Introduction and Chemistry (Ch1 2)
... compounds are soluble in water – Why? – What group might be the exception? ...
... compounds are soluble in water – Why? – What group might be the exception? ...
2.3: Carbon-Based Molecules
... Structure of Lipids • Fatty acids bonded to glycerol. – Glycerol-3 hydroxyl groups – Fatty acids-long carbon chains Two different types of fatty acids found in fats and oils: oSaturated fatty acids oUnsaturated fatty acids ...
... Structure of Lipids • Fatty acids bonded to glycerol. – Glycerol-3 hydroxyl groups – Fatty acids-long carbon chains Two different types of fatty acids found in fats and oils: oSaturated fatty acids oUnsaturated fatty acids ...
Biochemistry of Cells
... Substitution of one amino acid for another in hemoglobin causes sickle-cell disease ...
... Substitution of one amino acid for another in hemoglobin causes sickle-cell disease ...
Chapter 3 Review Questions
... 1. Which statement correctly describes how carbon’s ability to form four bonds makes it uniquely suited to form macromolecules? A. It forms short, simple carbon chains. B. It forms large, complex, diverse molecules. C. It forms covalent bonds with other carbon atoms. D. It forms covalent bonds that ...
... 1. Which statement correctly describes how carbon’s ability to form four bonds makes it uniquely suited to form macromolecules? A. It forms short, simple carbon chains. B. It forms large, complex, diverse molecules. C. It forms covalent bonds with other carbon atoms. D. It forms covalent bonds that ...
Chapter 1_summary notes
... Note: Lipids are not polymers as they are composed of distinct chemical groups of atoms. Monomers link together when the hydroxyl (-OH) group of one monomer reacts with the hydrogen of another monomer, forming a water molecule. This reaction is called condensation polymerisation. ...
... Note: Lipids are not polymers as they are composed of distinct chemical groups of atoms. Monomers link together when the hydroxyl (-OH) group of one monomer reacts with the hydrogen of another monomer, forming a water molecule. This reaction is called condensation polymerisation. ...
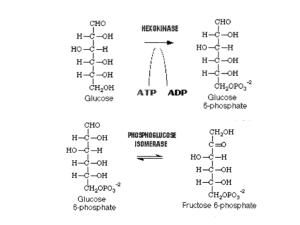
Glycolysis
... Energy for the body • Trapped in chemical bonds of fats, proteins, and carbs (potential) • liberate energy – break bonds – release energy, CO2 and H20 – Energy is transferred to ATP for use in the body ...
... Energy for the body • Trapped in chemical bonds of fats, proteins, and carbs (potential) • liberate energy – break bonds – release energy, CO2 and H20 – Energy is transferred to ATP for use in the body ...
Bio A
... Carbohydrates are made up of monomers called saccharides, that combine to make polysaccharides Saccaride rings can be formed into long straight or branched chains Carbohydrates can be considered simple or complex Simple carbohydrates are smaller molecules that are generally some type of sug ...
... Carbohydrates are made up of monomers called saccharides, that combine to make polysaccharides Saccaride rings can be formed into long straight or branched chains Carbohydrates can be considered simple or complex Simple carbohydrates are smaller molecules that are generally some type of sug ...
Elements Found in Living Things
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
MM Handouts
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
Elements Found in Living Things
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
Most common elements in living things are carbon, hydrogen
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
Most common elements in living things are carbon, hydrogen
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
... nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be classified in two broad categories --- organic and inorganic compounds. Organic compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form si ...
Glycolysis in the Cytoplasm
... 1. Glycolysis I - Energy Investment Phase ATP is used to split the 6-carbon molecule into two 3-carbon molecules 2. Glycolysis II - Energy Payoff Phase 2 ATP are produced and 2 molecules of the electron carrier NADH + H+ along with 2 molecules of pyruvate Pyruvate is an important branch point in met ...
... 1. Glycolysis I - Energy Investment Phase ATP is used to split the 6-carbon molecule into two 3-carbon molecules 2. Glycolysis II - Energy Payoff Phase 2 ATP are produced and 2 molecules of the electron carrier NADH + H+ along with 2 molecules of pyruvate Pyruvate is an important branch point in met ...
A Guided Reading on Macromolecules
... The four main classes of organic compounds (carbohydrates, lipids, proteins, and nucleic acids) that are essential to the proper functioning of all living things are known as polymers or macromolecules. All of these compounds are built primarily of carbon, hydrogen, and oxygen but in different ratio ...
