Biomolecule Notes
... A carbon atom can form chemical bonds with other carbon atoms in long chains or rings. Carbon Compounds in living things include: Carbohydrates, lipids, nucleic acids, and proteins (these are called macromolecules). ...
... A carbon atom can form chemical bonds with other carbon atoms in long chains or rings. Carbon Compounds in living things include: Carbohydrates, lipids, nucleic acids, and proteins (these are called macromolecules). ...
Carbon Compounds
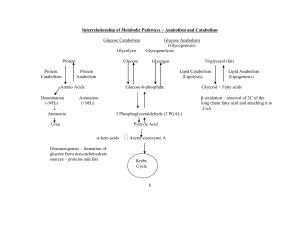
... – Glycogen stored in your liver is released when glucose in your blood runs low. ...
... – Glycogen stored in your liver is released when glucose in your blood runs low. ...
Macromolecule Notes
... Macromolecule Notes Macromolecules: Proteins, Lipids, Polysaccharides (Carbohydrates) and Nucleic Acids Monomer: single building block for a macromolecule Proteins (polypeptides) Monomer: amino acid (a.a.) Compound in your body with nitrogen, carbon, oxygen and hydrogen 20 essential amino acid ...
... Macromolecule Notes Macromolecules: Proteins, Lipids, Polysaccharides (Carbohydrates) and Nucleic Acids Monomer: single building block for a macromolecule Proteins (polypeptides) Monomer: amino acid (a.a.) Compound in your body with nitrogen, carbon, oxygen and hydrogen 20 essential amino acid ...
Lab Time
... Hydrolysis of triglycerides within adipose tissue releases glycerol and free fatty acids into the blood which are both used as an energy source by many organs; they can also be converted by the liver into derivatives called ketone bodies which circulate through the blood and used as an energy source ...
... Hydrolysis of triglycerides within adipose tissue releases glycerol and free fatty acids into the blood which are both used as an energy source by many organs; they can also be converted by the liver into derivatives called ketone bodies which circulate through the blood and used as an energy source ...
Macromolecules Quiz
... Matching--Select the macromolecule that best matches the statement. Letters may be used once, more than once or not at all. a. Proteins b. Carbohydrates c. Lipids d. Nucleic Acids 1. These macromolecules possess large nonpolar regions making them insoluble in water. 2. This macromolecule is compose ...
... Matching--Select the macromolecule that best matches the statement. Letters may be used once, more than once or not at all. a. Proteins b. Carbohydrates c. Lipids d. Nucleic Acids 1. These macromolecules possess large nonpolar regions making them insoluble in water. 2. This macromolecule is compose ...
Nucleic Acids
... contain an amino group (-NH2), a carboxyl group (-COOH) and a hydrogen atom, all bonded to a central carbon atom – twenty common AA grouped into five classes based on side groups nonpolar AA ...
... contain an amino group (-NH2), a carboxyl group (-COOH) and a hydrogen atom, all bonded to a central carbon atom – twenty common AA grouped into five classes based on side groups nonpolar AA ...
Teacher Quality Grant - Gulf Coast State College
... Items referring to the role of enzymes as catalysts will use a biological context and not require knowledge of specific enzymes. Items referring to the factors that affect enzyme activity are limited to concentration, pH, and temperature. Items will not require specific knowledge of how an enzyme re ...
... Items referring to the role of enzymes as catalysts will use a biological context and not require knowledge of specific enzymes. Items referring to the factors that affect enzyme activity are limited to concentration, pH, and temperature. Items will not require specific knowledge of how an enzyme re ...
THE Macromolecules PowerPoint - Panhandle Area Educational
... Items referring to the role of enzymes as catalysts will use a biological context and not require knowledge of specific enzymes. Items referring to the factors that affect enzyme activity are limited to concentration, pH, and temperature. Items will not require specific knowledge of how an enzyme re ...
... Items referring to the role of enzymes as catalysts will use a biological context and not require knowledge of specific enzymes. Items referring to the factors that affect enzyme activity are limited to concentration, pH, and temperature. Items will not require specific knowledge of how an enzyme re ...
C383 Study Guide for the Final Exam Spring 2017 Basic Information
... molecule that you store in your liver. Circle the pathways/cycles below that are part of this overall transformation. Cross out any that are not. Gluconeogenesis, pentose phosphate pathway, glycogen synthesis, glycolysis, citric acid cycle B. Trace the metabolic path of this glutamate molecule throu ...
