-position of an activated alkene and an aldehyde, or generally a carbon electrophile. Employing a nucleophilic catalyst, such as tertiary amine and phosphine, this reaction provides a densely functionalized product (e.g. functionalized allyl alcohol in the case of aldehyde as the electrophile). This reaction is also known as the Morita–Baylis–Hillman reaction or MBH reaction. It is named for the Japanese chemist Ken-ichi Morita, the British chemist Anthony B. Baylis and the German chemist Melville E. D. Hillman.DABCO is one of the most frequently used tertiary amine catalysts for this reaction. In addition, nucleophilic amines such as DMAP and DBU as well as phosphines have been found to successfully catalyze this reaction.MBH reaction has several advantages as a useful synthetic method: 1) It is an atom-economic coupling of easily prepared starting materials. 2) Reaction of a pro-chiral electrophile generates a chiral center, therefore an asymmetric synthesis is possible. 3) Reaction products usually contain multiple functionalities in a proximity so that a variety of further transformations are possible. 4) It can employ a nucleophilic organo-catalytic system without the use of heavy metal under mild conditions.Several reviews have been written.