
Full-Text PDF
... During the last three decades, thousands of papers have presented and described the olefin metathesis catalysis, by experimental synthesis and characterization [1], as well as in the validation of computational protocols [2–9]. However, neither a general catalyst for any metathesis reaction [10–12] ...
... During the last three decades, thousands of papers have presented and described the olefin metathesis catalysis, by experimental synthesis and characterization [1], as well as in the validation of computational protocols [2–9]. However, neither a general catalyst for any metathesis reaction [10–12] ...
Nucleophilic ring opening of aziridines
... their pharmacological effects in recent years has triggered considerable attention in asymmetric synthesis of these amines. One representative procedure is outlined in Scheme 1 using N-tosyl aziridines 1 derived from optically active amino acids as effective templates to undergo nucleophilic ring op ...
... their pharmacological effects in recent years has triggered considerable attention in asymmetric synthesis of these amines. One representative procedure is outlined in Scheme 1 using N-tosyl aziridines 1 derived from optically active amino acids as effective templates to undergo nucleophilic ring op ...
Lorell Thesis Final Version in PDF S
... overall. To my parents José and Betzaida, who always supported me in all aspects of life and made me what I am today. To my brother, José and sisters, Cheryl and Lorna for being there for me and giving me the unconditional love that only brothers can give. To my parents in law, Hilda and Luis, for a ...
... overall. To my parents José and Betzaida, who always supported me in all aspects of life and made me what I am today. To my brother, José and sisters, Cheryl and Lorna for being there for me and giving me the unconditional love that only brothers can give. To my parents in law, Hilda and Luis, for a ...
- University of Bath Opus
... After the initial reports it was almost 20 years before new catalysts appeared to improve and expand the scope of this type of C-N bond formation. Yamaguchi and Fujita were the first, demonstrating that [Cp*IrCl2]2, in the presence of an activating base, was capable of cyclizing amino alcohols to fo ...
... After the initial reports it was almost 20 years before new catalysts appeared to improve and expand the scope of this type of C-N bond formation. Yamaguchi and Fujita were the first, demonstrating that [Cp*IrCl2]2, in the presence of an activating base, was capable of cyclizing amino alcohols to fo ...
Proofs to - Research Explorer
... impeding the formation of an alkoxycarbene by-product [22b]. The reaction of [FeCl(dppe)Cp*] with HC≡CSiMe3 in refluxing MeOH has also been investigated [23] but again, only the vinylidene [Fe(=C=CH2)(dppe)Cp*][PF6] was isolated, consistent with the increased steric demand of the Cp* ring. The react ...
... impeding the formation of an alkoxycarbene by-product [22b]. The reaction of [FeCl(dppe)Cp*] with HC≡CSiMe3 in refluxing MeOH has also been investigated [23] but again, only the vinylidene [Fe(=C=CH2)(dppe)Cp*][PF6] was isolated, consistent with the increased steric demand of the Cp* ring. The react ...
Chapter 4 Alcohols and Alkyl Halides
... Step One: The SN1 mechanism under consideration here involves three separate steps. Step one is proton transfer from the hydrochloric acid to the oxygen lone pair of the alcohol. We protonate the hydroxyl group to make it into a good leaving group in step 2. CH3 CH3 ...
... Step One: The SN1 mechanism under consideration here involves three separate steps. Step one is proton transfer from the hydrochloric acid to the oxygen lone pair of the alcohol. We protonate the hydroxyl group to make it into a good leaving group in step 2. CH3 CH3 ...
Application of Novel Phosphine Ligands in Palladium
... rate-determining for the whole reaction. The third part of catalyzed reactions constitutes the transformations promoted by biomolecules, namely enzymes. Thus, nature can be considered as the world’s leading catalyst designer providing the most selective, active and complex catalysts. Biocatalysis is ...
... rate-determining for the whole reaction. The third part of catalyzed reactions constitutes the transformations promoted by biomolecules, namely enzymes. Thus, nature can be considered as the world’s leading catalyst designer providing the most selective, active and complex catalysts. Biocatalysis is ...
Efficient hydrogenation of organic carbonates, carbamates and
... recently, rare examples of the significantly more difficult hydrogenation of esters4,5 and amides6,7 have also been reported. However, the hydrogenation of organic carbonates and carbamates remains a major challenge. Indeed, as far as we know, catalytic hydrogenation of these important families of com ...
... recently, rare examples of the significantly more difficult hydrogenation of esters4,5 and amides6,7 have also been reported. However, the hydrogenation of organic carbonates and carbamates remains a major challenge. Indeed, as far as we know, catalytic hydrogenation of these important families of com ...