... molecule that you store in your liver. Circle the pathways/cycles below that are part of this overall transformation. Cross out any that are not. Gluconeogenesis, pentose phosphate pathway, glycogen synthesis, glycolysis, citric acid cycle B. Trace the metabolic path of this glutamate molecule throu ...
Biochemistry Note
... Macromolecules – Large organic molecules that contain dozens of carbon atoms and many functional groups. There are four major groups of biologically important macromolecules: ...
... Macromolecules – Large organic molecules that contain dozens of carbon atoms and many functional groups. There are four major groups of biologically important macromolecules: ...
Biochemistry Test Review (Vocabulary on the back page
... B2.2 Organic Molecules There are four major categories of organic molecules that make up living systems: carbohydrates, fats, proteins, and nucleic acids. B2.5 Living Organism Composition All living or once-living organisms are composed of carbohydrates, lipids, proteins, and nucleic acids. Carbohyd ...
... B2.2 Organic Molecules There are four major categories of organic molecules that make up living systems: carbohydrates, fats, proteins, and nucleic acids. B2.5 Living Organism Composition All living or once-living organisms are composed of carbohydrates, lipids, proteins, and nucleic acids. Carbohyd ...
Macromolecules biologyjunction
... oxygen, nitrogen, and phosphorus (CHON P). The body also needs trace amounts of other elements such as calcium, potassium, and sulfur for proper functioning of ...
... oxygen, nitrogen, and phosphorus (CHON P). The body also needs trace amounts of other elements such as calcium, potassium, and sulfur for proper functioning of ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034 Part A
... 6. Differentiate LDL from HDL. 7. What is chemi-osmotic hypothesis? 8. Comment on auto oxidation. 9. Write a note on Zimmermann reaction. 10. What are anti oxidants? ...
... 6. Differentiate LDL from HDL. 7. What is chemi-osmotic hypothesis? 8. Comment on auto oxidation. 9. Write a note on Zimmermann reaction. 10. What are anti oxidants? ...
Chapter 13: Carbohydrates
... Biochemistry Biochemistry is the study of the chemistry of living ...
... Biochemistry Biochemistry is the study of the chemistry of living ...
Most common elements in living things are carbon, hydrogen
... The four main classes of organic compounds (carbohydrates, lipids, proteins, and nucleic acids) that are essential to the proper functioning of all living things are known as polymers or macromolecules. All of these compounds are built primarily of carbon, hydrogen, and oxygen but in different ratio ...
... The four main classes of organic compounds (carbohydrates, lipids, proteins, and nucleic acids) that are essential to the proper functioning of all living things are known as polymers or macromolecules. All of these compounds are built primarily of carbon, hydrogen, and oxygen but in different ratio ...
Chapter 2-1 The Nature of Matter
... iv. saturated (max # of H bonds) and unsaturated (at least one double bond=) c. Nucleic acids i. H, O, N, C, P ii. Polymers of nucleotides – sugar, phosphate, and nitrogen base iii. Store and transmit hereditary or genetic ...
... iv. saturated (max # of H bonds) and unsaturated (at least one double bond=) c. Nucleic acids i. H, O, N, C, P ii. Polymers of nucleotides – sugar, phosphate, and nitrogen base iii. Store and transmit hereditary or genetic ...
Biomolecules Test Review
... _______________________ Pasta contains much of this. _______________________ Stores information in your cells in the form of a code. _______________________ Building block in an amino acid. _______________________ Butter contains much of this. _______________________ Used in the exoskeleton of insec ...
... _______________________ Pasta contains much of this. _______________________ Stores information in your cells in the form of a code. _______________________ Building block in an amino acid. _______________________ Butter contains much of this. _______________________ Used in the exoskeleton of insec ...
Fatty Acids: The lipid building blocks: The common building block for
... in the stomach and further digestion occurs in the small intestine. This process takes the proteins you consume and coverts them into the component amino acids by breaking the covalent bonds which connect the subunits of the proteins. alanine arginine asparagine aspartic acid ...
... in the stomach and further digestion occurs in the small intestine. This process takes the proteins you consume and coverts them into the component amino acids by breaking the covalent bonds which connect the subunits of the proteins. alanine arginine asparagine aspartic acid ...
Enzymes - flickbio
... • Hydrogen bonds – slightly positive, polar H atoms are attracted to polar negative atoms like O ...
... • Hydrogen bonds – slightly positive, polar H atoms are attracted to polar negative atoms like O ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.