... The four main classes of organic compounds (carbohydrates, lipids, proteins, and nucleic acids) that are essential to the proper functioning of all living things are known as polymers or macromolecules. All of these compounds are built primarily of carbon, hydrogen, and oxygen but in different ratio ...
Chapter 3
... -mainly C and H. Contain very little O. -source of long term energy storage for the body -lipids have 6x as much energy as carbs but are much harder for the body to break down. -also serve as protective layers (think whale blubber) and precursors of larger molecules -are hydrophobic…meaning they fea ...
... -mainly C and H. Contain very little O. -source of long term energy storage for the body -lipids have 6x as much energy as carbs but are much harder for the body to break down. -also serve as protective layers (think whale blubber) and precursors of larger molecules -are hydrophobic…meaning they fea ...
Nucleic acids
... Another important nucleic acid in biology is called Adenosine Triphosphate or ATP for short. ATP is the energy source of the bodies cells. ATP has three phosphates. Anytime the body needs energy, one of the phosphates are released from ATP, releasing energy and making a free phosphate and ADP (adeno ...
... Another important nucleic acid in biology is called Adenosine Triphosphate or ATP for short. ATP is the energy source of the bodies cells. ATP has three phosphates. Anytime the body needs energy, one of the phosphates are released from ATP, releasing energy and making a free phosphate and ADP (adeno ...
The Structure and Function of Large Biological Molecules
... digest β linkages, but promotes healthy digestion – Most abundant organic compound on Earth ...
... digest β linkages, but promotes healthy digestion – Most abundant organic compound on Earth ...
Nucleic acids
... Adenosine Triphosphate or ATP for short. ATP is the energy source of the bodies cells. ATP has three phosphates. Anytime the body needs energy, one of the phosphates are released from ATP, releasing energy and making a free phosphate and ADP (adenosine diphosphate) molecule. When energy is absorbed, ...
... Adenosine Triphosphate or ATP for short. ATP is the energy source of the bodies cells. ATP has three phosphates. Anytime the body needs energy, one of the phosphates are released from ATP, releasing energy and making a free phosphate and ADP (adenosine diphosphate) molecule. When energy is absorbed, ...
Organic Chemistry I. Organic compounds
... An amino group (NH2) 2. An acid (carboxyl) group (COOH) 3. Each of the twenty different amino acids have a different side chain (R) on their central carbon. ...
... An amino group (NH2) 2. An acid (carboxyl) group (COOH) 3. Each of the twenty different amino acids have a different side chain (R) on their central carbon. ...
Chapter 5: PROTEINS
... ● amino acids differ from each other at their “side” or “R” chains ● because they are so different, and can be put together in almost infinite combinations, proteins are among ...
... ● amino acids differ from each other at their “side” or “R” chains ● because they are so different, and can be put together in almost infinite combinations, proteins are among ...
Metabolism08
... To begin breaking down fat, the body breaks triglycerides into glycerol and fatty acids Most of the energy is stored in the fatty acids (glycerol can be converted to glucose or pyruvate) Fatty acids usually produce substantially more ATP than glucose (16 carbon fatty acid = 129 ATP) ...
... To begin breaking down fat, the body breaks triglycerides into glycerol and fatty acids Most of the energy is stored in the fatty acids (glycerol can be converted to glucose or pyruvate) Fatty acids usually produce substantially more ATP than glucose (16 carbon fatty acid = 129 ATP) ...
Lecture 1d Plant Diversity, Basic Chemistry
... loving). • A Nonpolar substance then lacks any charges and will not be able to interact with water. Nonpolar molecules are said to be Hydrophobic (water hating). ...
... loving). • A Nonpolar substance then lacks any charges and will not be able to interact with water. Nonpolar molecules are said to be Hydrophobic (water hating). ...
Macromolecules Quiz 1
... 2. A fat molecule with one double carbon bond is? a) Saturated b) Unsaturated 3. Match the level of protein structure with the description Primary ...
... 2. A fat molecule with one double carbon bond is? a) Saturated b) Unsaturated 3. Match the level of protein structure with the description Primary ...
Chemistry Of Life
... contained in the sequence of the bases. •Carries the information required for the growth and reproduction of all cells. •Stable, resistant to degradation. ...
... contained in the sequence of the bases. •Carries the information required for the growth and reproduction of all cells. •Stable, resistant to degradation. ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.