Document
... • When an OH group is bonded to a ring, the ring is numbered beginning with the OH group. • Because the functional group is at C1, the 1 is usually omitted from the name. • The ring is then numbered in a clockwise or counterclockwise fashion to give the next substituent the lowest number. ...
... • When an OH group is bonded to a ring, the ring is numbered beginning with the OH group. • Because the functional group is at C1, the 1 is usually omitted from the name. • The ring is then numbered in a clockwise or counterclockwise fashion to give the next substituent the lowest number. ...
Chapter 9 - ComSizo.com.br
... The outcome is that racemic mixture of (2S,3R) and (2R,3S). The reason is that: the first step can occur equally well from either face of the double bond, to give a 50:50 mixture of enantiomeric bromonium ions. Because the bromonium ions do not have symmetry planes, reaction on the left and right is ...
... The outcome is that racemic mixture of (2S,3R) and (2R,3S). The reason is that: the first step can occur equally well from either face of the double bond, to give a 50:50 mixture of enantiomeric bromonium ions. Because the bromonium ions do not have symmetry planes, reaction on the left and right is ...
aa-2005-38-71-negishi - University of Windsor
... cross-coupling tandem reaction were also reported in 1978.23 The use of Zn salts, such as ZnCl2 or ZnBr2, as additives or cocatalysts in the coupling step of this tandem reaction was shown to be highly desirable or even essential to observing satisfactory results. This study demonstrated, for the fi ...
... cross-coupling tandem reaction were also reported in 1978.23 The use of Zn salts, such as ZnCl2 or ZnBr2, as additives or cocatalysts in the coupling step of this tandem reaction was shown to be highly desirable or even essential to observing satisfactory results. This study demonstrated, for the fi ...
Comprehensive Organic Reactions in Aqueous Media
... body is roughly 65% water by weight. Some organs like the brain and lungs are composed of nearly 80% water.3 Water is the basis and bearer of life. For millions of years, water had been at work to prepare the earth for the evolution of life. It is the solvent in which numerous biochemical organic re ...
... body is roughly 65% water by weight. Some organs like the brain and lungs are composed of nearly 80% water.3 Water is the basis and bearer of life. For millions of years, water had been at work to prepare the earth for the evolution of life. It is the solvent in which numerous biochemical organic re ...
New Stereoselective Approaches to Highly Substituted
... why cyclisations o f (Z)-olefins 27a are generally slower than the (£)-olefins 27b. With a (Z)-olefin 27a, this substituent is in the axial position which presumably permits the intermolecular attack by small amounts o f water (arising from the neutralisation of hydrogen iodide by the base), to comp ...
... why cyclisations o f (Z)-olefins 27a are generally slower than the (£)-olefins 27b. With a (Z)-olefin 27a, this substituent is in the axial position which presumably permits the intermolecular attack by small amounts o f water (arising from the neutralisation of hydrogen iodide by the base), to comp ...
Chapter 17 Amines
... Secondary amines: both aryl amines and alkyl amines react with nitrous acid to yield N-nitrosoamines(亚硝胺) N O NHCH3 + NaNO2 + 2 HCl ...
... Secondary amines: both aryl amines and alkyl amines react with nitrous acid to yield N-nitrosoamines(亚硝胺) N O NHCH3 + NaNO2 + 2 HCl ...
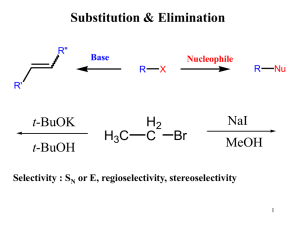
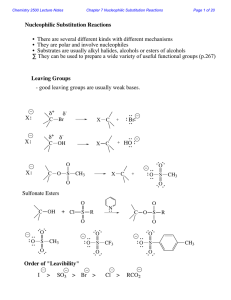
Nucleophilic Substitution Reactions
... Two mechanisms apply to most substitution reactions: SN1 and SN2 SN1: Substitution, nucleophilic, unimolecular SN2: Substitution, nucleophilic, bimolecular Which mechanism applies depends on the structure of your substrate. ...
... Two mechanisms apply to most substitution reactions: SN1 and SN2 SN1: Substitution, nucleophilic, unimolecular SN2: Substitution, nucleophilic, bimolecular Which mechanism applies depends on the structure of your substrate. ...
Three-dimensional Arrangement Of Atoms
... Addition of HBr to a chiral alkene: reactions of a chiral reactant with an achiral reagent gives diastereomeric products which may or may not be formed in equal amounts. The intermediate carbocation is asymmetric, therefore attack of Br- from the top or bottom faces may not be equally probable. The ...
... Addition of HBr to a chiral alkene: reactions of a chiral reactant with an achiral reagent gives diastereomeric products which may or may not be formed in equal amounts. The intermediate carbocation is asymmetric, therefore attack of Br- from the top or bottom faces may not be equally probable. The ...
Aldehydes and ketones
... the addition of R-OH to form an “acetal” the First step is the formation of a “hemiacetal” This is when an -OH and -OR are both on same C O CH3 CH + CH3 CH2 OH acetaldehyde ...
... the addition of R-OH to form an “acetal” the First step is the formation of a “hemiacetal” This is when an -OH and -OR are both on same C O CH3 CH + CH3 CH2 OH acetaldehyde ...
Nucleophilic Acyl Substitution
... The substituents attached to the acyl group strongly affect the reactivity of carbonyl compounds. Carbonyl compounds can be divided into two classes. Class I carbonyl compounds are those in which the acyl group is attached to an atom or a group that can be replaced by another group. Carboxylic acids ...
... The substituents attached to the acyl group strongly affect the reactivity of carbonyl compounds. Carbonyl compounds can be divided into two classes. Class I carbonyl compounds are those in which the acyl group is attached to an atom or a group that can be replaced by another group. Carboxylic acids ...
synthetic.applicatio..
... amines with predictable α and β stereochemistry. The increasing synthetic accessibility of chiral aziridines5−7 has propelled their use in ring opening reactions in organic synthesis. In general, two types of aziridine can be considered: activated and unactivated. The former contain substituents cap ...
... amines with predictable α and β stereochemistry. The increasing synthetic accessibility of chiral aziridines5−7 has propelled their use in ring opening reactions in organic synthesis. In general, two types of aziridine can be considered: activated and unactivated. The former contain substituents cap ...
CH221 CLASS 13
... Hydration of Alkenes Addition of water to alkenes to give alcohols is one of the most important reactions of alkenes. In industry, this is accomplished by the use of strong acid catalysts and high temperatures, but this is not really of much value in the laboratory. However, tertiary alcohols can b ...
... Hydration of Alkenes Addition of water to alkenes to give alcohols is one of the most important reactions of alkenes. In industry, this is accomplished by the use of strong acid catalysts and high temperatures, but this is not really of much value in the laboratory. However, tertiary alcohols can b ...
The story of V
... do not have left reactive groups in the matrix: no reactive end groups (-OH, -COOH), no unreacted carboncarbon double bonds (= unsaturated, (vinyl group = ethenyl group = unsaturated end-group)). Vinyl-Ester (VE) systems, like most resin systems, shrink during the curing (polymerisation). The volume ...
... do not have left reactive groups in the matrix: no reactive end groups (-OH, -COOH), no unreacted carboncarbon double bonds (= unsaturated, (vinyl group = ethenyl group = unsaturated end-group)). Vinyl-Ester (VE) systems, like most resin systems, shrink during the curing (polymerisation). The volume ...
Baylis–Hillman reaction

The Baylis–Hillman reaction is a carbon-carbon bond forming reaction between the α-position of an activated alkene and an aldehyde, or generally a carbon electrophile. Employing a nucleophilic catalyst, such as tertiary amine and phosphine, this reaction provides a densely functionalized product (e.g. functionalized allyl alcohol in the case of aldehyde as the electrophile). This reaction is also known as the Morita–Baylis–Hillman reaction or MBH reaction. It is named for the Japanese chemist Ken-ichi Morita, the British chemist Anthony B. Baylis and the German chemist Melville E. D. Hillman.DABCO is one of the most frequently used tertiary amine catalysts for this reaction. In addition, nucleophilic amines such as DMAP and DBU as well as phosphines have been found to successfully catalyze this reaction.MBH reaction has several advantages as a useful synthetic method: 1) It is an atom-economic coupling of easily prepared starting materials. 2) Reaction of a pro-chiral electrophile generates a chiral center, therefore an asymmetric synthesis is possible. 3) Reaction products usually contain multiple functionalities in a proximity so that a variety of further transformations are possible. 4) It can employ a nucleophilic organo-catalytic system without the use of heavy metal under mild conditions.Several reviews have been written.












![[Ru(Triphos)H2(CO)] Characterisation - Durham e](http://s1.studyres.com/store/data/017676948_1-4352644236c53cc416f065328f560d26-300x300.png)







